The fundamental building blocks of life, proteins, are intricate molecular machines that perform a staggering array of functions within all living organisms. From catalyzing biochemical reactions to providing structural support and transporting vital molecules, proteins are indispensable. The remarkable diversity and complexity of proteins arise from their unique linear sequences of amino acids, which are linked together by a specific type of chemical bond: the peptide bond. Understanding the visual and structural characteristics of this bond is crucial for comprehending protein folding, function, and the very essence of biological processes.
The Anatomy of an Amino Acid
Before delving into the peptide bond itself, it’s essential to understand the structure of its constituent units, amino acids. Amino acids are organic molecules that share a common structural framework. Each amino acid possesses a central carbon atom, known as the alpha-carbon (α-carbon). Attached to this α-carbon are four key groups:
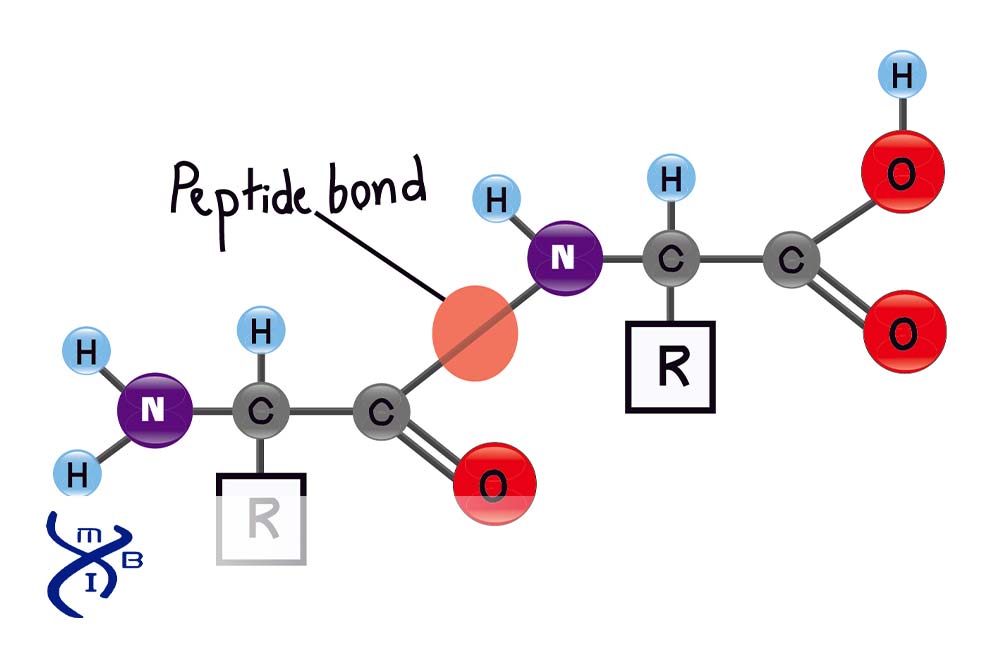
- An amino group (-NH₂): This is a nitrogen atom bonded to two hydrogen atoms. In biological systems, at physiological pH, this group is typically protonated, existing as an ammonium group (-NH₃⁺).
- A carboxyl group (-COOH): This is a carbon atom double-bonded to one oxygen atom and single-bonded to a hydroxyl group (-OH). At physiological pH, this group is usually deprotonated, existing as a carboxylate group (-COO⁻).
- A hydrogen atom (-H): A simple, uncharged hydrogen atom.
- A side chain (R-group): This is the variable component of the amino acid and is what differentiates one amino acid from another. The R-group can range in complexity from a single hydrogen atom (as in glycine) to elaborate ring structures, and it dictates many of the physical and chemical properties of the amino acid, including its polarity, charge, and size.
This tetrasubstituted carbon atom, the α-carbon, is chiral in all amino acids except glycine, meaning it has four different groups attached to it, leading to stereoisomers. However, in biological systems, only L-amino acids are incorporated into proteins.
