In the realm of Tech & Innovation, particularly concerning advanced drone development, understanding fundamental chemical principles like polar and nonpolar bonds is crucial. These atomic-level interactions dictate the properties of materials, from the robust composites forming a drone’s frame to the intricate electrolytes powering its batteries and the sophisticated sensors gathering data. While seemingly abstract, the nature of these bonds directly influences a drone’s durability, efficiency, and operational capabilities, pushing the boundaries of what autonomous flight can achieve.
The Fundamentals of Chemical Bonding
At its core, a chemical bond is the force that holds atoms together to form molecules or compounds. This force arises from the interaction of electrons, specifically the valence electrons in the outermost shell of an atom. Atoms seek to achieve a stable electron configuration, typically by having a full outer shell, which often means having eight valence electrons (the octet rule). They accomplish this by either transferring electrons (forming ionic bonds) or sharing electrons (forming covalent bonds). Our focus for understanding drone materials and innovations lies predominantly with covalent bonds, where the distinction between polar and nonpolar interactions becomes paramount.

Covalent Bonds: The Shared Electron Pair
Covalent bonds are formed when two atoms share one or more pairs of electrons. This sharing allows each atom to effectively complete its valence shell, achieving greater stability. The shared electrons are mutually attracted to the nuclei of both atoms, creating a strong bond that holds the molecule together. Examples are abundant in nature and technology, from the water we drink to the carbon-fiber composites used in high-performance drones. The stability and integrity of these shared electron pairs are not always uniform, leading to the crucial distinction of polarity.
Electronegativity: The Electron-Pulling Power
The concept of electronegativity is central to understanding bond polarity. Electronegativity is a measure of an atom’s ability to attract shared electrons in a chemical bond. Developed by Linus Pauling, this scale helps predict how electrons will be distributed between two bonded atoms. Atoms with high electronegativity, like oxygen or fluorine, exert a stronger pull on shared electrons than atoms with lower electronegativity, such as hydrogen or carbon. The difference in electronegativity between two bonded atoms is the primary factor determining whether a covalent bond will be polar or nonpolar, fundamentally shaping the material’s interaction with its environment and its overall performance in advanced drone systems.
Unpacking Nonpolar Covalent Bonds
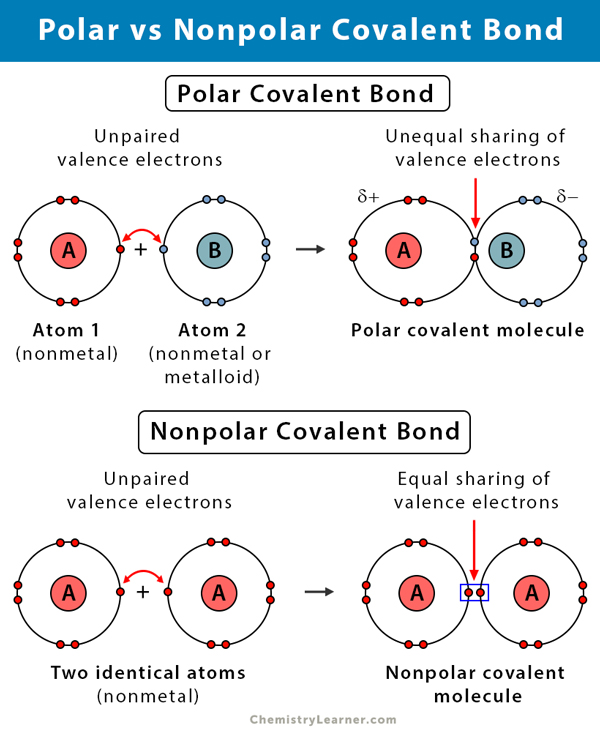
A nonpolar covalent bond occurs when electrons are shared equally between two atoms. This typically happens when the two bonded atoms are identical or have very similar electronegativities. In such cases, there is no significant difference in their ability to attract the shared electron pair, leading to a symmetrical distribution of electron density across the bond.
Symmetrical Electron Distribution
Consider a molecule of oxygen (O₂), where two oxygen atoms are bonded together. Since both atoms are identical, their electronegativity values are the same. Consequently, they share the bonding electrons perfectly equally. The electron cloud is evenly distributed around both nuclei, resulting in no net positive or negative charge accumulation at either end of the bond. The molecule as a whole, therefore, has no distinct poles of charge. This symmetrical electron distribution is a hallmark of nonpolar bonds and influences how these materials behave, particularly in environments relevant to drone operations.
Examples in Drone Materials
Nonpolar bonds are critical in the engineering of many drone components, especially where stability and resistance to polar solvents are desired.
