Agitation, a seemingly simple concept, plays a profoundly critical role in the realm of chemistry. At its core, agitation refers to the process of stirring or shaking a substance or a mixture to enhance its properties, facilitate reactions, or achieve specific physical states. While often taken for granted, the deliberate application of agitation is a cornerstone of countless chemical processes, from laboratory-scale experiments to large-scale industrial manufacturing. Understanding the principles and applications of agitation is essential for chemists and chemical engineers seeking to optimize efficiency, yield, and product quality. This exploration delves into the multifaceted nature of agitation in chemistry, examining its fundamental mechanisms, diverse applications, and the advanced technologies that govern its precise control.
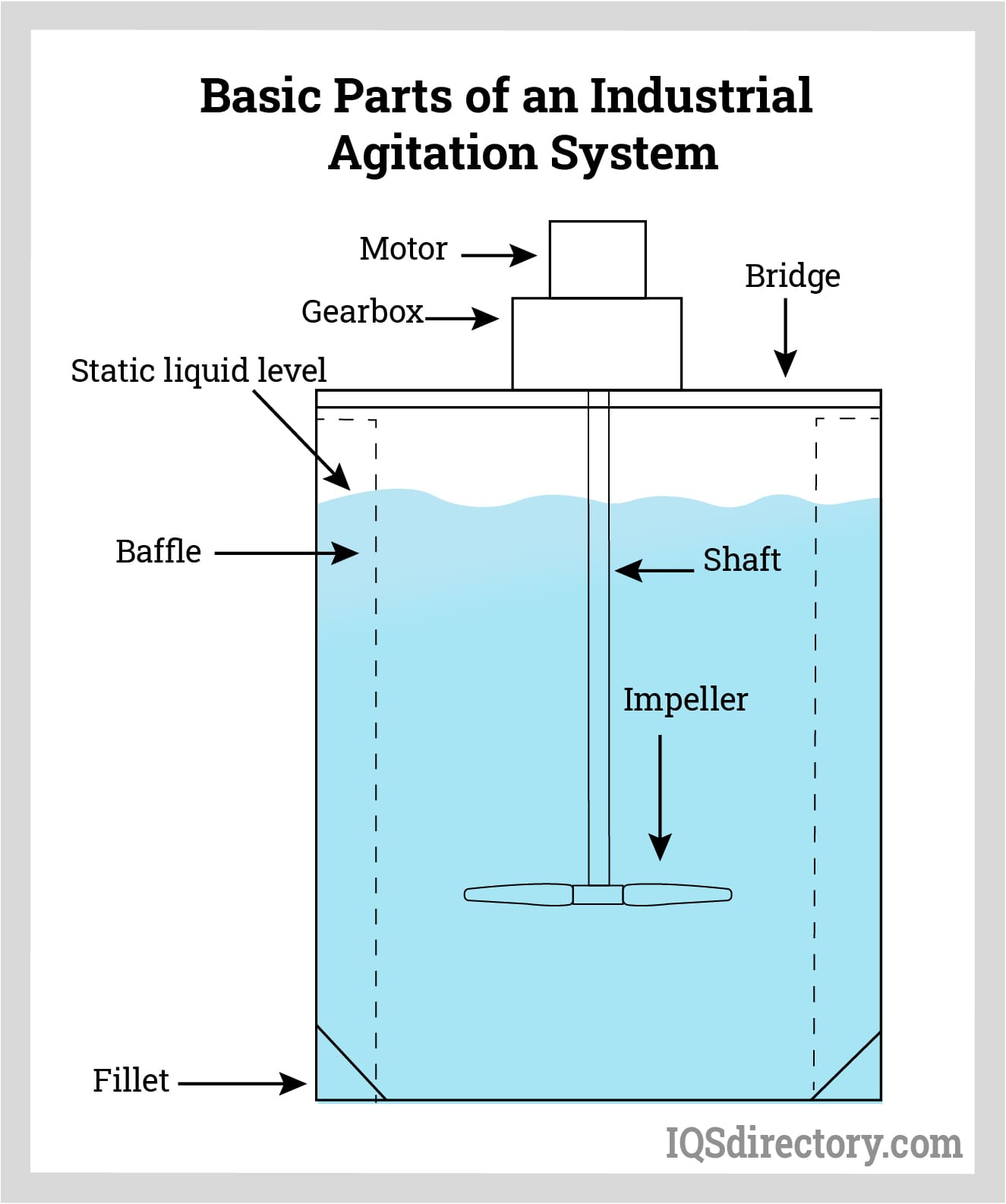
The Fundamental Science Behind Agitation
At its most basic, agitation introduces kinetic energy into a system, inducing motion among its constituent particles. This motion serves several vital purposes, primarily related to enhancing mass and heat transfer.
