Transmembrane proteins are specialized proteins that span the entire biological membrane, serving as critical conduits and mediators between a cell’s interior and its external environment. Integral to virtually all cellular processes, these proteins are deeply embedded within or pass completely through the lipid bilayer, making them uniquely positioned to facilitate communication, transport, and signal transduction across the otherwise impermeable membrane barrier. Their sophisticated architecture and diverse functionalities underpin the very essence of cellular life, from nutrient uptake and waste expulsion to sensing external cues and orchestrating complex intracellular responses. Understanding transmembrane proteins is fundamental to grasping cellular physiology, and their pivotal roles make them central figures in contemporary biotechnology and medical innovation.
The Fundamental Architecture of Life
The defining characteristic of a transmembrane protein is its ability to interact simultaneously with both the hydrophobic core of the lipid bilayer and the aqueous environments on either side. This dual affinity dictates their complex structural arrangements and their crucial placement within the cellular membrane. The lipid bilayer itself is a formidable barrier, primarily composed of phospholipids with hydrophilic heads facing outward and hydrophobic tails forming the interior. Transmembrane proteins navigate this environment through specific structural motifs that allow them to integrate seamlessly while maintaining their functional integrity.
Hydrophobic and Hydrophilic Interactions
A transmembrane protein’s structure is a masterclass in accommodating contrasting environments. The segments of the protein that traverse the lipid bilayer are typically rich in hydrophobic amino acids. These nonpolar residues readily interact with the fatty acid tails of the phospholipids, allowing the protein to embed stably within the membrane’s hydrophobic interior. Conversely, the portions of the protein exposed to the aqueous extracellular or intracellular environments are rich in hydrophilic amino acids. These polar and charged residues can form hydrogen bonds and ionic interactions with water molecules and other polar components, ensuring their solubility and functionality in these aqueous phases. This strategic distribution of amino acids is a key determinant of a transmembrane protein’s stability and proper insertion into the membrane.
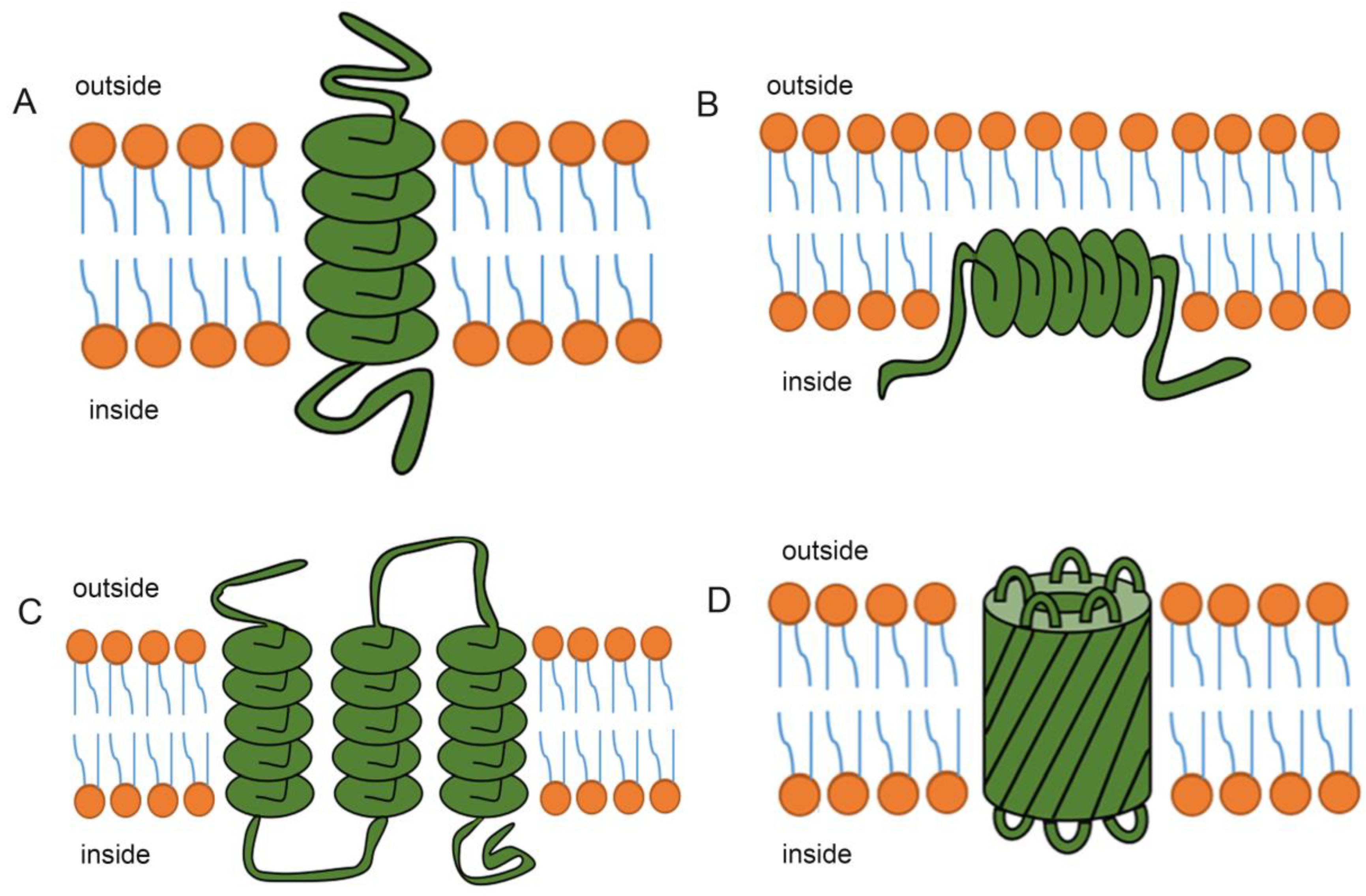
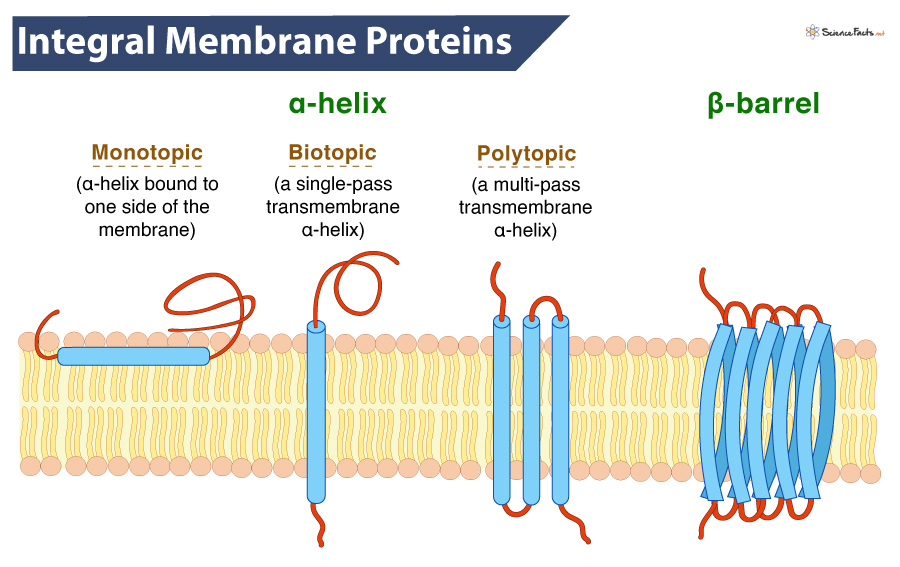
Common Structural Motifs
Transmembrane proteins predominantly adopt one of two main structural motifs to cross the lipid bilayer: alpha-helices or beta-barrels. Alpha-helical bundles are by far the more common structure in eukaryotic cells and span the membrane with one or more helical segments. These helices are typically 20-25 amino acids long, just enough to span the ~30-ångström thickness of the membrane’s hydrophobic core. Multiple alpha-helices can then pack together, forming channels, pores, or receptor complexes. Beta-barrels, on the other hand, are more frequently found in the outer membranes of Gram-negative bacteria, mitochondria, and chloroplasts. They consist of a sheet of beta-strands rolled into a barrel shape, forming a central pore. The side chains facing the hydrophobic lipid interior are typically nonpolar, while those facing the aqueous pore and the periplasmic or cytoplasmic spaces are polar, facilitating the passage of specific molecules. The choice between these motifs depends on the protein’s evolutionary origin, functional requirements, and the specific membrane it inhabits.
