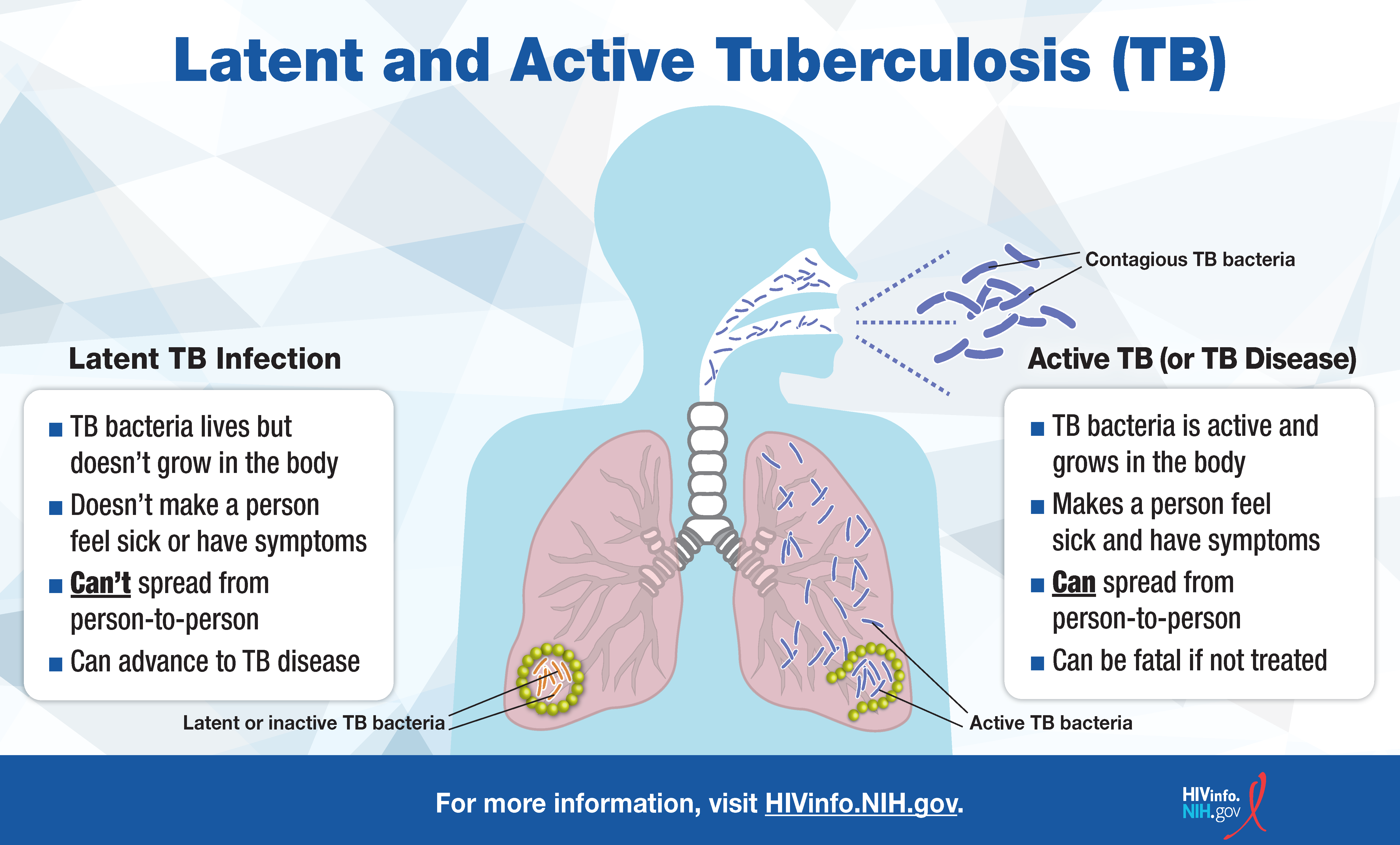
Latent Tuberculosis Infection (LTBI) represents a formidable global health challenge, affecting an estimated one-quarter of the world’s population. Unlike active tuberculosis disease, LTBI is characterized by the presence of Mycobacterium tuberculosis (Mtb) bacteria in the body without outward symptoms or contagiousness. However, individuals with LTBI carry a 5-10% lifetime risk of progressing to active TB disease, a risk that significantly escalates in immunocompromised individuals. Understanding what latent tuberculosis is, therefore, is not merely a medical inquiry but a critical area for technological intervention and innovation. In an era defined by rapid technological advancement, from artificial intelligence to sophisticated biosensors, the fight against LTBI is increasingly being waged on the digital and biotechnological frontiers. This article explores latent tuberculosis through the lens of Tech & Innovation, examining how cutting-edge technologies are reshaping our approach to detection, monitoring, and future eradication strategies for this silent pandemic.
