The density of a material is a fundamental physical property that describes how much mass is contained within a given volume. For silver, a precious metal highly valued for its conductivity, reflectivity, and aesthetic appeal, understanding its density is crucial across a range of scientific, industrial, and even artistic applications. When discussing density, it is typically expressed in units of mass per unit volume. For silver, this is most commonly measured in grams per cubic centimeter (g/cm³) or kilograms per cubic meter (kg/m³). This article delves into the precise density of silver in grams, exploring the factors that influence it and its significance in various contexts.
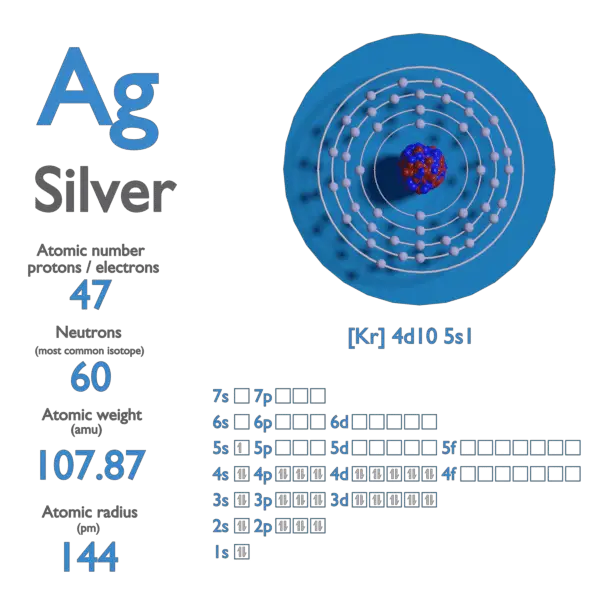
The Intrinsic Density of Pure Silver
At its core, the density of silver is an intrinsic property of the element itself. Pure elemental silver (Ag), with an atomic number of 47, has a well-defined crystalline structure. This structure, coupled with the atomic mass of silver, dictates its bulk density.
Standard Temperature and Pressure (STP) Considerations
Scientific measurements are often standardized to specific conditions to ensure comparability. For density, the most common standard conditions are Standard Temperature and Pressure (STP). STP is defined by IUPAC (International Union of Pure and Applied Chemistry) as a temperature of 273.15 K (0 °C, 32 °F) and a pressure of 100 kPa (1 bar, approximately 0.987 atm).
Under these conditions, the density of pure, solid silver is approximately 10.49 grams per cubic centimeter (g/cm³). This value is a widely accepted and scientifically determined figure.
Variations in Density Measurement
While 10.49 g/cm³ is the standard, it’s important to note that slight variations can occur due to several factors:
- Purity: The presence of impurities, even in small amounts, can alter the density. Alloys of silver, such as sterling silver (which contains 92.5% silver and 7.5% other metals, typically copper), will have a different density than pure silver. Copper, for instance, is less dense than silver, so sterling silver will have a slightly lower density than pure silver.
- Temperature: Density is temperature-dependent. As temperature increases, materials generally expand, meaning their volume increases while their mass remains constant. This leads to a decrease in density. Conversely, at lower temperatures, materials contract, and their density increases. While the change in density of solid silver with typical temperature fluctuations is relatively small, it is a factor in highly precise measurements.
- Pressure: Pressure also affects density, though its impact on solid materials like silver is less pronounced than on gases. Increased pressure can compress a material, leading to a slight increase in density.
- Crystal Structure and Allotropy: While silver primarily exists in a face-centered cubic (FCC) crystal structure under normal conditions, extreme pressures or temperatures could theoretically induce allotropic transformations, leading to different crystalline forms with potentially different densities. However, for practical purposes, the FCC structure is the relevant one.
Understanding the Grams Unit
The unit “grams” in the context of density refers to the mass component. A cubic centimeter (cm³) is a unit of volume. Therefore, a density of 10.49 g/cm³ signifies that a 1-centimeter cube of pure silver would have a mass of approximately 10.49 grams. This provides a tangible understanding of how much “stuff” is packed into a given space.
Factors Influencing Silver’s Density in Real-World Applications
Beyond the theoretical density of pure silver, real-world applications often involve silver in forms or alloys that can exhibit slightly different density values. This understanding is critical for engineers, metallurgists, and designers.
