Understanding Heparin and Its Paradoxical Complication
Heparin-induced thrombocytopenia (HIT) is a serious, immune-mediated adverse drug reaction that can occur in patients receiving heparin, a commonly used anticoagulant. While heparin is indispensable in preventing and treating various thrombotic disorders, HIT presents a paradoxical challenge: it leads to a reduction in platelet count (thrombocytopenia) and an increased risk of new or worsening thrombosis, rather than its intended anticoagulant effect. Understanding this complex condition is crucial for timely diagnosis and effective management, directly impacting patient outcomes in critical care settings, surgical recovery, and long-term anticoagulation.
The Role of Heparin
Heparin, available as unfractionated heparin (UFH) and low molecular weight heparin (LMWH), works by binding to antithrombin, a natural anticoagulant protein, thereby accelerating its inhibitory effect on various clotting factors, particularly thrombin (Factor IIa) and Factor Xa. It is widely used for conditions like deep vein thrombosis (DVT), pulmonary embolism (PE), myocardial infarction, and during surgical procedures like cardiac bypass. Its rapid onset of action and reversibility make it a cornerstone of modern medicine. However, despite its benefits, heparin administration is not without risks, with bleeding being the most common adverse effect. HIT, though less frequent, represents a far more dangerous complication due to its prothrombotic nature.

Atypical Platelet Response
The hallmark of HIT is a significant drop in platelet count, typically occurring 5 to 10 days after heparin initiation, though it can manifest sooner in patients with prior heparin exposure. This thrombocytopenia is not benign; it acts as a precursor to life-threatening thrombotic events. Unlike other forms of thrombocytopenia, which often increase bleeding risk, HIT paradoxically leads to an increased propensity for clot formation in both arterial and venous systems. This unique characteristic underscores the complexity of HIT and necessitates a high index of suspicion from clinicians. Without appropriate recognition and intervention, HIT can lead to severe and potentially fatal complications, including limb ischemia, stroke, myocardial infarction, and extensive venous thromboembolism.
Pathophysiology: The Immune-Mediated Mechanism
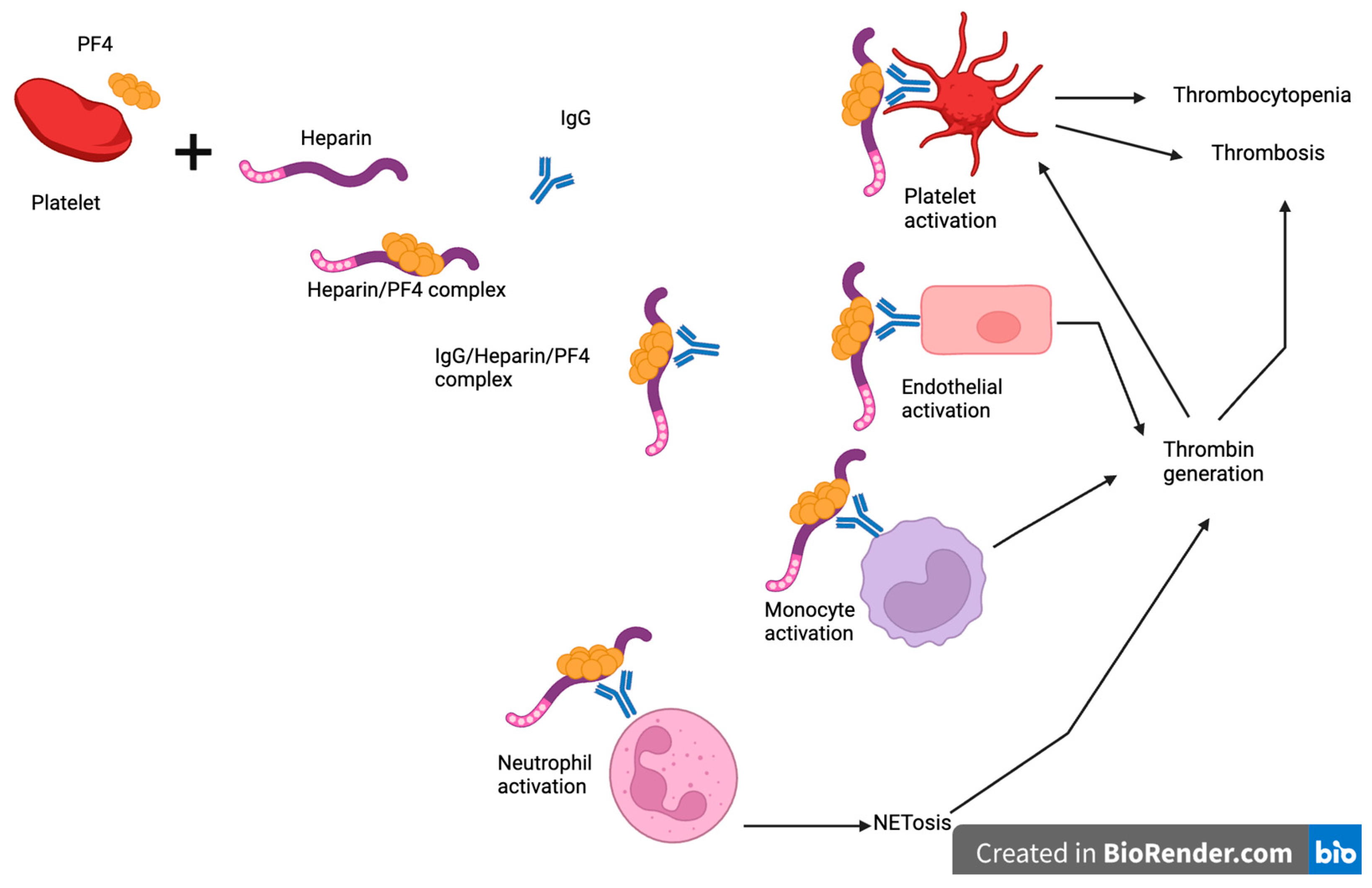