The Silent Threat: Reimagining Diagnosis Through Innovation
The insidious nature of latent tuberculosis lies in its asymptomatic state, making traditional diagnosis challenging and often reliant on reactive measures. For decades, the primary tools for identifying LTBI have been the Tuberculin Skin Test (TST) and Interferon-Gamma Release Assays (IGRAs). While foundational, these methods possess inherent limitations, prompting a pressing need for more accurate, accessible, and rapid diagnostic innovations. The global burden of LTBI, particularly in low- and middle-income countries, demands solutions that transcend geographical and infrastructural barriers, pushing the boundaries of what’s possible with modern technology.
Limitations of Traditional Detection Methods
Traditional methods, such as the TST, suffer from variable sensitivity and specificity, often complicated by cross-reactivity with BCG vaccination or exposure to non-tuberculous mycobacteria. This can lead to false positives, unnecessarily subjecting individuals to drug regimens, or false negatives, leaving infected individuals undiagnosed and at risk of progression. IGRAs, while more specific, are blood-based tests that require laboratory infrastructure, skilled personnel, and are often more expensive, limiting their widespread adoption in resource-constrained settings. These limitations underscore a critical unmet need for non-invasive, point-of-care, and highly accurate diagnostic tools that can be deployed at scale. The drive for innovation in this space is not just about incremental improvements but about a fundamental shift in how we detect and manage LTBI.
AI and Machine Learning for Enhanced Screening
The advent of Artificial Intelligence (AI) and Machine Learning (ML) is profoundly transforming medical diagnostics, and LTBI detection is no exception. AI algorithms can be trained on vast datasets of medical images, clinical histories, and genomic information to identify subtle patterns indicative of LTBI that might elude the human eye or conventional analysis. For instance, AI-powered analysis of chest X-rays, even in seemingly healthy individuals, can detect early fibrotic changes or calcifications often associated with past TB exposure or LTBI, thereby improving the predictive value of imaging for identifying high-risk individuals.
Furthermore, ML models can integrate various risk factors—demographic data, exposure history, immunological markers, and co-morbidities—to create predictive analytics tools. These tools can stratify populations based on their likelihood of having LTBI or progressing to active disease, enabling targeted screening and preventive therapy. This intelligent risk assessment can optimize resource allocation, ensuring that interventions are directed towards those who stand to benefit most, thereby enhancing the efficiency and impact of public health programs globally.
Advanced Imaging Techniques: Beyond the X-ray
While AI enhances the utility of existing imaging, innovation also extends to the development of entirely new imaging modalities or the refinement of current ones. Beyond standard X-rays, advanced techniques like low-dose CT scans are being explored for more precise detection of pulmonary lesions consistent with LTBI. Although CT scans pose radiation exposure concerns and are cost-prohibitive for mass screening, innovative approaches such as ultra-low-dose protocols or AI-enhanced image reconstruction could make them more viable for specific high-risk groups or research settings.
Moreover, the frontier of molecular imaging holds promise. Positron Emission Tomography (PET) scans, combined with specific radiotracers that bind to Mtb-infected cells or inflammatory markers, could offer unprecedented insights into the metabolic activity and immunological state of granulomas in LTBI. Such techniques could differentiate between truly latent and incipient active disease, revolutionizing personalized risk assessment and treatment strategies. These sophisticated imaging innovations, while still largely in research phases, represent a significant leap towards non-invasive and highly specific detection of LTBI.
Biosensors and Point-of-Care Diagnostics: Democratizing Detection
The ultimate goal for widespread LTBI detection is the development of rapid, accurate, and affordable point-of-care (POC) diagnostics. These technologies aim to decentralize testing from specialized laboratories to community clinics, pharmacies, and even homes, making diagnosis accessible to underserved populations. The innovation in this domain leverages advancements in microfluidics, nanotechnology, and electrical engineering to create sophisticated yet user-friendly devices.
Non-Invasive Biomarker Detection
The search for novel non-invasive biomarkers is paramount. Researchers are exploring volatile organic compounds (VOCs) released in breath, specific DNA/RNA fragments in saliva or urine, and unique protein signatures in blood, all indicative of Mtb infection. The innovation lies in developing highly sensitive biosensors capable of detecting these minute quantities of biomarkers. Nanotechnology plays a crucial role here, with nanoparticles engineered to capture and amplify signals from target molecules, dramatically improving detection limits. For instance, handheld devices utilizing electrochemical or optical biosensors are being developed to detect Mtb-specific antigens or host immune responses from a simple finger-prick blood sample, delivering results in minutes rather than days.
Lab-on-a-Chip and Microfluidic Devices
“Lab-on-a-chip” technologies exemplify the miniaturization trend, integrating multiple laboratory functions onto a single, small chip. These microfluidic devices can perform complex assays, such as sample preparation, nucleic acid amplification, and detection, using minimal reagents and power. For LTBI, this translates into portable devices that can rapidly process samples, identify Mtb DNA, or measure specific immune responses, offering a complete diagnostic workflow in a self-contained unit. Such innovations drastically reduce the need for bulky lab equipment and trained technicians, making advanced diagnostics deployable in the most remote settings. The integration of smartphone compatibility further enhances their utility, allowing for automated result interpretation, data logging, and seamless reporting to healthcare providers, effectively bridging diagnostic gaps.
