Alcohol distillation, at its core, is a sophisticated process of separating components of a liquid mixture based on their differing boiling points. While the term “distillation” itself is broadly applied across various scientific and industrial fields, in the context of “what is alcohol distillation,” the focus narrows significantly to the production of alcoholic beverages and, by extension, its role in various technological and innovative applications where precise separation is paramount. This article will delve into the fundamental principles of alcohol distillation, its historical significance, the scientific underpinnings that make it possible, and its modern-day relevance, particularly as it intersects with areas of tech and innovation.
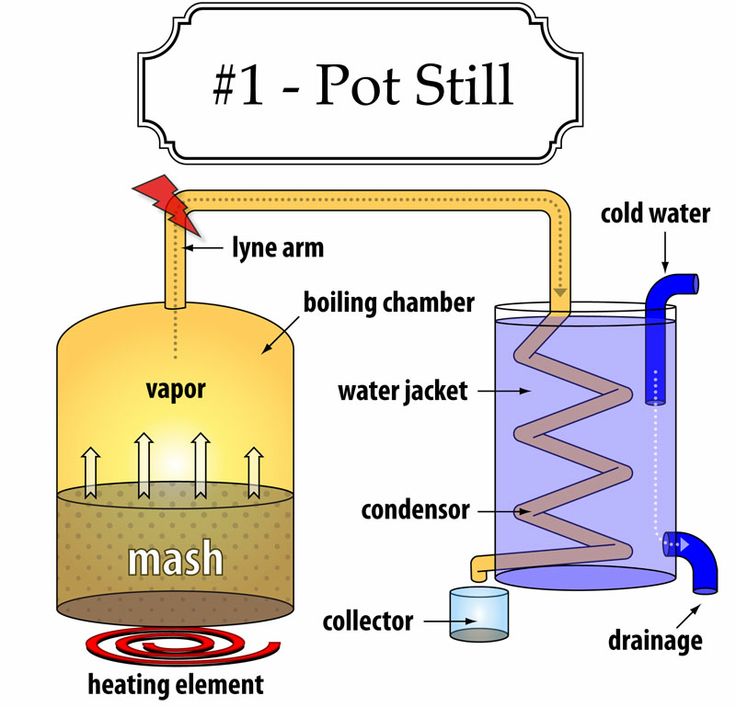
The Fundamental Principles of Distillation
At the heart of alcohol distillation lies a simple yet powerful scientific principle: vapor pressure. Every liquid has a tendency to evaporate, a phenomenon related to its vapor pressure. Liquids with higher vapor pressures evaporate more readily. Crucially, different substances have different boiling points – the temperature at which their vapor pressure equals the surrounding atmospheric pressure, causing them to turn into a gas.
For alcohol (specifically ethanol in the context of beverages), its boiling point is approximately 78.37 degrees Celsius (173.07 degrees Fahrenheit), while water boils at 100 degrees Celsius (212 degrees Fahrenheit). This difference is the linchpin of the distillation process.
The basic setup for distillation involves a heated vessel containing the liquid mixture (often called a “wash” or “mash” in brewing and distilling), a condenser to cool the vapor, and a collection vessel for the condensed liquid, known as the distillate.
-
Heating and Vaporization: The liquid mixture is heated. As the temperature rises, the component with the lower boiling point (ethanol) will vaporize at a proportionally higher rate than the component with the higher boiling point (water). The vapor that rises from the heated liquid is therefore enriched in the more volatile component.
-
Condensation: This ethanol-rich vapor travels upwards and into a condenser. The condenser is typically a cool tube or coil through which a cooling fluid (usually water) circulates. As the hot vapor comes into contact with the cool surfaces of the condenser, it loses heat and condenses back into a liquid.
-
Collection: This condensed liquid, now significantly richer in ethanol than the original mixture, drips into a collection vessel. This is the distillate.
This process can be repeated multiple times (known as “re-distillation” or “multiple distillations”) to achieve even higher concentrations of ethanol. Each distillation further refines the separation, removing more water and other less volatile compounds.
