Oligonucleotides, often abbreviated as oligos, are short, synthetic or naturally occurring strands of nucleic acids, either DNA or RNA. Their relatively small size, typically ranging from 2 to 50 nucleotides, sets them apart from the long polymer chains that constitute the genetic material of living organisms (genomic DNA) or the messenger molecules that carry genetic information for protein synthesis (mRNA). This characteristic brevity, however, belies their profound significance and burgeoning applications across a vast spectrum of scientific disciplines, particularly within the realm of biotechnology, molecular biology, and cutting-edge therapeutics.
The fundamental building blocks of oligonucleotides are nucleotides. Each nucleotide consists of three components: a phosphate group, a five-carbon sugar (deoxyribose for DNA, ribose for RNA), and a nitrogenous base (adenine, guanine, cytosine, thymine for DNA; adenine, guanine, cytosine, uracil for RNA). These nucleotides are linked together by phosphodiester bonds to form a polynucleotide chain. The sequence of these bases along the oligonucleotide chain is what dictates its unique properties and functions, much like the sequence of letters in a word defines its meaning.
The ability to synthesize oligonucleotides with precise and custom sequences has revolutionized molecular biology research. Scientists can now design and produce oligos that are complementary to specific target sequences within DNA or RNA molecules. This complementarity allows oligos to bind selectively to their intended targets through base pairing (A with T/U, and G with C). This fundamental principle underpins many powerful molecular techniques.
Applications in Molecular Biology and Diagnostics
The controlled design and synthesis of oligonucleotides have made them indispensable tools in a myriad of molecular biology applications, ranging from fundamental research to sophisticated diagnostic assays and therapeutic development. Their specificity and adaptability are key to their widespread utility.
PCR and qPCR: Amplifying Genetic Information
One of the most ubiquitous applications of oligonucleotides is in Polymerase Chain Reaction (PCR). PCR is a revolutionary technique that enables the amplification of specific DNA sequences exponentially. In a standard PCR reaction, two short, synthetic DNA oligonucleotides, known as primers, are crucial. These primers are designed to be complementary to the flanking regions of the DNA segment that needs to be amplified.
- Forward and Reverse Primers: The primers act as starting points for DNA polymerase, an enzyme that synthesizes new DNA strands. A forward primer binds to one strand of the DNA template, and a reverse primer binds to the complementary strand, defining the boundaries of the region to be copied.
- Exponential Amplification: Through repeated cycles of denaturation (separating DNA strands), annealing (primers binding to the template), and extension (DNA polymerase synthesizing new strands), the target DNA sequence is copied billions of times. This amplification is essential for detecting and analyzing even minute quantities of DNA, which is critical in fields like forensics, genetic testing, and pathogen detection.
Quantitative PCR (qPCR), also known as real-time PCR, builds upon the principles of PCR and uses oligonucleotides in conjunction with fluorescent probes or dyes to monitor the amplification process in real-time. This allows for the quantification of the initial amount of target DNA or RNA, providing valuable insights into gene expression levels or viral load.
Gene Sequencing and Genotyping
Oligonucleotides play a vital role in determining the exact order of nucleotides within a DNA or RNA molecule, a process known as gene sequencing. Sanger sequencing, a foundational method, relies on dideoxynucleotides (modified nucleotides that terminate DNA synthesis), which are essentially short, custom-synthesized DNA fragments. More modern high-throughput sequencing technologies, such as next-generation sequencing (NGS), also extensively utilize custom-designed oligonucleotides for library preparation, adapter ligation, and amplification steps, enabling the rapid sequencing of entire genomes.
Furthermore, oligonucleotides are used in genotyping, the process of determining the specific alleles (variations of a gene) an individual possesses. Techniques like DNA microarrays and SNP (single-nucleotide polymorphism) arrays employ thousands of immobilized oligonucleotides, each designed to bind to a specific genetic variant. By observing which oligos hybridize to a sample’s DNA, researchers can infer an individual’s genotype.
Blotting Techniques: Visualizing Nucleic Acids
Techniques like Southern blotting (for DNA) and Northern blotting (for RNA) also depend on the specificity of oligonucleotide probes. These methods involve transferring separated nucleic acid fragments onto a membrane, followed by hybridization with a labeled oligonucleotide probe that is complementary to the target sequence of interest. The label (often radioactive or fluorescent) allows researchers to detect and visualize the presence and location of the target nucleic acid on the membrane, providing insights into gene structure, expression, and organization.
Diagnostic Assays and Biosensors
The exquisite specificity of oligonucleotide hybridization makes them ideal for diagnostic applications. Diagnostic kits for detecting infectious diseases, genetic predispositions to certain conditions, or cancer biomarkers often employ oligonucleotides. For instance, in nucleic acid testing (NAT), primers and probes are used to amplify and detect the genetic material of pathogens like viruses (e.g., HIV, SARS-CoV-2) or bacteria.