- Carbon-Fiber Composites: The backbone of many advanced drone frames consists of carbon-fiber reinforced polymers. The C-C bonds within the carbon fibers, and to a large extent the C-H bonds in the polymer matrices (like epoxies), are predominantly nonpolar. This nonpolar nature contributes to the material’s strength, rigidity, and lightweight properties. These materials exhibit excellent chemical stability, resisting interaction with water and other polar substances, which is vital for drones operating in diverse weather conditions.
- Lubricants and Coatings: Many lubricants and protective coatings used on drone motors or sensitive electronic components are designed with nonpolar characteristics. Hydrocarbons, which feature long chains of C-C and C-H nonpolar bonds, are excellent nonpolar solvents and repellents for water. This ensures proper functioning of mechanical parts and shields delicate electronics from moisture and corrosion, enhancing the drone’s operational lifespan and reliability.
- Propeller Materials: High-performance propellers often utilize engineered plastics or composites with significant nonpolar components. Their nonpolar nature ensures mechanical integrity and resistance to environmental degradation, maintaining aerodynamic efficiency under stress.
Exploring Polar Covalent Bonds
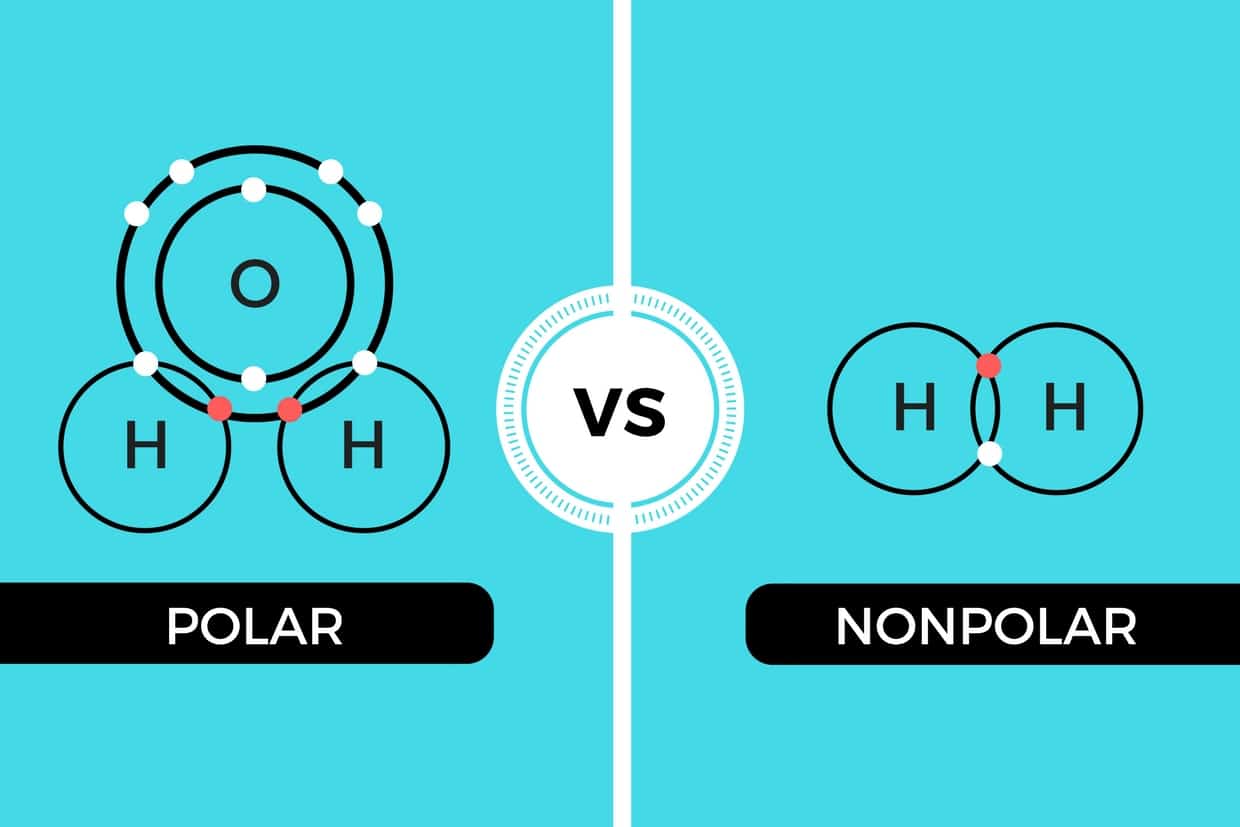
In contrast to nonpolar bonds, a polar covalent bond forms when electrons are shared unequally between two atoms. This imbalance arises from a significant difference in their electronegativity, meaning one atom pulls the shared electron pair closer to itself than the other.