Innovation in Monitoring and Management
Detecting LTBI is only the first step; effective management requires consistent monitoring and adherence to often lengthy preventive treatment regimens. Innovation in this space focuses on leveraging digital health and wearable technologies to support patients and healthcare systems in ensuring successful treatment outcomes and preventing progression to active disease.
Digital Health Platforms for Adherence and Tracking
Digital health interventions, including mobile applications and web-based platforms, are increasingly vital in managing chronic conditions, including LTBI. These platforms can facilitate medication reminders, provide educational content about LTBI and treatment adherence, and enable secure communication between patients and healthcare providers. Personalized dashboards can track treatment progress, log side effects, and monitor adherence rates, offering real-time data for both patients and clinicians.
Furthermore, these platforms can integrate with national TB programs for centralized data collection and epidemiological surveillance. This allows public health officials to identify geographical hotspots, track treatment outcomes across large populations, and implement data-driven strategies for LTBI control. The secure handling of sensitive patient data, leveraging blockchain technology for data integrity and privacy, represents another layer of innovation in this critical area.
Wearable Technology in High-Risk Populations
Wearable devices, such as smartwatches and fitness trackers, are moving beyond wellness into clinical applications. For LTBI, wearables could offer a non-invasive method for continuous physiological monitoring, particularly for individuals undergoing preventive therapy. While not directly diagnosing LTBI, changes in sleep patterns, heart rate variability, body temperature, or activity levels, when integrated with other health data, could potentially signal early physiological shifts that warrant medical attention, especially if an individual is at high risk of progressing to active disease.
In research settings, wearables could also be utilized to objectively measure medication adherence (e.g., through pill box sensors or ingested sensors) and to monitor for subtle side effects that might impact quality of life and adherence. While the application of wearables for LTBI is still nascent, their potential to generate real-world, longitudinal health data offers new avenues for personalized care and early intervention, particularly for vulnerable populations where regular clinic visits might be challenging.
Future Horizons: Precision Medicine and Eradication Strategies
The ultimate vision for tackling LTBI extends beyond improved diagnosis and management to personalized interventions and, eventually, global eradication. This ambitious goal is being propelled by breakthroughs in genomics, synthetic biology, and advanced immunological research, paving the way for a new era of precision medicine and innovative vaccine development.
Genomic Sequencing for Personalized Treatment
The genomic revolution offers profound insights into both the host and the pathogen. Whole-genome sequencing of Mtb strains from infected individuals can identify specific genetic markers related to drug resistance or virulence, informing more effective and personalized preventive therapy regimens. Simultaneously, host genomics can elucidate individual susceptibility to LTBI progression, identifying genetic predispositions that make certain people more likely to develop active disease.
Integrating these genomic insights allows for precision medicine approaches: tailoring preventive therapy based on an individual’s unique genetic makeup and the specific Mtb strain they carry. This could mean shorter, more potent drug regimens for some, and different drug combinations for others, minimizing side effects and maximizing efficacy. The application of big data analytics and bioinformatics is crucial for processing and interpreting these vast genomic datasets, translating complex genetic information into actionable clinical decisions.
Vaccine Development Leveraging Biotech
The holy grail in the fight against TB, including LTBI, is a highly effective vaccine that can prevent both initial infection and progression to active disease. Current vaccine development leverages advanced biotechnological approaches, moving beyond the attenuated live vaccine (BCG) to subunit vaccines, viral vector vaccines, and mRNA vaccines, drawing inspiration from recent successes in other infectious diseases.
Biotechnology enables the design and synthesis of specific Mtb antigens that elicit strong and durable immune responses. Through genetic engineering, researchers are developing novel vaccine candidates that target Mtb during its latent phase, aiming to boost host immunity to contain the dormant bacteria indefinitely or even clear them from the body. Innovations in vaccine delivery systems, such as nanoparticle formulations or mucosal vaccines, are also being explored to enhance immunogenicity and ease of administration. The combination of structural biology, immunology, and synthetic biology is accelerating the pace of vaccine discovery, offering renewed hope for a future free from the shadow of tuberculosis.
Conclusion
Latent tuberculosis, once a silent and often underestimated threat, is now at the forefront of global health innovation. The convergence of advanced diagnostics, digital health platforms, and biotechnological breakthroughs is rapidly transforming our capacity to detect, monitor, and ultimately prevent the progression of LTBI to active disease. From AI-powered screening and sophisticated biosensors to personalized genomic medicine and next-generation vaccines, technology is not just augmenting existing strategies; it is fundamentally redefining the battle against Mtb. As these innovations mature and become more accessible, the vision of a world free from tuberculosis—active and latent—moves closer to becoming a reality, underscored by the relentless drive of human ingenuity and technological progress.
