The humble 1.5-volt battery, a ubiquitous power source for countless everyday devices, often goes unnoticed in its visual simplicity. While drone enthusiasts are typically concerned with higher voltage lithium-polymer (LiPo) batteries powering their sophisticated aerial machines, understanding the common 1.5-volt cell offers a foundational perspective on battery technology. This exploration delves into the typical forms and internal structures of 1.5-volt batteries, providing context for the evolution and diversity of battery designs that ultimately lead to the powerful energy solutions used in modern drones.
The Familiar Forms: Cylindrical Cells
When one thinks of a 1.5-volt battery, the most immediate visual is the familiar cylindrical shape. These batteries, commonly referred to by their size designations, have become household staples.
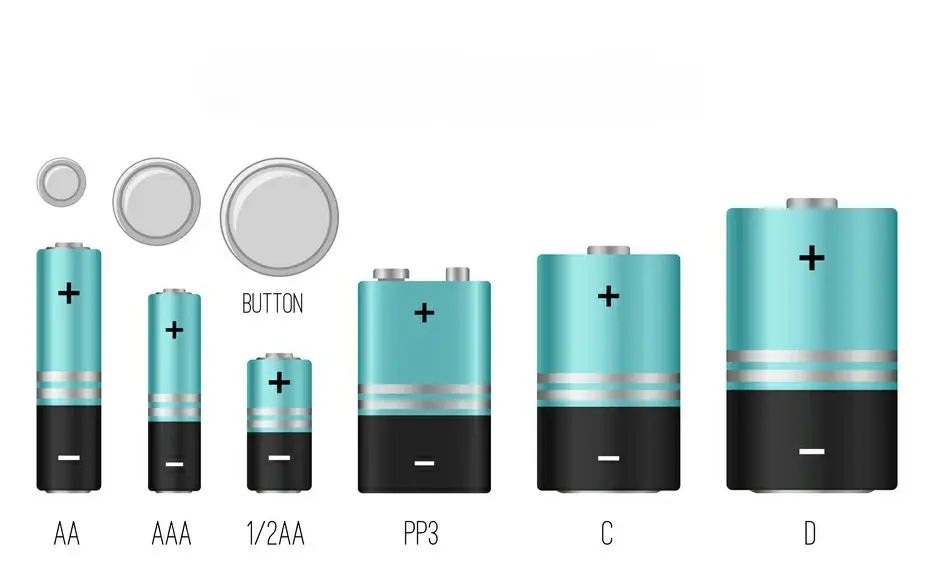
AA Batteries: The Workhorse
The AA battery is perhaps the most iconic 1.5-volt cell. Slightly larger than AAA, AAs are cylindrical with a positive terminal at one end (a small raised button) and a flat negative terminal at the other. Their dimensions are standardized, measuring approximately 50.5 mm in length and 14.5 mm in diameter. This standardized sizing ensures compatibility across a vast array of electronic devices, from remote controls and children’s toys to wall clocks and portable radios. Internally, the AA battery houses a complex electrochemical system designed to deliver a steady 1.5 volts. The common alkaline AA battery utilizes zinc as the anode and manganese dioxide as the cathode, separated by an electrolyte, typically potassium hydroxide. The cylindrical form factor is optimal for packing these components efficiently while maintaining structural integrity and ease of handling. The positive terminal is designed to make reliable contact with the positive terminal of the device, while the negative terminal connects to the device’s negative contact point.
AAA Batteries: The Compact Cousin
Smaller and more slender than AAs, AAA batteries offer a 1.5-volt output in a more compact form factor. They share the same basic cylindrical design and terminal configuration as AAs but are significantly shorter, measuring around 44.5 mm in length and 10.3 mm in diameter. This miniaturization makes them ideal for smaller, power-conscious devices such as wireless mice, compact flashlights, and certain types of remote controls. Despite their smaller size, the internal chemistry of alkaline AAA batteries is largely the same as their AA counterparts, offering a similar energy density relative to their volume. The reduced size necessitates a more tightly packed internal structure, but the fundamental principles of electrochemical energy conversion remain the same.
Other Cylindrical Variants: C, D, and N Cells
Beyond AAs and AAAs, the 1.5-volt cylindrical battery family includes other less common but still relevant sizes. C batteries, larger than AAs, and D batteries, even larger still, were historically used in higher-drain devices like flashlights, portable radios, and early electronic toys. They offer greater capacity due to their larger volume, allowing for longer operation times. N cells, on the other hand, are significantly smaller than AAAs, used in very specific low-drain applications like personal organizers or keychains. The visual distinction of these cells is primarily their size, with the positive and negative terminals maintaining the same design principle – a raised button for the positive and a flat surface for the negative. Their larger diameters and lengths accommodate more active material, leading to higher ampere-hour ratings.