Mass Transfer Enhancement
In heterogeneous systems, where different phases (solid-liquid, liquid-liquid, gas-liquid) exist, the interface between these phases is often the bottleneck for reaction rates. Agitation effectively breaks down larger particles or droplets, creating a larger surface area for interaction. This increased interfacial area allows reactants to come into closer contact more rapidly, accelerating the rate at which they can diffuse across the boundaries and react.
Consider a solid catalyst suspended in a liquid reactant. Without agitation, only the outer surface of the catalyst is exposed, leading to slow reaction rates. Stirring suspends the catalyst particles throughout the liquid, bringing fresh reactant to the catalyst surface and carrying away products. Similarly, in immiscible liquid systems, agitation creates smaller droplets of one liquid dispersed within the other, dramatically increasing the surface area for mass transfer between the two phases.
Heat Transfer Improvement
Chemical reactions often generate or consume heat. Efficient removal or addition of heat is crucial for controlling reaction temperature, preventing runaway reactions, and ensuring optimal product formation. Agitation plays a significant role in heat transfer by:
- Reducing Thermal Gradients: By homogenizing the bulk of the fluid, agitation prevents the formation of hot or cold spots. This uniform temperature distribution is vital for consistent reaction kinetics and preventing localized degradation of reactants or products.
- Increasing Heat Transfer Coefficient: Agitation promotes turbulent flow in the fluid, which breaks up stagnant boundary layers that form at heat exchange surfaces (e.g., the walls of a reactor or immersed heating/cooling coils). This turbulence enhances the rate at which heat can be transferred between the fluid and the surface.
Improved Mixing and Homogenization
Many chemical processes require thorough mixing of reactants to ensure uniform concentration and prevent side reactions or incomplete conversion. Agitation achieves this by:
- Dispersion: Breaking down aggregates of solid particles or creating fine emulsions of immiscible liquids.
- Distribution: Spreading components evenly throughout the mixture.
- Homogenization: Achieving a uniform concentration and temperature throughout the reactor volume.
The degree of mixing achieved by agitation can range from simple blending to complex homogenization, depending on the type of agitator, its speed, and the rheological properties of the fluid.
Diverse Applications of Agitation in Chemical Processes
The utility of agitation spans the entire spectrum of chemical operations. Its presence is felt in research laboratories, pilot plants, and full-scale industrial facilities across numerous sectors.
Reaction Engineering
This is perhaps the most direct and critical application of agitation. In stirred-tank reactors, the impeller’s design and speed are carefully chosen to ensure:
- Reactant Contact: Adequate mixing of liquid-liquid, solid-liquid, or gas-liquid reactants to achieve desired reaction rates.
- Temperature Control: Efficient heat removal or addition to maintain optimal reaction temperature.
- Suspension of Solids: Keeping solid catalysts or reactants uniformly dispersed.
- Gas Dispersion: Breaking down gas bubbles for improved mass transfer in gas-liquid reactions.
Separation Processes
Agitation is integral to various separation techniques:
- Extraction: In liquid-liquid extraction, agitation creates intimate contact between two immiscible phases, facilitating the transfer of a solute from one phase to the other. The mixture is then allowed to settle, and the phases are separated.
- Crystallization: Gentle agitation during crystallization can control crystal size distribution and prevent agglomeration, leading to purer and more easily filterable crystals. Vigorous agitation might be used to induce nucleation or break down agglomerates.
- Dissolution: Agitation significantly speeds up the dissolution of solid solutes in liquids by continuously bringing fresh solvent into contact with the solid surface and sweeping away saturated solution.
- Filtration and Sedimentation: While not direct agitation, the preceding mixing steps that prepare slurries for filtration or sedimentation often involve agitation to ensure uniform suspension.
Material Processing
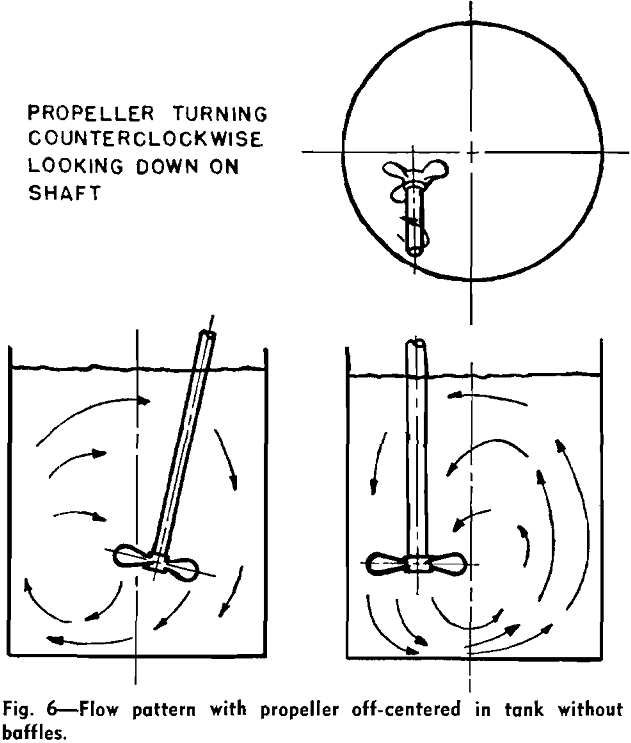
Beyond reactions and separations, agitation is crucial for modifying the physical properties of materials:
- Blending and Formulation: In the production of paints, cosmetics, pharmaceuticals, and food products, agitation is essential for creating homogeneous mixtures of various ingredients, ensuring consistent texture, color, and efficacy.