The Role of Azeotropes
A critical consideration in alcohol distillation is the concept of the azeotrope. An azeotrope is a mixture of two or more substances that has a constant boiling point. For ethanol and water, this occurs at approximately 95.6% ethanol by mass at standard atmospheric pressure. This means that through simple distillation, one cannot achieve 100% pure ethanol. The vapor produced when a 95.6% ethanol-water mixture boils will also be 95.6% ethanol. To achieve higher purities, specialized techniques or different separation methods are required.
Historical Significance and Evolution
The art and science of alcohol distillation have a long and rich history, predating many modern scientific understandings. While the exact origins are debated, evidence suggests its development in the Hellenistic world around the 1st century CE, with refinements and wider adoption occurring in the Islamic world and later in Europe.
Initially, distillation was primarily used for medicinal purposes, extracting essential oils from plants and for creating potent spirits for therapeutic use. Alchemists, in their quest for the “elixir of life,” inadvertently unlocked the secrets of separating and concentrating alcohol.
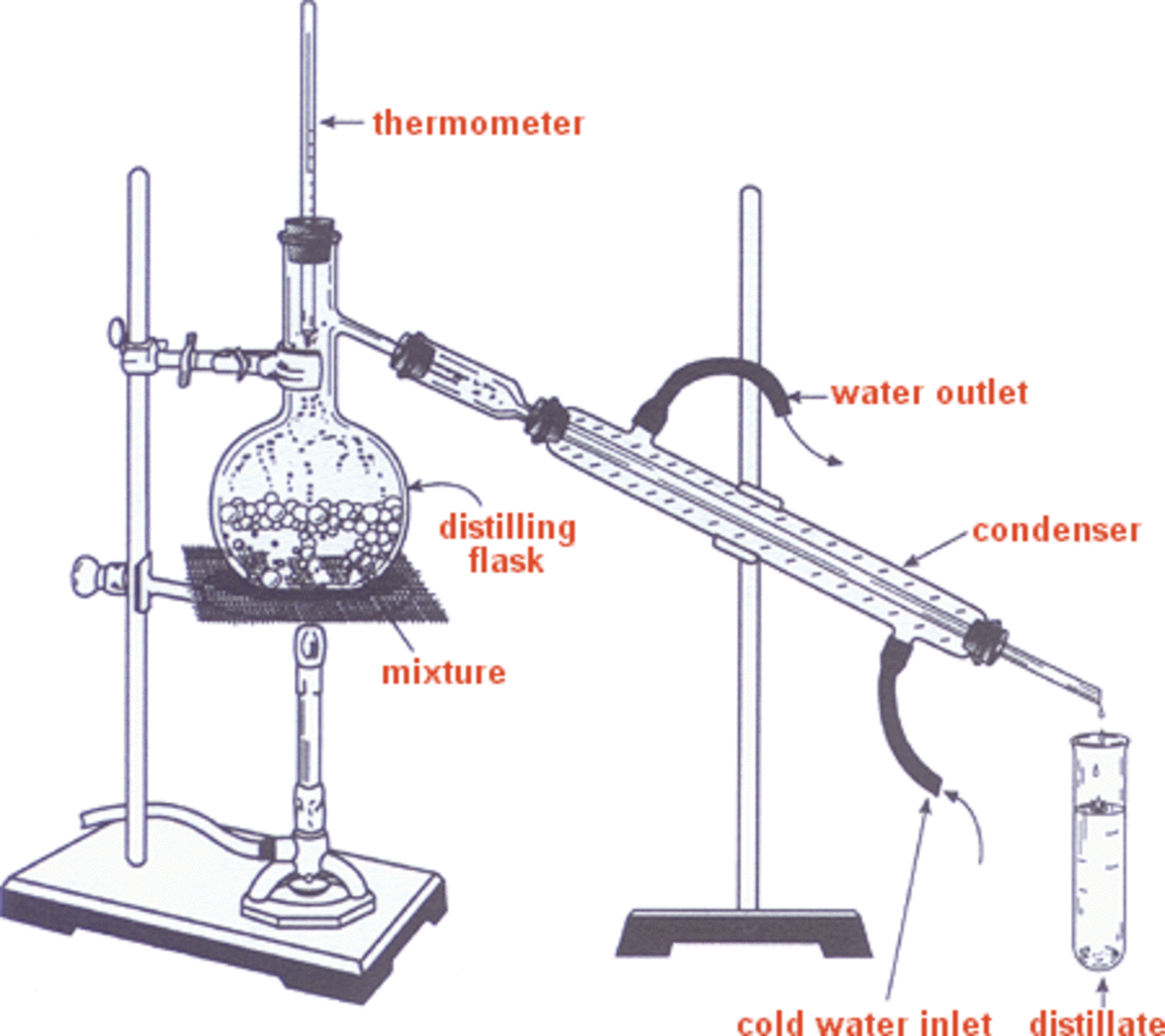
From Alchemy to Industrial Production
Over centuries, the techniques evolved. The development of more efficient stills, such as the alembic still, allowed for better control over the heating and cooling processes, leading to more consistent and potent spirits. As scientific understanding grew, particularly with the work of scientists like Antoine Lavoisier in the 18th century, the chemical principles behind distillation became clearer.