Beyond Cylinders: Less Common Geometries
While cylindrical cells dominate the 1.5-volt landscape, other shapes exist, often tailored for specific applications where space or unique form factors are paramount.
Button Cells: The Low-Profile Powerhouses

Small, coin-shaped batteries, commonly known as button cells or coin cells, also often operate at 1.5 volts, although many common button cells are actually 3-volt lithium types. However, older or specialized mercury-based button cells, and some silver-oxide cells, operated at a nominal 1.5 volts. These batteries are characterized by their extremely low profile, making them suitable for very slim devices. Visually, they are flat discs with a diameter ranging from a few millimeters to over an inch, and a thickness of just a few millimeters. The positive terminal is typically the entire top surface of the cell, while the negative terminal is the flat bottom surface. Their internal construction often involves a layered arrangement of anode, cathode, and electrolyte within a sealed casing. While less common for general-purpose 1.5-volt applications today due to the prevalence of alkaline cylinders and higher-voltage lithium options, they represent an important category of compact power.
Speciality and Custom Designs
In certain niche applications, manufacturers may produce custom-shaped 1.5-volt batteries. These are less about standardized form factors and more about fitting into unique electronic devices. Visually, they can vary widely, potentially being rectangular, oval, or even custom-molded to interlock with specific components within a device. The internal chemistry would likely remain similar to standard alkaline or zinc-carbon cells to achieve the 1.5-volt output. These are less about what a “typical” 1.5-volt battery looks like and more about the engineering flexibility of battery design to meet specific product requirements.
Internal Structure: The Core of the Cell
Understanding what a 1.5-volt battery looks like on the outside is only part of the story. Its internal composition is what enables its function.
The Basic Alkaline Cell Construction
The most prevalent 1.5-volt batteries are alkaline cells. Their construction is relatively standardized:
- Casing: Typically made of steel, this cylindrical shell serves as the battery’s structural housing and also acts as the negative terminal.
- Anode (Negative Electrode): Usually powdered zinc. This is where oxidation occurs, releasing electrons.
- Cathode (Positive Electrode): Typically manganese dioxide. This is where reduction occurs, accepting electrons.
- Electrolyte: A moist alkaline paste, most commonly potassium hydroxide. This conductive medium allows ions to move between the anode and cathode, completing the electrical circuit internally.
- Separator: A porous material, often a non-woven fabric, placed between the anode and cathode to prevent short-circuiting while allowing ion flow.
- Current Collector: A metal rod or strip that connects the anode to the negative terminal of the battery.
- Positive Terminal: A cap at the top of the battery, usually made of brass or steel, which makes contact with the positive terminal of the device. It’s designed with a raised button to facilitate easy connection.
The interplay of these components within the cylindrical structure is what allows for the sustained generation of 1.5 volts through a controlled electrochemical reaction.
Zinc-Carbon (Heavy Duty) Cells
Older and less powerful than alkaline batteries are zinc-carbon cells, often labeled “Heavy Duty.” While they also output 1.5 volts, their internal chemistry and construction differ, leading to lower capacity and shelf life. They typically use a paste of ammonium chloride or zinc chloride as the electrolyte, and a carbon rod acts as the current collector for the manganese dioxide cathode. Visually, they may appear similar to alkaline cells, but their performance characteristics are distinct, and they are increasingly being replaced by alkaline technology.

The Leap to Higher Voltages for Drones
It is crucial to differentiate these common 1.5-volt cells from the batteries powering modern drones. Drone batteries are almost universally Lithium Polymer (LiPo) batteries, which operate at much higher voltages, typically ranging from 7.4 volts (2S configuration) up to 22.2 volts (6S configuration) or even higher. These higher voltages are necessary to provide the significant power required to lift and maneuver a drone, especially one equipped with cameras and other payloads. Visually, LiPo batteries are distinct, often appearing as flat, multi-celled packs with a balance connector in addition to the main power leads. The technology, chemistry, and safety requirements are vastly different from the simple 1.5-volt cells that power everyday electronics. The evolution from the basic 1.5-volt cell to the sophisticated, high-discharge LiPo batteries used in drones represents a significant advancement in electrochemical energy storage, driven by the demands of aerial mobility and advanced capabilities.
