Electron shielding is a fundamental concept in atomic physics that profoundly influences the behavior of electrons within atoms and molecules. While not directly a subject within drone technology, flight systems, cameras, accessories, aerial filmmaking, or even broader tech and innovation categories, understanding electron shielding provides crucial foundational knowledge for advancements in materials science, electronics, and sensor development – areas that are indirectly but significantly impacting the technological evolution of all these drone-related fields. For instance, the efficiency and reliability of the microelectronics powering advanced drone navigation, the sensitivity of optical sensors in cameras, and the development of novel battery chemistries are all underpinned by principles of atomic and molecular behavior, where electron shielding plays a vital role.

The Core Concept of Electron Shielding
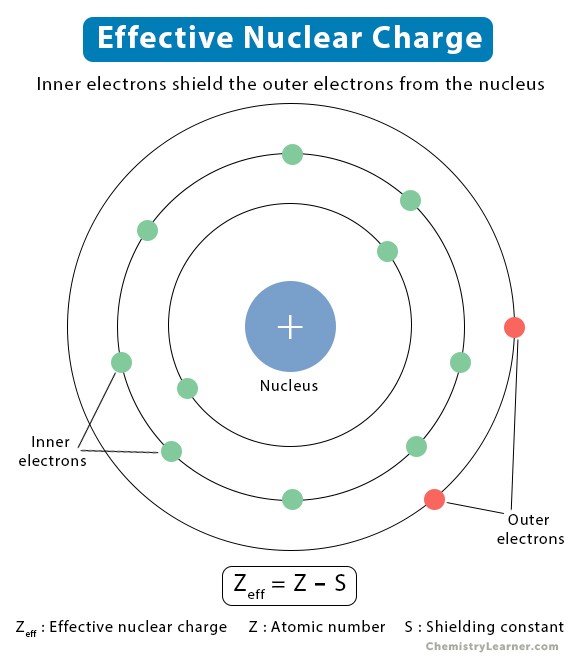
At its heart, electron shielding, also known as atomic screening, describes the phenomenon where inner-shell electrons partially block or “shield” the electrostatic attraction between the nucleus and outer-shell electrons. The nucleus, with its positive charge, exerts a pull on all electrons within the atom. However, the electrons in shells closer to the nucleus experience this pull more directly than those in outer shells. The inner electrons, being between the nucleus and the outer electrons, create a repulsive effect that counteracts the nuclear attraction on the outer electrons. This effectively reduces the net positive charge (the effective nuclear charge) experienced by the outer valence electrons.
The Nucleus and Its Influence
The nucleus of an atom contains protons, which are positively charged, and neutrons, which have no charge. The number of protons determines the atomic number of an element and dictates the total positive charge of the nucleus. This positive charge is the primary force attracting the negatively charged electrons orbiting the nucleus.
Inner vs. Outer Electrons: A Tale of Two Forces
Electrons are arranged in distinct energy levels or shells around the nucleus. The closer an electron is to the nucleus, the stronger the attractive force it experiences. Conversely, electrons in shells farther from the nucleus are less strongly bound. Electron shielding is the consequence of the electron-electron repulsion between electrons in different shells. Imagine a line of people holding hands. The person at the front is directly connected to the person at the back (the nucleus), but the people in the middle (inner electrons) reduce the direct impact of the front person’s grip on the last person in line (outer electrons).
Effective Nuclear Charge ($Z_{eff}$)
The concept of effective nuclear charge ($Z_{eff}$) is a direct result of electron shielding. It represents the net positive charge experienced by an electron in a multi-electron atom. It is always less than the actual nuclear charge ($Z$, the atomic number) because of the shielding effect of the inner electrons. The formula often used to conceptualize this is:
$Z_{eff} = Z – S$
Where:
- $Z$ is the atomic number (the number of protons in the nucleus).
- $S$ is the shielding constant, which represents the average number of electrons between the nucleus and the electron in question. This constant is difficult to calculate precisely and is often estimated using rules like Slater’s rules.
The greater the shielding, the lower the $Z_{eff}$, and thus the weaker the attraction between the nucleus and the outer electron. This has significant implications for various atomic and chemical properties.