Forming the Peptide Bond: The Dehydration Reaction
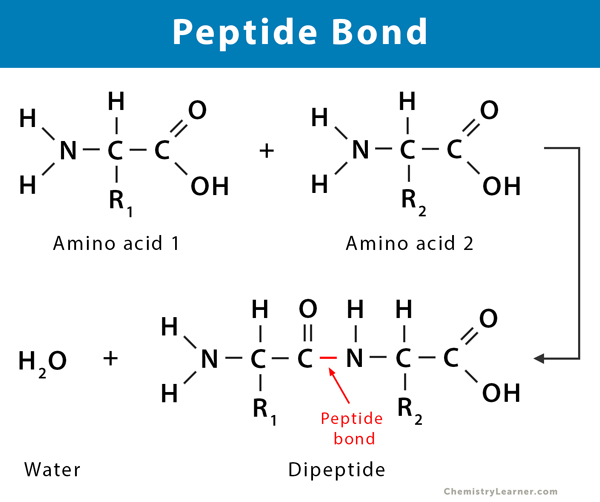
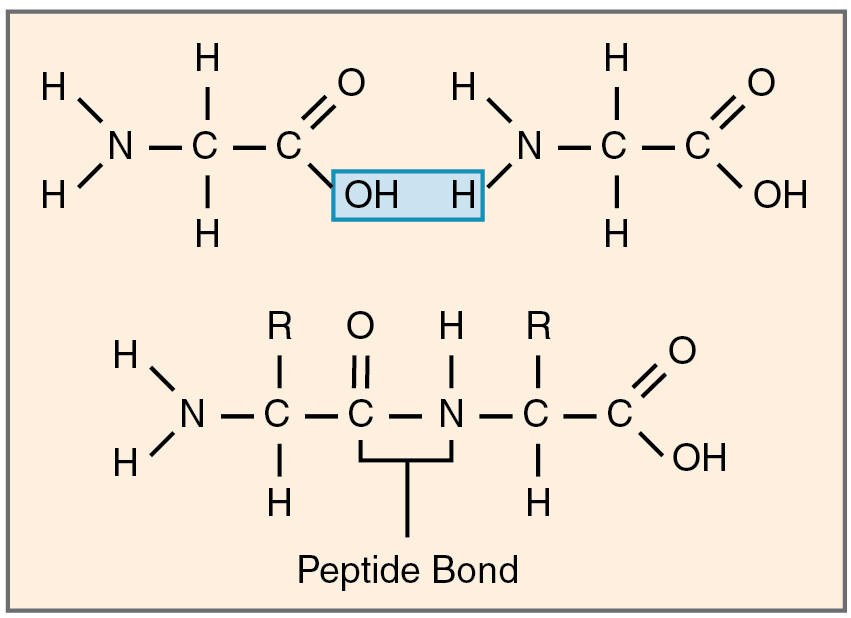
The formation of a peptide bond is a classic example of a dehydration (or condensation) reaction. This process involves the joining of two amino acids with the elimination of a water molecule. Specifically, the carboxyl group of one amino acid reacts with the amino group of another amino acid.
Let’s visualize this reaction:
- The Carboxyl Group’s Role: The hydroxyl (-OH) portion of the carboxyl group of the first amino acid is released.
- The Amino Group’s Role: One of the hydrogen atoms from the amino group of the second amino acid is released.
- Water Formation: The released -OH and -H combine to form a molecule of water (H₂O), which is eliminated from the reaction.
- Bond Formation: The remaining carbon atom from the carboxyl group and the remaining nitrogen atom from the amino group now form a covalent bond. This is the peptide bond.
This reaction results in the formation of a dipeptide, a molecule composed of two amino acids linked by a single peptide bond. The process can be repeated sequentially, adding more amino acids to form polypeptides, which are the long chains that constitute proteins.
The Visual Appearance of the Peptide Bond
The peptide bond itself, chemically known as an amide bond, has a distinctive planar structure due to resonance. Let’s break down its visual characteristics:
The Amide Linkage
The core of the peptide bond involves a carbon atom double-bonded to an oxygen atom and single-bonded to a nitrogen atom. This carbon-nitrogen linkage (-CO-NH-) is the peptide bond. The oxygen atom is typically depicted as a double-bonded oxygen (C=O), and the nitrogen atom is bonded to a hydrogen atom.
Resonance and Partial Double Bond Character
A critical aspect of the peptide bond’s structure is its partial double bond character, arising from resonance. The lone pair of electrons on the nitrogen atom of the amino group can delocalize into the adjacent carbonyl group. This delocalization can be represented by resonance structures:
-
Structure 1 (Dominant): The standard depiction with a C=O double bond and a C-N single bond.
O⁻
||
C – N
| |
R₁ H -
Structure 2 (Resonance Contributor): Here, the lone pair on nitrogen contributes to a double bond between the carbon and nitrogen, while the C=O double bond becomes a C-O single bond with a negative charge on the oxygen.