Oligonucleotide-based biosensors are also emerging as powerful tools for rapid and sensitive detection of analytes. These sensors leverage the binding affinity of oligonucleotides to their targets to generate a measurable signal, which can be electrical, optical, or acoustic. This technology holds promise for point-of-care diagnostics, environmental monitoring, and food safety.
Therapeutic Applications: The Rise of Oligonucleotide Drugs
Beyond their utility in research and diagnostics, oligonucleotides are increasingly at the forefront of therapeutic innovation. Their ability to precisely interact with genetic material opens up new avenues for treating a wide range of diseases, particularly those with a genetic or molecular basis.
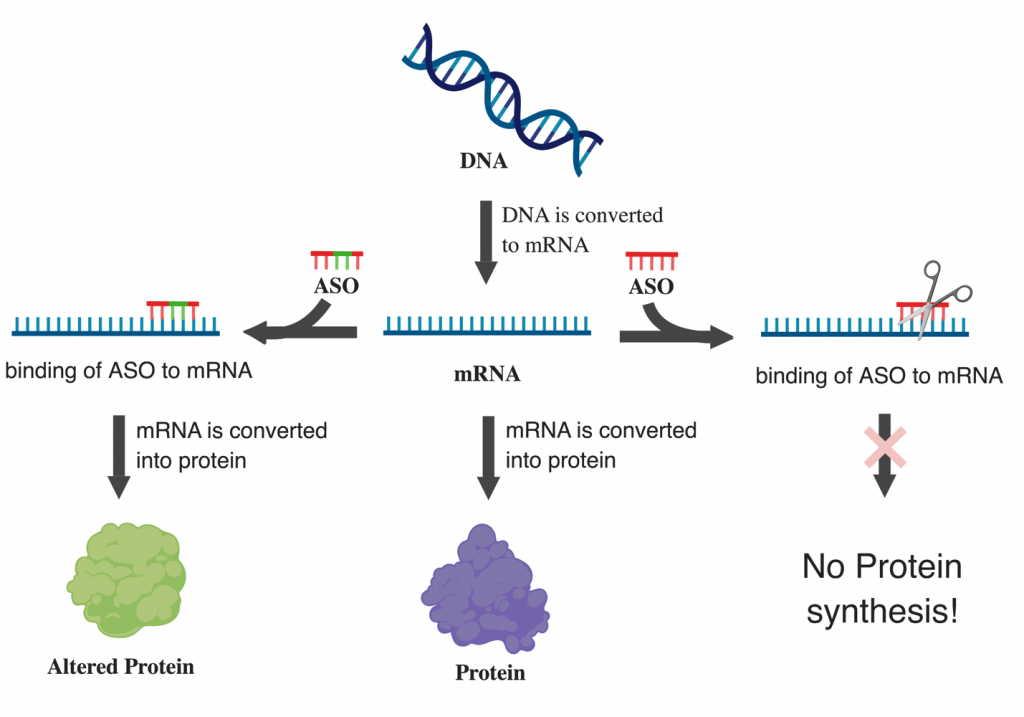
Antisense Oligonucleotides (ASOs)
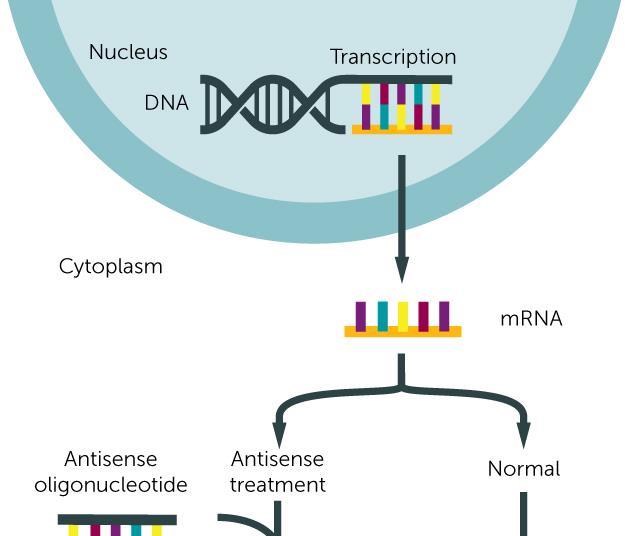
Antisense oligonucleotides are single-stranded DNA or RNA molecules designed to bind to a specific messenger RNA (mRNA) molecule. By binding to the target mRNA, ASOs can interfere with its function in several ways:
- Degradation of mRNA: Some ASOs recruit cellular enzymes that degrade the bound mRNA, effectively silencing the gene.
- Blocking Translation: Other ASOs can sterically hinder the ribosome from translating the mRNA into a protein, thereby reducing protein production.