Factors Influencing Electron Shielding
The degree of electron shielding is not uniform across all atoms or even within a single atom. Several factors contribute to how effectively inner electrons shield outer electrons.
Shell Number and Penetration
The principal quantum number, or shell number ($n$), is a primary determinant. Electrons in lower shells (smaller $n$) are closer to the nucleus and therefore contribute more significantly to shielding outer electrons. Moreover, the shape of electron orbitals plays a role. Electrons in s orbitals penetrate closer to the nucleus than those in p, d, or f orbitals of the same shell. This means that an electron in an s orbital experiences less shielding and a higher $Z_{eff}$ compared to an electron in a p, d, or f orbital of the same shell, assuming they are at similar distances from the nucleus. This difference in penetration is why subshells within the same shell have different energies.
The Nature of the Shielding Electrons
The electrons that provide shielding are typically those in inner shells. Electrons within the same shell also contribute to shielding, but their effect is less pronounced than that of electrons in inner shells. The repulsive force between electrons in the same subshell is generally considered to be about half as effective as the repulsion from electrons in an inner shell.
Relationship to Atomic Size and Ionization Energy
Electron shielding has a direct impact on atomic radius and ionization energy.
-
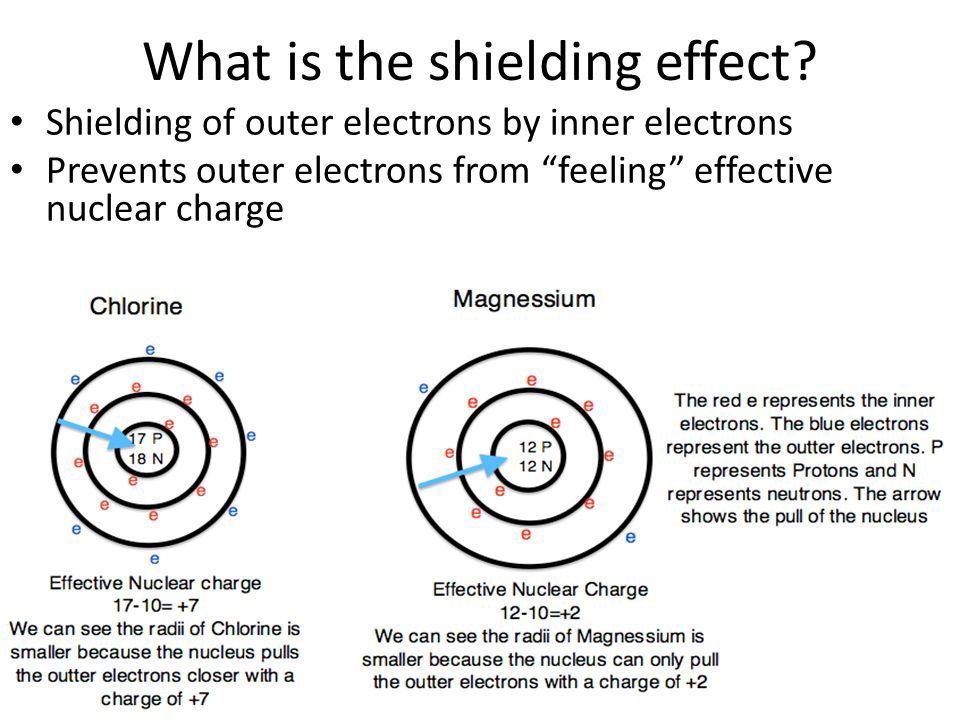
Atomic Radius: As you move down a group in the periodic table, the principal quantum number ($n$) increases, meaning valence electrons are in shells farther from the nucleus. While nuclear charge also increases, the effect of the additional electron shells and the increased shielding from these inner shells outweighs the increased nuclear attraction, leading to larger atomic radii. Conversely, moving across a period, the principal quantum number remains the same, but the nuclear charge increases, and the shielding effect within the same shell is less effective. This results in a stronger attraction between the nucleus and valence electrons, leading to smaller atomic radii.