Alloys and Impurities
As mentioned, most silver used in jewelry, silverware, and certain industrial applications is not 100% pure. Sterling silver is the most common example. The addition of copper to silver increases its hardness and durability, making it more practical for everyday use. Because copper has a density of approximately 8.96 g/cm³, sterling silver will have a density lower than pure silver. The exact density of sterling silver can vary slightly depending on the precise alloy composition, but it typically falls in the range of 9.8 to 10.3 g/cm³.
Other silver alloys might be used for specific properties:
- Silver-Gold Alloys: These might be used in specialized electronics or dentistry and would have a density influenced by both metals.
- Silver-Copper-Germanium Alloys: Used in brazing, their density would also be an average of the constituent elements.
When calculating the density of an alloy, a weighted average based on the mass fractions of each component element can provide a good approximation.
Manufacturing Processes
The manufacturing process can also subtly influence the density of silver components:
- Powder Metallurgy: If silver is processed using powder metallurgy techniques, the density achieved can depend on the particle size, shape, and the degree of compaction during sintering. Porosity within the material can reduce the overall bulk density.
- Casting and Machining: While casting generally produces dense materials, microscopic voids or inclusions can sometimes occur, slightly affecting density. Machining, while removing material, doesn’t inherently change the density of the remaining material itself but can alter the overall density of the finished part if significant material is removed unevenly.
- Nanostructured Silver: In advanced applications, silver might be engineered at the nanoscale. The density of nanostructured materials can sometimes deviate from bulk materials due to surface effects and different packing arrangements of nanoparticles, though this is a specialized area.
Porosity and Surface Treatments
The presence of internal voids or porosity significantly impacts the effective density of a silver object. A porous piece of silver will have a lower bulk density than a solid piece of the same material. Surface treatments, such as plating or the application of coatings, can also add mass to the object, potentially altering its measured density if not accounted for.
Practical Significance of Silver’s Density
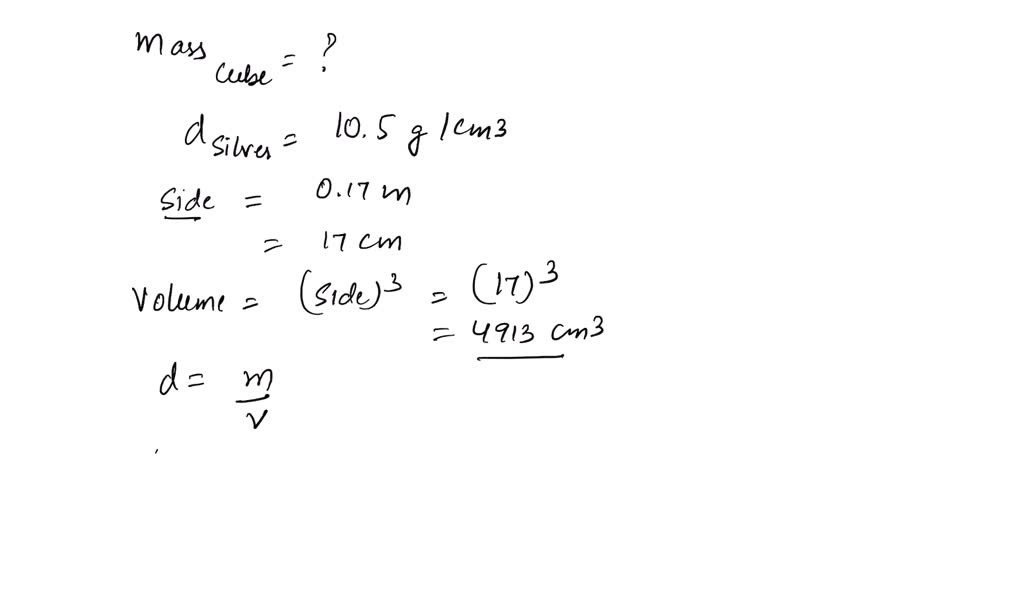
The density of silver, expressed in grams, has profound implications across numerous fields. It’s not just an academic curiosity but a practical parameter that informs design, manufacturing, and application.
Metallurgy and Material Science
In metallurgy, density is a key indicator of material quality and composition.
- Alloy Verification: Measuring the density of a sample can be a quick way to verify its composition or detect significant deviations from the expected alloy.
- Process Control: For manufacturers, consistent density is often a critical quality control parameter. Variations could indicate issues in the melting, casting, or alloying process.
- Material Selection: When designing components where weight is a factor (e.g., in aerospace or specialized electronics), knowing the density is essential for calculating mass and ensuring structural integrity.