- Modulating Splicing: Certain ASOs can alter the way pre-mRNA is spliced, leading to the production of different protein isoforms or preventing the formation of non-functional proteins.
This targeted approach allows ASOs to modulate gene expression at the post-transcriptional level, offering a way to treat diseases caused by the overproduction or malfunction of specific proteins. Several ASO-based drugs have already been approved for treating conditions such as spinal muscular atrophy (SMA), hereditary transthyretin amyloidosis, and Duchenne muscular dystrophy.
Small Interfering RNAs (siRNAs)
Small interfering RNAs (siRNAs) are another class of therapeutic oligonucleotides that function through RNA interference (RNAi). siRNAs are typically short, double-stranded RNA molecules that are processed by cellular machinery into single-stranded guide RNAs. These guide RNAs then direct a protein complex called the RNA-induced silencing complex (RISC) to a complementary mRNA target. Once bound, RISC cleaves the target mRNA, leading to its degradation and subsequent silencing of the corresponding gene.
siRNA therapeutics are being developed for a broad array of diseases, including viral infections, genetic disorders, and cancers. Their potent gene-silencing capabilities offer a promising strategy for targeting diseases where specific protein production needs to be suppressed.
Aptamers: Molecular Recognition Molecules
Aptamers are short, single-stranded oligonucleotides (DNA or RNA) that can fold into complex three-dimensional structures capable of binding to specific target molecules with high affinity and specificity, much like antibodies. This process of selecting aptamers is known as SELEX (Systematic Evolution of Ligands by Exponential Enrichment).
Aptamers can bind to a diverse range of targets, including proteins, small molecules, and even cells. Their therapeutic applications are rapidly expanding, with aptamers being explored as:
- Anticoagulants: By binding to specific coagulation factors.
- Anticancer agents: By targeting tumor-specific proteins or delivering cytotoxic payloads.
- Diagnostic agents: For detecting disease biomarkers.
- Antiviral agents: By inhibiting viral entry or replication.
The advantages of aptamers include their relatively easy synthesis, stability, and low immunogenicity compared to antibodies.
CRISPR-Cas Gene Editing: The Role of Guide RNAs
While not strictly therapeutic oligonucleotides in the same vein as ASOs or siRNAs, the guide RNA (gRNA) component of the CRISPR-Cas gene editing system is a crucial oligonucleotide. The gRNA directs the Cas9 enzyme to a specific DNA sequence, enabling precise cutting of the genome. Researchers are leveraging this technology to correct genetic mutations, disable disease-causing genes, and introduce new genetic material. The precise design and synthesis of these gRNAs are fundamental to the success and specificity of CRISPR-based gene editing therapies.
Chemical Modifications and Delivery Challenges
The inherent nature of oligonucleotides presents certain challenges that need to be addressed for their effective therapeutic use. Their susceptibility to degradation by nucleases (enzymes that break down nucleic acids) in the bloodstream and their inability to readily cross cell membranes are significant hurdles.
To overcome these limitations, chemists have developed a wide array of chemical modifications that can be incorporated into the oligonucleotide backbone or bases. These modifications can:
- Increase Nuclease Resistance: Modifications like phosphorothioate linkages (replacing a non-bridging oxygen with sulfur) or 2′-O-methyl modifications enhance stability against enzymatic degradation.
- Improve Cellular Uptake: Conjugating oligonucleotides with specific ligands (e.g., GalNAc for liver targeting) or incorporating specific chemical motifs can facilitate their entry into cells.
- Enhance Binding Affinity: Certain modifications can increase the binding strength of the oligonucleotide to its target, improving its efficacy.
- Reduce Off-Target Effects: Careful design and modifications can help minimize unintended interactions with other nucleic acid sequences, thus reducing potential side effects.
Delivery remains a critical aspect of oligonucleotide therapeutics. Strategies include systemic administration with appropriate chemical modifications and delivery vehicles (e.g., lipid nanoparticles, viral vectors) or localized delivery to specific tissues or organs. The liver is a primary target for many oligonucleotide therapies due to its high uptake of modified oligonucleotides and its role in metabolism.
The Future Landscape of Oligonucleotide Technology
The field of oligonucleotide science is dynamic and rapidly evolving. Ongoing research is focused on developing novel oligonucleotide chemistries, improving delivery systems, and expanding the range of therapeutic targets. The increasing understanding of the complex regulatory mechanisms of gene expression, coupled with advances in synthesis and computational design, portends a future where oligonucleotides play an even more central role in medicine.
From unraveling the mysteries of the genome to offering groundbreaking treatments for previously intractable diseases, oligonucleotides stand as a testament to the power of molecular design and chemical ingenuity. As research continues to push the boundaries of what’s possible, these short nucleic acid sequences are poised to revolutionize healthcare and our understanding of life itself.