Diverse Functions Essential for Cellular Survival
The sheer diversity in structure of transmembrane proteins is mirrored by the extensive range of functions they perform. These proteins are not mere structural components; they are active participants in almost every aspect of cellular existence, acting as gatekeepers, communicators, and catalysts. Their strategic location allows them to mediate interactions that are vital for maintaining cellular homeostasis, responding to stimuli, and coordinating multicellular activities.
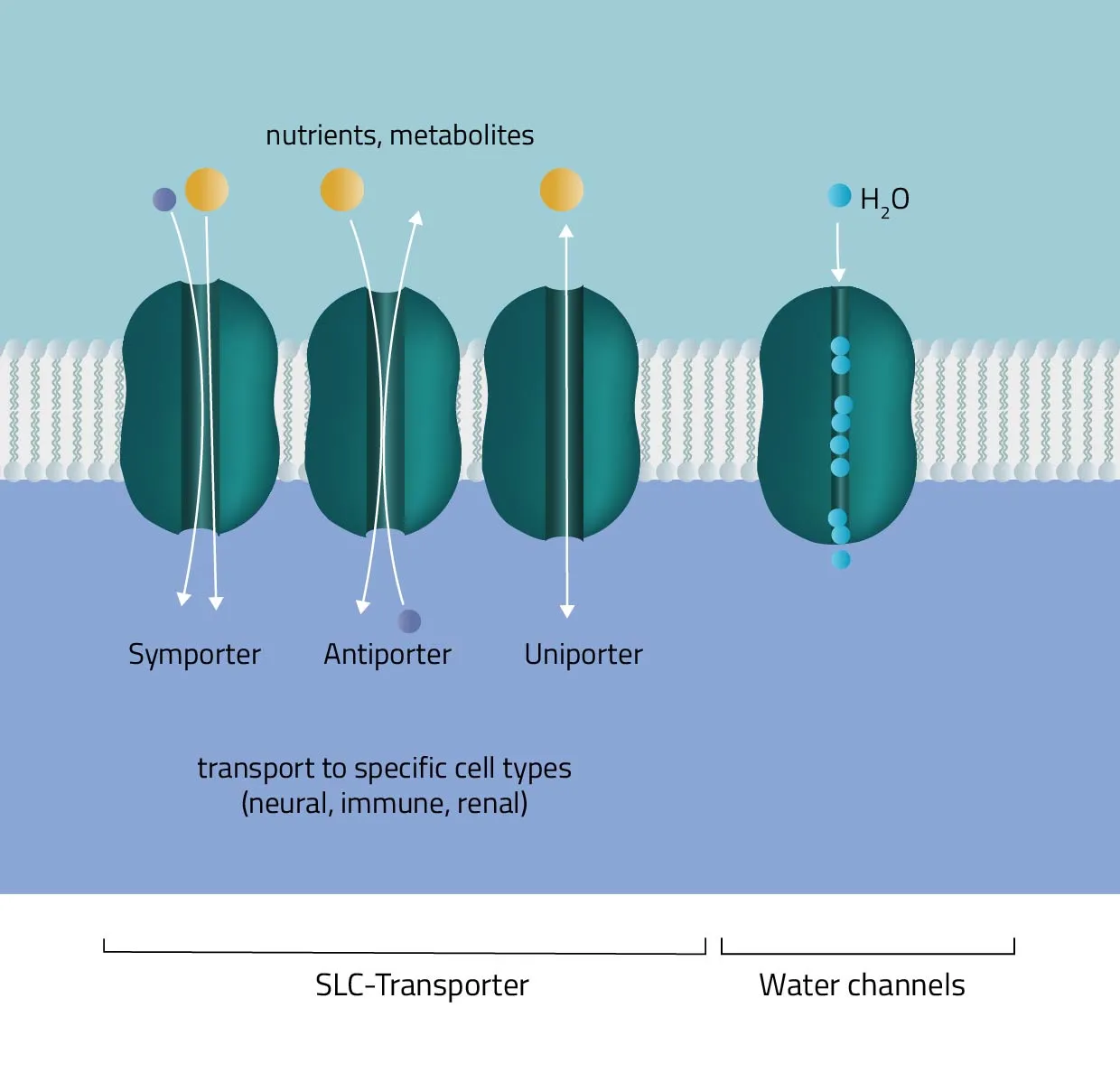
Channels, Pumps, and Transporters
One of the most critical roles of transmembrane proteins is the regulated movement of molecules across the cell membrane. The lipid bilayer is largely impermeable to ions and most polar molecules, necessitating specialized mechanisms for their passage.