Jewelry and Precious Metals
For precious metals, density plays a vital role in valuation and authenticity.
- Hallmarking and Purity: While hallmarking guarantees purity, density can be used as a supplementary check. A piece claiming to be pure silver but found to be significantly less dense might warrant further investigation.
- Weight and Value: The value of precious metals is often tied to their weight. Knowing the density allows for accurate calculation of the amount of silver present in an item, contributing to its appraisal and market price. For instance, a solid silver statue would have a predictable weight based on its dimensions and the density of silver.
Electronics and Conductivity
Silver’s exceptional electrical and thermal conductivity makes it indispensable in electronics.
- Component Design: In applications like high-frequency connectors, electrical contacts, and superconducting elements, the precise mass and volume of silver components are critical. Density calculations help determine the exact amount of silver needed for a specific function and to manage weight in compact electronic devices.
- Thermal Management: Silver’s high thermal conductivity means it’s used in heat sinks and thermal interface materials. Its density influences the mass and thermal capacity of these components.
Scientific Instrumentation and Measurement
In scientific research and metrology, precision is paramount.
- Calibration Standards: High-purity silver can be used as a reference material in certain calibration processes. Its known density contributes to the accuracy of these standards.
- Buoyancy and Hydrostatics: The principles of buoyancy, which depend on density, are used in various measurement techniques. For instance, Archimedes’ principle can be employed to determine the density of an unknown substance by measuring its mass and the mass of the water it displaces. Conversely, knowing the density of silver is crucial if it’s part of a scientific apparatus where buoyancy effects are relevant.
Art and Sculpture
Artists and sculptors working with silver appreciate its properties.
- Material Estimation: For large sculptures or intricate pieces, understanding the volume and density allows artists to estimate the amount of silver required and the final weight of the artwork. This is important for structural planning and transportation.
- Aesthetic Qualities: While not directly a density application, the metallic luster and malleability of silver, coupled with its density, contribute to the perceived solidity and value of artistic creations.
Quantifying Density: Grams per Cubic Centimeter
To reiterate, the standard and most commonly cited density for pure silver at room temperature and standard pressure is approximately 10.49 grams per cubic centimeter (g/cm³). This figure is derived from fundamental physics and chemistry.
The Concept of Specific Gravity
Related to density is the concept of specific gravity. Specific gravity is the ratio of the density of a substance to the density of a reference substance, usually water. Since the density of water is approximately 1 g/cm³ at 4 °C, the specific gravity of a substance is numerically very close to its density in g/cm³. For pure silver, the specific gravity is also approximately 10.49.
Measurement Techniques
Determining the density of a silver sample can be achieved through various methods:
- Direct Measurement: This involves precisely measuring the mass of the sample using a calibrated balance and its volume using geometric measurements (for regular shapes) or displacement methods (like Archimedes’ principle using water or another liquid of known density).
- Mass Measurement: A high-precision digital scale is used to determine the mass in grams.
- Volume Measurement:
- For Regular Shapes: If the silver object is a cube, cylinder, or other precisely definable geometric form, its dimensions can be measured with calipers or micrometers to calculate its volume in cm³.
- For Irregular Shapes (Archimedes’ Principle): The sample is first weighed in air. Then, it is submerged in a liquid (typically distilled water) of known density, and its apparent weight is measured. The difference in weight (buoyant force) is equal to the weight of the displaced liquid. Knowing the density of the liquid, the volume of the displaced liquid, and thus the volume of the silver sample, can be calculated.
- X-ray Diffraction (XRD): For highly precise measurements of crystalline materials, XRD can determine the lattice parameters of the crystal structure. Knowing the atomic mass of silver and the dimensions of its unit cell, the theoretical density can be calculated with great accuracy.

The Importance of Precision
In specialized fields like microelectronics manufacturing, nanotechnology, or advanced material science, even minor deviations in density can have significant consequences. For instance, in the production of precise medical implants or high-performance electrical contacts, materials are often selected and processed to achieve a very specific density profile.
In summary, the density of pure silver in grams, a cornerstone of its material properties, is approximately 10.49 g/cm³ under standard conditions. This value, while seemingly straightforward, is influenced by purity, temperature, and manufacturing processes, making it a critical parameter for a vast array of scientific, industrial, and artistic endeavors. Understanding and accurately measuring this density ensures the integrity, functionality, and value of silver in its myriad applications.