The pathogenesis of HIT is rooted in an intricate immune response triggered by heparin. It is classified as an antibody-mediated disorder, where the body’s immune system mistakenly targets a complex formed between heparin and a platelet-derived protein, leading to widespread platelet activation and subsequent hypercoagulability. This sequence of events is central to understanding why a drug designed to prevent clots can, in some individuals, provoke their formation.
Formation of PF4/Heparin Complexes
The initial step in HIT involves the binding of heparin, particularly unfractionated heparin, to platelet factor 4 (PF4), a protein released from alpha granules of activated platelets. PF4 is a positively charged chemokine that readily forms complexes with the negatively charged heparin molecule. These heparin-PF4 complexes are then presented on the surface of platelets and endothelial cells. In some susceptible individuals, these complexes are recognized as foreign by the immune system, leading to the production of antibodies. Low molecular weight heparins are less likely to form these immunogenic complexes due to their smaller size and reduced binding affinity for PF4, accounting for their lower incidence of HIT.
Antibody Production and Platelet Activation
The immune system, upon recognizing the heparin-PF4 complex as an antigen, produces specific antibodies, primarily of the IgG class, though IgA and IgM antibodies can also be involved. These anti-heparin/PF4 antibodies bind to the complexes on the platelet surface, leading to the activation of platelets via their FcγIIa receptors. This activation is a critical step, as it causes platelets to release more PF4, perpetuating a vicious cycle of complex formation, antibody binding, and further platelet activation. The activated platelets also release procoagulant microparticles and aggregate, further reducing circulating platelet counts and promoting clot formation.
Thrombus Formation
The widespread activation and aggregation of platelets, combined with the release of procoagulant factors, create a highly thrombogenic state. The activated platelets also damage the endothelial lining of blood vessels, contributing to the development of thrombi. These thrombi can form in both arteries and veins, leading to a spectrum of clinical manifestations. Venous thromboses, such as DVT and PE, are common, but arterial thromboses, which can cause devastating consequences like stroke, myocardial infarction, and limb gangrene, are also significant risks. The consumption of platelets during this process is what ultimately leads to the thrombocytopenia observed in HIT, a paradoxical scenario where low platelet count is associated with high clotting risk.