The Industrial Revolution marked a significant shift, transforming distillation from a craft-based practice into a large-scale industrial process. The invention of continuous stills, like the Coffey still (or column still) in the 1830s, revolutionized production. Unlike traditional pot stills, which operate in batches, continuous stills allow for a constant flow of raw material and a continuous output of distillate. This dramatically increased efficiency and reduced production costs, paving the way for the widespread availability of spirits and industrial ethanol.
Scientific Underpinnings and Modern Applications
The principles of distillation are not confined to the production of alcoholic beverages. They form the bedrock of countless chemical engineering processes and are increasingly integrated into advanced technological fields.
Chemical Engineering and Purification
In chemical engineering, distillation is one of the most fundamental and widely used separation techniques. It’s crucial for:
- Petroleum Refining: Crude oil is a complex mixture of hydrocarbons. Distillation towers separate crude oil into various fractions like gasoline, kerosene, diesel fuel, and lubricating oils based on their boiling points. This is a prime example of large-scale industrial distillation.
- Chemical Production: Many industrial chemicals are produced through processes involving distillation for purification or separation of reactants and products.
- Water Purification: While not producing alcoholic distillate, desalination plants and industrial water purification systems often employ distillation to separate pure water from dissolved salts and impurities.
Tech & Innovation: Distillation Beyond Beverages
The concept of precise separation, inherent in distillation, finds a unique resonance in the realm of “Tech & Innovation,” particularly in areas that leverage advanced analytical or purification techniques. While not directly about making spirits, the underlying scientific principles of separating components based on distinct properties are analogous.
-
Analytical Chemistry: Techniques like Gas Chromatography (GC) and High-Performance Liquid Chromatography (HPLC) often employ principles closely related to distillation for separating complex mixtures into their constituent parts for analysis. These methods are indispensable in research and development, quality control, and diagnostics across numerous industries. For example, in advanced materials science or pharmaceuticals, highly accurate separation is crucial for identifying and quantifying specific compounds.
-
Biotechnology and Pharmaceuticals: In the production of complex pharmaceuticals, including biopharmaceuticals like monoclonal antibodies, purification processes are paramount. While often involving chromatography and filtration, the fundamental drive to isolate a desired component from a complex biological matrix echoes the spirit of distillation – a targeted separation based on physical or chemical properties. Researchers might explore novel separation technologies that draw inspiration from distillation’s efficiency and scalability for extracting high-value compounds from biological sources.
-
Food Science and Flavor Extraction: Beyond alcoholic beverages, distillation is used to extract volatile aroma compounds from various food products, creating concentrated flavorings or essential oils. This requires a deep understanding of the boiling points and vapor pressures of diverse organic molecules, a direct application of distillation principles. Innovative methods are continuously being explored to enhance the efficiency and selectivity of these extraction processes.
-
Environmental Technology: In some environmental remediation processes, distillation can be used to recover solvents or separate pollutants from wastewater. This application highlights the versatility of the technique for purification and resource recovery, aligning with the innovation drive to create more sustainable industrial practices.
The pursuit of higher purities, greater efficiency, and novel separation methods in these advanced fields reflects a continuous innovation cycle, much like the historical evolution of the distillation apparatus itself. Understanding the foundational science of alcohol distillation provides a crucial perspective on the broader applications of separation technologies in modern innovation. The ability to precisely manipulate and separate components based on their inherent properties remains a cornerstone of technological advancement, from refining fuels to developing life-saving medicines.