O
|
C = N⁺ – H
| |
R₁
This resonance means that the electrons are shared between the oxygen, carbon, and nitrogen atoms. Consequently, the C-N bond in the peptide linkage is shorter and stronger than a typical single bond but longer than a typical double bond. This partial double bond character restricts rotation around the C-N bond, a crucial factor in protein folding.
Planarity
The partial double bond character also forces the atoms involved in the peptide bond, along with the α-carbon atoms of the adjacent amino acids and the hydrogen attached to the nitrogen, to lie in the same plane. This planar arrangement consists of six atoms: the carbonyl carbon (C), the carbonyl oxygen (O), the amide nitrogen (N), the amide hydrogen (H), and the α-carbons of the two connected amino acids. This planarity significantly constrains the possible conformations of a polypeptide chain, influencing how it can fold into its three-dimensional structure.
Bond Angles and Distances
The peptide bond geometry is well-defined:
- Bond length of the C-N bond: Approximately 1.33 Å (Angstroms), which is intermediate between a typical C-N single bond (around 1.47 Å) and a C=N double bond (around 1.27 Å).
- Bond length of the C=O bond: Slightly longer than a typical carbonyl double bond, reflecting the partial single bond character.
- Bond length of the C-O bond: Slightly shorter than a typical C-O single bond.
The bond angles around the atoms within the peptide plane are also fixed, contributing to the rigidity of this unit. The planarity and restricted rotation make the peptide bond a stable and rigid structural element within a protein.
The “Backbone” of the Protein
When multiple amino acids are linked together, the peptide bonds, along with the α-carbon atoms and their attached hydrogens, form the repeating “backbone” of the polypeptide chain. The sequence of repeating units is often described as -N-Cα-C-, where ‘N’ represents the nitrogen atom of the amino group, ‘Cα’ is the alpha-carbon, and ‘C’ is the carbonyl carbon. The R-groups, the variable side chains, then project outwards from this backbone, allowing for diverse interactions that drive protein folding and function.
Implications for Protein Structure and Function
The characteristics of the peptide bond are not merely academic details; they have profound implications for how proteins achieve their complex three-dimensional structures and, consequently, how they perform their biological roles.
Restricted Rotation and Conformation
As mentioned, the partial double bond character of the peptide C-N bond significantly restricts rotation around this axis. Rotation is possible around the bonds connecting the α-carbon to the carbonyl carbon (the phi, φ, angle) and the α-carbon to the amino nitrogen (the psi, ψ, angle). These angles define the local conformation of the polypeptide chain. The limited rotational freedom imposed by the planar peptide bond, coupled with the steric hindrance of the R-groups, dictates the allowable conformations, playing a critical role in how a polypeptide chain folds into its native, functional structure.
Hydrogen Bonding Potential
The peptide bond possesses both a hydrogen bond donor (the N-H group) and a hydrogen bond acceptor (the C=O group). These potential hydrogen bonds are instrumental in stabilizing secondary structures, such as alpha-helices and beta-sheets. Within an alpha-helix, for instance, the N-H group of one amino acid forms a hydrogen bond with the C=O group of an amino acid four residues further down the chain. In beta-sheets, hydrogen bonds form between the C=O and N-H groups of different polypeptide strands or different regions of the same strand. These interactions, facilitated by the peptide bond’s structure, are fundamental to protein stability and conformation.
The Trans Configuration
Peptide bonds almost exclusively exist in the trans configuration, where the α-carbons of the two adjacent amino acids are on opposite sides of the peptide bond. The cis configuration, where they are on the same side, is energetically unfavorable due to steric clashes between the bulky R-groups. While there are rare exceptions, particularly when proline is involved, the overwhelming preference for the trans configuration further contributes to the predictable folding patterns of polypeptide chains.
Conclusion: A Humble Yet Powerful Link
In essence, the peptide bond, a seemingly simple amide linkage, is a cornerstone of molecular biology. Its planar structure, partial double bond character, and restricted rotation are not limitations but rather fundamental determinants of protein architecture. The way a peptide bond “looks” – its rigidity, its potential for hydrogen bonding, and its preferred trans configuration – dictates how a linear chain of amino acids can fold into the precise three-dimensional structures required for life’s diverse functions. Understanding this fundamental chemical connection is key to unraveling the complexities of protein science, from drug design to understanding genetic diseases.