Clinical Manifestations and Diagnostic Challenges
Recognizing HIT promptly is paramount due to its severe prothrombotic potential. The clinical presentation can vary, but a consistent pattern of thrombocytopenia in a patient receiving heparin should immediately raise suspicion. Diagnosis is often a multi-faceted process involving clinical assessment, scoring systems, and laboratory confirmation.
Recognising the Symptoms
The most characteristic symptom of HIT is a new onset of thrombocytopenia, typically defined as a platelet count drop of more than 50% from baseline, or a fall below 100,000/µL, occurring 5 to 10 days after heparin initiation. In patients with recent heparin exposure (within the last 30 to 100 days), HIT can manifest much sooner, even within hours of re-exposure, a phenomenon known as “rapid onset HIT.” Beyond the platelet drop, patients may present with signs of new thrombosis, despite ongoing anticoagulation. These can include:
- Venous thrombosis: Deep vein thrombosis (DVT) in the limbs, pulmonary embolism (PE), and less commonly, cerebral venous sinus thrombosis or splanchnic vein thrombosis.
- Arterial thrombosis: Stroke, myocardial infarction, acute limb ischemia leading to gangrene, or mesenteric ischemia.
- Skin lesions: Necrotic lesions at heparin injection sites, or diffuse erythema, which are rare but highly specific indicators of HIT.
- Adrenal hemorrhage: A severe, albeit rare, complication of thrombotic microangiopathy in the adrenal glands.
Any new thrombotic event in a patient receiving heparin, especially in conjunction with thrombocytopenia, demands immediate evaluation for HIT.
Diagnostic Criteria: The 4T’s Score
Given the need for rapid assessment, clinicians often use the 4T’s score as a pre-test probability tool to estimate the likelihood of HIT. This scoring system evaluates four key parameters:
- Thrombocytopenia: The extent and timing of the platelet count drop.
- Timing of platelet count fall: When the thrombocytopenia occurred relative to heparin exposure.
- Thrombosis or other sequelae: Presence of new thrombosis, skin lesions, or acute systemic reaction after heparin bolus.
- Other causes for thrombocytopenia: Absence of other clear reasons for the low platelet count.
Each parameter is scored 0, 1, or 2 points, resulting in a total score ranging from 0 to 8. A high 4T’s score (6-8 points) indicates a high probability of HIT, a intermediate score (4-5 points) suggests moderate probability, and a low score (0-3 points) indicates a low probability. A low probability score usually allows for ruling out HIT, while intermediate and high scores warrant further laboratory testing.
Laboratory Confirmation
Laboratory tests for HIT fall into two main categories: antigen assays and functional assays.
- Antigen Assays (e.g., ELISA): These tests detect the presence of antibodies against the heparin-PF4 complex. They are highly sensitive, meaning they can effectively rule out HIT if negative. However, they are less specific, as many patients may have these antibodies without developing clinical HIT. A positive antigen assay indicates the presence of antibodies but does not confirm their pathogenic activity.
- Functional Assays (e.g., Heparin-Induced Platelet Aggregation [HIPA], Serotonin Release Assay [SRA]): These tests are more specific as they detect antibodies that can actually activate platelets in the presence of heparin. They are considered the gold standard for confirming HIT. However, functional assays are technically demanding, expensive, and often take several days to yield results, which can delay critical treatment decisions.
Typically, a high or intermediate 4T’s score prompts the immediate discontinuation of heparin and initiation of alternative anticoagulation, while laboratory tests are pending to confirm the diagnosis.
Management Strategies and Prevention
Managing HIT requires immediate action to halt the immune process and prevent thrombotic complications. The cornerstone of treatment involves discontinuing all heparin products and initiating alternative non-heparin anticoagulation. Prevention is also a key consideration, especially in high-risk patients.
Immediate Cessation of Heparin
Upon suspicion or diagnosis of HIT, all forms of heparin, including heparin flushes, heparin-coated catheters, and both UFH and LMWH, must be immediately discontinued. This is crucial to remove the antigenic trigger for the immune response and prevent further antibody-mediated platelet activation. Failure to stop heparin can exacerbate thrombocytopenia and significantly increase the risk of thrombosis.