- Channels are transmembrane proteins that form hydrophilic pores, allowing specific ions or small molecules to diffuse rapidly down their electrochemical gradient. Ligand-gated channels open in response to chemical signals, while voltage-gated channels respond to changes in membrane potential, playing crucial roles in nerve impulse transmission and muscle contraction.
- Pumps (also known as active transporters) utilize energy, often from ATP hydrolysis, to move ions or molecules against their concentration gradient. The sodium-potassium pump, for instance, is vital for maintaining cell volume and establishing electrochemical gradients essential for nerve function.
- Transporters facilitate the movement of specific solutes across the membrane. They bind their cargo on one side of the membrane, undergo a conformational change, and release it on the other side. This process can be either passive (facilitated diffusion) or active (secondary active transport, using the energy from an ion gradient).
Receptors and Signal Transduction
Cells are constantly bombarded with signals from their environment, and transmembrane receptors are the primary sensors that detect and relay this information into the cell. These proteins have an extracellular domain that specifically binds to signaling molecules (ligands) such as hormones, neurotransmitters, or growth factors. Upon ligand binding, the receptor undergoes a conformational change that triggers a cascade of events within the cell. This signal transduction pathway ultimately leads to specific cellular responses, such as changes in gene expression, metabolism, or cell division. Examples include G protein-coupled receptors (GPCRs), receptor tyrosine kinases (RTKs), and ligand-gated ion channels, all of which are pivotal in physiological regulation and disease pathology.
Enzymes and Structural Support
While transport and signaling are dominant functions, some transmembrane proteins also serve as enzymes, catalyzing specific biochemical reactions directly within the membrane environment. For example, some enzymes involved in lipid synthesis or electron transport chains are membrane-bound. Beyond their active roles, certain transmembrane proteins contribute to the structural integrity of the cell, anchoring the cytoskeleton to the membrane or mediating cell-to-cell adhesion, which is crucial for tissue formation and communication. Integrins, for instance, link the extracellular matrix to the intracellular cytoskeleton, playing key roles in cell migration, differentiation, and tissue repair.
Transmembrane Proteins as Pillars of Biotechnology and Medicine
The profound and multifaceted roles of transmembrane proteins make them exceptionally compelling targets and tools across various fields of technology and innovation. Their intricate involvement in disease pathogenesis and their accessibility on the cell surface position them as central figures in modern drug discovery, diagnostics, and synthetic biology. The ability to manipulate, study, or interact with these proteins offers unprecedented opportunities for advancements in human health and beyond.
Drug Targets and Therapeutic Development
Remarkably, transmembrane proteins are the targets of over 60% of all currently marketed drugs. Their critical roles in signal transduction pathways, nutrient uptake, and ion homeostasis mean that modulating their activity can profoundly impact cellular function. GPCRs, in particular, are the most targeted protein family, playing roles in diverse conditions from hypertension and allergies to pain management and psychiatric disorders. Innovations in structural biology, such as cryo-electron microscopy and X-ray crystallography, are enabling researchers to determine the precise 3D structures of these complex proteins, even in their native membrane environment. This structural information is invaluable for rational drug design, allowing scientists to develop highly specific small molecules or biologics that can activate, inhibit, or modulate the function of disease-relevant transmembrane proteins, leading to more effective therapies with fewer side effects.