Uneven Electron Sharing and Dipole Moments
When there’s a noticeable electronegativity difference, the atom with the higher electronegativity will attract the shared electrons more strongly. This creates a partial negative charge (δ⁻) on the more electronegative atom and a partial positive charge (δ⁺) on the less electronegative atom. The bond effectively develops two poles, much like a tiny magnet, resulting in a bond dipole moment. A classic example is the water molecule (H₂O). Oxygen is significantly more electronegative than hydrogen, pulling the shared electrons closer to itself. This makes the O-H bonds polar, creating a partial negative charge on the oxygen and partial positive charges on the hydrogens. The overall bent shape of the water molecule further ensures that these individual bond dipoles do not cancel out, making water a highly polar molecule.
Impact on Material Properties for Drones
The presence of polar bonds has profound implications for the physical and chemical properties of materials, which are extensively leveraged in drone technology:
- Adhesion and Bonding: Polar bonds play a crucial role in adhesives and coatings where strong intermolecular forces are desired. For instance, the adhesion of protective layers to drone components, or the bonding between different material layers in composites, often relies on the attractive forces between polar molecules or regions.
- Sensor Technology: Many advanced sensors for environmental monitoring (e.g., humidity sensors, gas sensors) exploit the interaction of target molecules with polar surfaces. The specific dipole moments of these sensor materials allow them to selectively attract and detect polar molecules in the air, providing drones with crucial data for agricultural applications, air quality monitoring, or search and rescue operations.
- Dielectric Properties: Materials with highly polar bonds can exhibit high dielectric constants. These are vital for capacitors in drone electronics, allowing them to store more electrical energy in a smaller space, which is critical for miniaturization and efficiency. They also play a role in insulating materials, where their ability to dissipate electrical fields is paramount for protecting sensitive components.
- Moisture Absorption: While nonpolar materials repel water, polar materials can absorb it. This can be a double-edged sword: certain battery separators might benefit from controlled moisture content, while structural components must be designed to resist moisture ingress to prevent degradation. Understanding bond polarity helps engineers choose the right materials for specific applications to either attract or repel water as needed.
Ionic Bonds: The Extreme Polarity
While the primary focus is on covalent bonds, it’s worth noting the extreme end of the polarity spectrum: ionic bonds. These occur when the electronegativity difference between two atoms is so vast that one atom effectively transfers one or more electrons to the other, rather than sharing them. This results in the formation of fully charged ions (cations and anions) that are held together by strong electrostatic attraction. Sodium chloride (table salt) is a common example. While not typically forming the structural backbone of drones, ionic compounds can be found in certain battery electrolytes (e.g., lithium salts in lithium-ion batteries) or in specialized sensor components, where their charge-carrying capabilities are essential for electrochemical processes. The strong electrostatic forces in ionic bonds contribute to the high melting points and hardness of such materials, though their brittleness often limits their use in primary structural roles.
The Technological Ramifications for Drone Innovation
The precise understanding and manipulation of polar and nonpolar bonds are foundational to the continuous innovation driving the drone industry forward. From extending flight times to enhancing payload capabilities and improving resilience in harsh environments, molecular-level design is key.
Advanced Polymers and Composites
Future drone designs demand materials that are increasingly lighter, stronger, and more resilient. Innovations in advanced polymers and composites critically depend on engineering the right balance of polar and nonpolar bonds. For example, creating self-healing polymers involves designing macromolecules with specific polar interactions that allow damaged sections to re-form bonds. Developing hydrophobic (water-repelling) coatings for drone electronics relies on surfaces rich in nonpolar bonds, preventing water damage. Conversely, designing smart materials that can change properties in response to stimuli often involves tuning the polarity of specific molecular segments, allowing for adaptive wing structures or deployable mechanisms.
Battery Chemistry and Thermal Management
The performance of drone batteries, particularly lithium-ion and solid-state alternatives, is heavily influenced by the polarity of their constituent materials. Electrolytes in lithium-ion batteries often utilize polar solvents to dissolve and transport lithium ions efficiently, allowing for high power output. Research into solid-state batteries explores new solid electrolytes where the nature of ionic and polar interactions dictates ion mobility and overall battery safety and energy density. Furthermore, thermal management strategies often involve materials with specific thermal conductivities, which can be influenced by how atoms are bonded and how easily vibrations (heat) can propagate through polar or nonpolar molecular networks.
Sensor Technology and Environmental Resilience
The next generation of drone sensors will be more sensitive, specific, and capable of operating in extreme conditions. This advancement hinges on understanding and leveraging bond polarity. For instance, developing highly selective gas sensors for detecting trace amounts of pollutants or specific biological agents relies on designing active sensor surfaces with precisely tailored polar characteristics that interact uniquely with target molecules. Similarly, for drones operating in marine environments or extreme temperatures, creating surfaces that resist fouling or maintain structural integrity under thermal stress requires materials engineered at the molecular level with robust polar or nonpolar bond networks, ensuring longevity and reliable data acquisition. The ability to precisely control these fundamental bond characteristics is unlocking new possibilities for autonomous monitoring, data collection, and mission execution.