-
Ionization Energy: Ionization energy is the minimum energy required to remove an electron from an atom in its gaseous state. Electrons that are less tightly bound (due to lower $Z_{eff}$ and greater distance) require less energy to remove. Therefore, elements with significant electron shielding of their valence electrons will have lower ionization energies. This explains why alkali metals (Group 1) are highly reactive; their single valence electron is well-shielded and easily removed.
The Role of Electron Shielding in Chemical Bonding and Reactivity
The effective nuclear charge experienced by valence electrons dictates how readily an atom will participate in chemical reactions and the types of bonds it will form.
Valence Electrons and Chemical Behavior
Valence electrons are the outermost electrons of an atom and are the primary participants in chemical bonding. The extent to which these valence electrons are shielded by inner electrons is a critical factor in determining an atom’s chemical reactivity.
-
High Shielding: If valence electrons are heavily shielded by inner electrons, they are less attracted to the nucleus. This makes them more easily removed, leading to a tendency to form positive ions (cations) and participate in ionic bonding. Elements with low ionization energies, due to effective shielding, are typically metals found on the left side of the periodic table.
-
Low Shielding: If valence electrons experience a strong attraction to the nucleus (low shielding), they are less likely to be lost. Instead, these atoms tend to gain electrons to achieve a stable electron configuration or share electrons to form covalent bonds. Elements with high ionization energies, due to less effective shielding, are typically nonmetals found on the right side of the periodic table.
Electronegativity and Electron Shielding
Electronegativity, the measure of an atom’s ability to attract shared electrons in a chemical bond, is also influenced by electron shielding. Atoms with a higher effective nuclear charge experienced by their valence electrons tend to be more electronegative because the nucleus exerts a stronger pull on electrons, both its own and those in a shared bond. Therefore, elements with less shielding and higher $Z_{eff}$ values for their valence electrons generally exhibit higher electronegativity.
Electron Shielding in Materials Science and Beyond
While the direct application of “electron shielding” as a term might not be prevalent in every field, the underlying principles are crucial for understanding the properties of materials used in advanced technologies.
Semiconductor Properties
The performance of semiconductors, the backbone of modern electronics (including those in drones), is dictated by the electronic band structure, which is a direct consequence of how electrons interact within the crystal lattice. Electron shielding within individual atoms and the resulting electron-electron repulsions influence the energy levels and the delocalization of electrons, which in turn defines whether a material is a conductor, semiconductor, or insulator. Understanding these atomic-level interactions is essential for designing and fabricating more efficient microprocessors, sensors, and power management systems.
Development of Advanced Sensors
Many sensors used in flight technology and aerial imaging rely on the interaction of electromagnetic radiation or chemical species with materials. The response of these sensors is often tied to the electronic configuration and bonding of the sensing material. Electron shielding affects the energy levels of electrons within the sensing material, influencing its absorption and emission spectra, its conductivity changes in response to stimuli, and its overall sensitivity and selectivity. For example, in optical sensors used for imaging, the way light interacts with the material is fundamentally linked to electron behavior, which is modulated by shielding effects.
Battery Technology and Energy Storage
The development of advanced batteries with higher energy density and faster charging capabilities hinges on understanding electrochemical processes at the atomic and molecular level. Electron shielding plays a role in the stability of electrode materials and the ease with which ions and electrons can move within the battery. For instance, the way lithium ions interact with the cathode and anode materials is influenced by the electronic environment, where electron shielding contributes to the overall potential energy landscape and kinetics of charge transfer.
Future Innovations
As we push the boundaries of technology, the need for deeper understanding of fundamental physics becomes increasingly important. From developing next-generation materials for lighter and stronger drone frames to creating more sophisticated AI-driven flight control systems that can adapt to complex environments, the underlying physics of electron behavior, including electron shielding, remains a critical, albeit often implicit, foundation for innovation. Advances in quantum computing, for instance, are entirely predicated on manipulating quantum states of electrons, where the precise understanding of their interactions, including shielding, is paramount.
In conclusion, while “electron shielding” might sound like an esoteric concept divorced from the tangible world of drones, its principles are woven into the fabric of the very technologies that make modern aerial platforms possible. From the microchips that govern flight to the sensors that capture breathtaking imagery, the understanding of how electrons interact and influence each other is a fundamental building block of technological progress.