Alternative Anticoagulation
Once heparin is stopped, immediate initiation of an alternative, non-heparin anticoagulant is critical, regardless of the platelet count, to mitigate the high thrombotic risk. Platelet transfusions are generally avoided in HIT unless there is active bleeding or a planned invasive procedure, as they can potentially fuel thrombosis by providing more target platelets for the antibodies. The preferred alternative anticoagulants include:
- Direct Thrombin Inhibitors (DTIs): Argatroban and bivalirudin are intravenous DTIs that directly inhibit thrombin without requiring antithrombin, thus circumventing the heparin-PF4 antibody mechanism. Argatroban is often used in patients with renal impairment, while bivalirudin is preferred in those with hepatic dysfunction.
- Factor Xa Inhibitors: Fondaparinux, a synthetic pentasaccharide, indirectly inhibits Factor Xa and has a lower propensity to cause HIT antibodies. Direct oral anticoagulants (DOACs) like rivaroxaban, apixaban, edoxaban, and dabigatran (after initial parenteral therapy) are increasingly used for long-term management once the acute phase is resolved and platelet counts have recovered.
The goal of alternative anticoagulation is to maintain a therapeutic level of anticoagulation until the platelet count recovers and the acute thrombotic risk subsides.
Monitoring and Long-Term Care
Patients with HIT require close monitoring of their platelet counts and clinical status for signs of new or worsening thrombosis. Once the platelet count has stabilized and recovered (typically above 150,000/µL or returned to baseline), patients can usually be transitioned to an oral anticoagulant. Vitamin K antagonists (VKAs) like warfarin are often used but should only be initiated after platelet recovery and overlapping with a non-heparin parenteral anticoagulant for at least 5 days. Initiating warfarin too early in HIT can paradoxically exacerbate the thrombotic state by transiently reducing protein C, a natural anticoagulant. Patients typically require at least 3 to 6 months of therapeutic anticoagulation. Future heparin exposure should be strictly avoided in patients with a history of HIT, as re-exposure can lead to rapid onset and severe reactions.
Prophylactic Considerations
While complete prevention of HIT is not possible, certain strategies can reduce its incidence or severity. These include using LMWH instead of UFH for routine thromboprophylaxis when appropriate, as LMWH has a lower incidence of HIT. Minimizing the duration of heparin exposure and careful monitoring of platelet counts in all heparinized patients are also crucial. For patients undergoing procedures requiring anticoagulation and with a history of HIT, alternative anticoagulants like DTIs are the preferred choice.
Prognosis and Complications
The prognosis of HIT is largely dependent on the promptness of diagnosis and initiation of appropriate management. Untreated or delayed diagnosis can lead to severe and potentially fatal complications. Even with appropriate treatment, patients who have experienced HIT may face long-term challenges.
Potential Long-Term Sequelae
The most significant long-term sequelae of HIT are related to its thrombotic complications. Patients can suffer from post-thrombotic syndrome after DVT, characterized by chronic pain, swelling, and skin changes in the affected limb. Arterial thromboses can result in permanent organ damage, such as stroke with neurological deficits, myocardial infarction with reduced cardiac function, or limb loss due to severe ischemia. Recurrence of thrombosis is also a concern if anticoagulation is not managed appropriately during the acute and subacute phases. Patients with a history of HIT also face a lifelong risk of recurrence if re-exposed to heparin.
Impact on Patient Outcomes
HIT significantly increases morbidity and mortality. Studies show that patients with HIT have a higher risk of death, amputation, and long-term disability compared to those without the condition. The condition also leads to prolonged hospital stays and increased healthcare costs. The psychological impact on patients, including anxiety about future thrombotic events and the need for long-term anticoagulation, should also not be underestimated. Therefore, continuous vigilance, rapid diagnostic algorithms, and effective treatment protocols are essential to mitigate the devastating impact of heparin-induced thrombocytopenia and improve patient outcomes.
