The Microbiome’s Window into Health
The human gut microbiome, a complex ecosystem of trillions of microorganisms residing primarily in the large intestine, plays a profound role in our overall health. Far from being passive inhabitants, these microbes are active participants in digestion, nutrient absorption, immune system development, and even the regulation of mood and behavior. Consequently, analyzing stool samples, essentially a collection of these microbial communities and their byproducts, offers a powerful and non-invasive method to gain insights into the intricate workings of our gut and, by extension, our systemic health. This article delves into the diverse applications of stool analysis, highlighting its capacity to detect a wide array of conditions and inform personalized health strategies.
Unveiling Pathogens and Infections
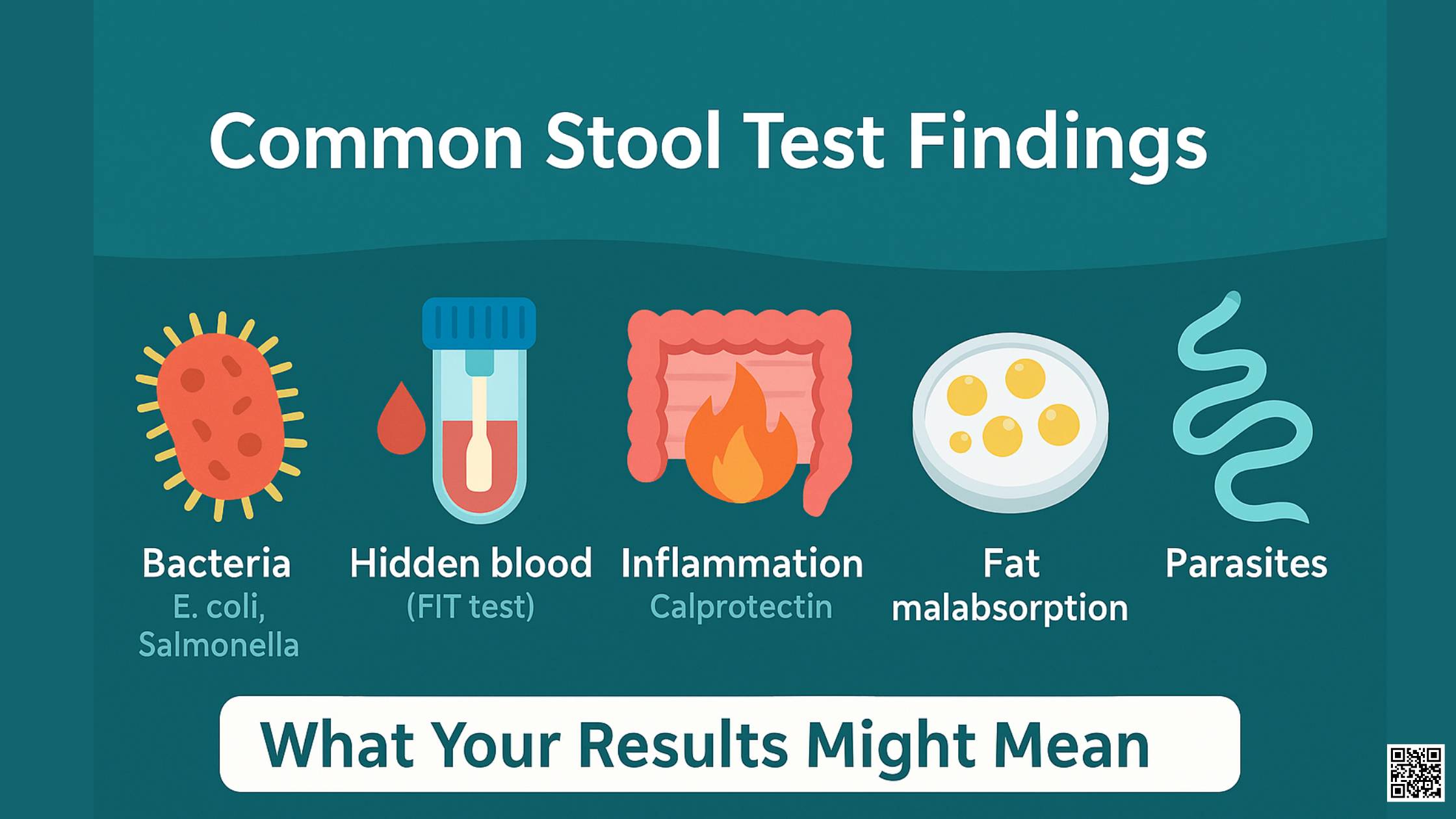
One of the most direct and long-standing applications of stool analysis is the identification of infectious agents. Pathogenic bacteria, viruses, and parasites can cause a spectrum of gastrointestinal illnesses, ranging from mild food poisoning to severe, life-threatening infections. Traditional methods, such as microscopy and culture-based techniques, are still valuable for detecting common culprits like Salmonella, Shigella, Campylobacter, and E. coli O157:H7, which are frequent causes of bacterial gastroenteritis.