- Drying: In certain drying operations, agitated dryers (like paddle dryers or rotary dryers) tumble and mix the material, exposing fresh surfaces to the drying medium and accelerating the removal of moisture.
- Gas Absorption: In scrubbing towers and absorption columns, agitation (often in the form of bubble agitation or mechanical stirring) is used to increase the contact area between a gas and a liquid, facilitating the absorption of gaseous components into the liquid.
Types of Agitators and Their Design Considerations
The effectiveness of agitation is highly dependent on the type of agitator used and its operational parameters. The selection process involves considering the fluid properties, vessel geometry, and the specific process objectives.
Impeller Types
Impellers are the rotating components that generate the flow and turbulence. Common types include:
- Propellers: High speed, axial flow, suitable for low-viscosity fluids and general blending.
- Pitched-Blade Turbines (PBT): Axial flow, good for blending, solid suspension, and gas dispersion. Offers a balance between flow and turbulence.
- Flat-Blade Turbines (FBT) / Rushton Turbines: Radial flow, high shear, good for gas dispersion and high-viscosity mixing where significant turbulence is needed.
- Anchor Impellers: Move slowly along the vessel wall, excellent for high-viscosity fluids and heat transfer, preventing material build-up.
- Helical Ribbon Impellers: Create axial flow and good axial blending, ideal for very high viscosity materials.
Baffle Systems
Baffles are stationary plates installed on the reactor walls that disrupt the swirling motion of the fluid, forcing it to flow radially and axially. This interruption promotes turbulence and improves mixing efficiency, especially in tall vessels. Without baffles, the fluid would simply rotate with the impeller, leading to poor axial mixing and reduced energy transfer.
Design Parameters
Key parameters influencing agitation performance include:
- Impeller Diameter (D) and Vessel Diameter (T): The D/T ratio is crucial for determining flow patterns and mixing intensity.
- Impeller Speed (N): Directly relates to the kinetic energy imparted to the fluid.
- Impeller Type and Number of Blades: Dictates the flow regime (axial vs. radial) and shear.
- Baffle Configuration: Affects the degree of turbulence and flow patterns.
- Fluid Properties: Viscosity, density, and non-Newtonian behavior significantly impact agitator selection and performance.
Advanced Control and Measurement of Agitation
In modern chemical engineering, agitation is not merely about spinning a propeller. Sophisticated systems are employed to precisely control and monitor agitation parameters for optimal outcomes.
Computational Fluid Dynamics (CFD)
CFD simulations are invaluable tools for designing and optimizing agitator systems. By modeling fluid flow patterns, shear rates, and mixing times within a specific vessel geometry and with given fluid properties, engineers can:
- Predict mixing performance before physical construction.
- Optimize impeller design and placement.
- Identify potential dead zones or areas of poor mixing.
- Scale up mixing processes from laboratory to industrial scale with greater confidence.
Rheological Characterization
Understanding how a fluid behaves under shear is critical. Rheometers are used to measure viscosity, yield stress, and other rheological parameters. This data is essential for selecting the appropriate agitator type and speed for non-Newtonian fluids, which exhibit viscosity that changes with the rate of shear.
Process Analytical Technology (PAT)
PAT tools, such as in-situ probes for monitoring concentration, temperature, or particle size, can be integrated with agitation systems. This allows for real-time feedback and adjustment of agitation parameters to maintain optimal conditions and ensure consistent product quality. For instance, if a probe detects incomplete dissolution, the agitation speed can be automatically increased.

Scale-Up Challenges
Scaling up an agitated process from a small laboratory beaker to a large industrial reactor presents significant challenges. Simply multiplying dimensions and speeds often leads to different mixing characteristics due to changes in surface-area-to-volume ratios and flow regimes. Engineers rely on dimensionless numbers (like the Power Number and Reynolds Number) and CFD to maintain similar mixing intensity and efficiency during scale-up.
In conclusion, agitation is a fundamental yet sophisticated aspect of chemical engineering. Its impact on reaction kinetics, separation efficiency, and material processing is undeniable. By understanding the underlying scientific principles, carefully selecting appropriate agitator designs, and leveraging advanced control technologies, chemists and engineers can harness the power of agitation to drive innovation and achieve superior results across a vast array of chemical applications.