Biosensors and Diagnostics
The inherent ability of transmembrane proteins to specifically bind ligands and transduce signals makes them ideal components for advanced biosensing technologies. Engineered receptor proteins can be integrated into synthetic membranes or immobilized surfaces to detect the presence of specific molecules, such as biomarkers for disease, environmental toxins, or even pathogens. For instance, modified ion channels or GPCRs could be designed to produce a measurable electrical or optical signal upon binding a target analyte. This offers the potential for highly sensitive and rapid diagnostic tools, moving towards point-of-care testing and early disease detection. Innovations in microfluidics and nanotechnology are further enhancing the integration of these protein-based sensors into compact, high-throughput diagnostic platforms.
Synthetic Biology and Nanotechnology
Beyond their natural roles, transmembrane proteins are increasingly being utilized and engineered within the realms of synthetic biology and nanotechnology. Researchers are exploring ways to design and synthesize novel transmembrane proteins with custom functionalities. This includes creating artificial channels for targeted drug delivery, developing membrane-bound enzymes for biocatalysis in controlled environments, or even constructing de novo protein assemblies that can interact with and manipulate cellular membranes. In nanotechnology, these proteins can serve as building blocks for creating biomimetic nanostructures, such as vesicles with specific permeabilities or self-assembling protein arrays for advanced material science. The ability to precisely control the insertion and function of these proteins opens avenues for designing artificial cells, advanced bioreactors, and novel drug delivery vehicles.
Future Frontiers in Research and Innovation
The field of transmembrane protein research continues to be a vibrant area of scientific and technological innovation. Advances in experimental techniques, computational modeling, and interdisciplinary approaches are constantly pushing the boundaries of what we understand about these vital molecules and how we can harness their potential for future applications. The complexity of these proteins, often requiring a lipid environment for stability and function, presents unique challenges, but also promises extraordinary rewards for those who can unravel their remaining mysteries.
Structural Biology Breakthroughs
Recent years have witnessed a revolution in structural biology, particularly with the advent of cryo-electron microscopy (cryo-EM). This technology has proven exceptionally powerful for resolving the high-resolution structures of large, dynamic, and membrane-embedded protein complexes that are often recalcitrant to traditional X-ray crystallography. These structural insights are not merely academic; they provide atomic-level blueprints that inform rational drug design, uncover mechanisms of action for existing drugs, and guide the engineering of proteins with enhanced or novel functions. As cryo-EM continues to improve in resolution and accessibility, an ever-increasing number of transmembrane protein structures will be elucidated, driving a new era of targeted therapeutic development.
Precision Medicine and Personalized Therapies
Variations in an individual’s transmembrane proteins can significantly impact their susceptibility to disease and their response to medications. Pharmacogenomics, the study of how genes affect a person’s response to drugs, frequently identifies genetic polymorphisms in transmembrane transporters and receptors that influence drug metabolism, efficacy, and toxicity. Future innovations will leverage this understanding to develop precision medicine approaches. By profiling an individual’s unique set of transmembrane proteins, clinicians could personalize drug dosages, select the most effective therapies, and predict adverse reactions, leading to highly tailored and optimized patient care. This also extends to developing gene therapies or CRISPR-based interventions to correct dysfunctional transmembrane proteins directly.
Engineering Novel Biological Systems
The principles learned from studying natural transmembrane proteins are inspiring the creation of entirely novel biological systems. Synthetic biologists are not just modifying existing proteins but are designing proteins from scratch that can insert into membranes and perform custom tasks. This includes building artificial ion channels that respond to specific light wavelengths for optogenetics, creating membrane-bound logic gates for advanced cellular computing, or designing protein assemblies that can self-organize into complex bioreactors. These ambitious projects aim to engineer cells or even acellular systems with capabilities far beyond their natural counterparts, paving the way for revolutionary advances in areas such as bio-manufacturing, sustainable energy solutions, and advanced medical diagnostics. The journey to fully understand and harness transmembrane proteins is ongoing, promising a future rich with scientific discovery and technological innovation.