However, advancements in molecular diagnostics have revolutionized the detection of infectious agents. Polymerase Chain Reaction (PCR) and other nucleic acid amplification tests (NAATs) allow for the rapid and highly sensitive detection of viral pathogens, including norovirus and rotavirus, which are common causes of outbreaks. Furthermore, these techniques can identify protozoa like Giardia lamblia and Cryptosporidium, which are often acquired from contaminated water or food.
Beyond identifying the specific causative agent, stool analysis can also provide clues about the severity and nature of an infection. The presence of white blood cells (leukocytes) in stool, for instance, often indicates an inflammatory or invasive bacterial infection, prompting a different clinical approach than a purely secretory diarrhea. Similarly, occult blood testing can reveal subtle bleeding, which might be associated with conditions like inflammatory bowel disease or even certain types of colorectal cancer, necessitating further investigation.
Assessing Digestive Function and Malabsorption
The ability of the digestive system to break down and absorb nutrients is critical for maintaining health. Stool analysis offers valuable insights into various aspects of digestive function and can help identify conditions characterized by malabsorption.
One key marker is the presence and quantity of fat in the stool (fecal fat). Excessive fat in the stool, known as steatorrhea, is a hallmark of malabsorption and can be caused by a variety of conditions. Pancreatic insufficiency, where the pancreas does not produce enough digestive enzymes, is a common cause, often seen in cystic fibrosis or chronic pancreatitis. Bile acid malabsorption, which impairs fat digestion, can result from liver disease, gallbladder removal, or damage to the small intestine. Celiac disease, an autoimmune disorder triggered by gluten, also leads to damage in the small intestine, impairing nutrient absorption, including fats. Measuring fecal fat can guide diagnostic efforts toward these underlying causes.
Beyond fat, stool analysis can also assess the digestion of other macronutrients and the presence of inflammation indicative of damage. Elastase levels in stool, for example, are a sensitive marker of pancreatic exocrine function. Low elastase levels strongly suggest pancreatic insufficiency, prompting investigation into its cause. Undigested carbohydrates can also be detected, indicating malabsorption issues with sugars like lactose or fructose. The presence of specific inflammatory markers, such as calprotectin, is particularly important in the context of inflammatory bowel disease (IBD), such as Crohn’s disease and ulcerative colitis. Calprotectin is released by activated neutrophils in the intestinal lining, and elevated levels correlate with active inflammation, helping to distinguish IBD from other causes of diarrhea and monitor treatment response.
The Microbiome’s Intricate Balance: Dysbiosis and Beyond
The advent of advanced sequencing technologies, particularly 16S rRNA gene sequencing and shotgun metagenomics, has revolutionized our understanding of the gut microbiome. These techniques allow for the comprehensive profiling of the microbial communities present in stool, identifying not only the types of bacteria, archaea, fungi, and viruses but also their relative abundance and functional potential. This has opened up a new frontier in detecting imbalances, collectively termed dysbiosis, which are increasingly linked to a wide range of health conditions.
Dysbiosis is characterized by alterations in the composition and function of the gut microbiome, often involving a decrease in beneficial microbes and an increase in potentially harmful ones. This imbalance can arise from various factors, including diet, antibiotic use, stress, and underlying diseases.
Key insights from microbiome analysis include:
- Reduced Diversity: A healthy microbiome is characterized by high diversity. A significant reduction in the variety of microbial species can be an indicator of vulnerability to certain diseases or a sign of ongoing inflammation.
- Altered Ratios: The relative abundance of different microbial groups can be highly informative. For example, an overgrowth of certain bacteria, such as Clostridium difficile after antibiotic treatment, can lead to severe gastrointestinal issues. Conversely, a depletion of beneficial bacteria like Bifidobacterium and Lactobacillus species is often observed in individuals with irritable bowel syndrome (IBS) or IBD.
- Functional Capacity: Shotgun metagenomics goes beyond identifying species to analyze the genes present in the microbiome. This provides insights into the metabolic capabilities of the microbial community, revealing whether they are producing beneficial short-chain fatty acids (SCFAs) like butyrate, which nourish gut cells and have anti-inflammatory properties, or producing potentially harmful metabolites.
- Links to Chronic Diseases: Emerging research has implicated microbiome dysbiosis in a growing list of chronic conditions beyond the gut. These include metabolic disorders like obesity and type 2 diabetes, autoimmune diseases, neurological conditions such as Parkinson’s disease and even depression and anxiety through the gut-brain axis. Stool analysis is a primary tool for investigating these connections.
By characterizing the microbiome’s composition and function, stool samples can help identify individuals at risk for developing these conditions or provide targeted therapeutic strategies aimed at restoring microbial balance, such as through probiotics, prebiotics, or fecal microbiota transplantation (FMT).
Early Detection of Colorectal Cancer
Perhaps one of the most critical applications of stool analysis is in the early detection of colorectal cancer (CRC). Colorectal cancer often develops from precancerous polyps, which can bleed over time. This bleeding may not be visible to the naked eye, making stool-based screening tests invaluable for early detection when the cancer is most treatable.
Two primary types of stool-based CRC screening tests exist:
- Fecal Occult Blood Test (FOBT): These tests detect the presence of hidden blood in the stool. There are two main types: guaiac-based FOBT (gFOBT) and fecal immunochemical test (FIT). FIT is generally preferred due to its higher specificity and sensitivity, as it specifically detects human hemoglobin, unaffected by dietary influences.
- Stool DNA Tests: These more advanced tests detect both occult blood and altered DNA shed from cancerous or precancerous cells in the stool. By identifying specific genetic mutations or changes associated with CRC, these tests offer a higher sensitivity for detecting advanced adenomas and early-stage cancers.
Regular screening with these stool-based tests is recommended for individuals over a certain age or those with increased risk factors. A positive result from any of these stool tests does not confirm cancer but indicates the need for a diagnostic colonoscopy to visualize the colon and take biopsies if necessary. Early detection through these non-invasive methods significantly improves survival rates for colorectal cancer.
Beyond Traditional Applications: Emerging Insights
The utility of stool samples extends beyond the well-established applications mentioned above. Ongoing research is continuously uncovering new ways in which stool analysis can inform health and disease.
- Drug Metabolism and Efficacy: The gut microbiome can significantly influence how the body metabolizes certain medications. Analyzing the microbiome’s composition might help predict an individual’s response to specific drugs or identify potential drug-drug interactions.
- Nutrient Deficiencies: While not a primary diagnostic tool for all nutrient deficiencies, certain patterns in stool can indirectly suggest issues with absorption or gut health that might contribute to deficiencies. For example, severe malabsorption can lead to deficiencies in fat-soluble vitamins.
- Personalized Nutrition: Understanding an individual’s unique gut microbiome composition can inform personalized dietary recommendations. For instance, certain microbes may be better at breaking down specific types of fiber, suggesting dietary adjustments to optimize nutrient intake and gut health.
- Monitoring Treatment Response: For various gastrointestinal conditions, including IBD, IBS, and even infections, serial stool analysis can be used to monitor the effectiveness of treatment. Changes in inflammatory markers, pathogen load, or microbiome composition can indicate whether a therapy is working or needs to be adjusted.
In conclusion, stool samples are far more than just a waste product. They are a rich source of information about our health, offering a non-invasive yet powerful lens through which to view the complex ecosystem of the gut. From diagnosing infections and identifying malabsorption to assessing the intricate balance of the microbiome and screening for colorectal cancer, stool analysis plays a vital role in modern diagnostics and the pursuit of personalized health. As our understanding of the gut microbiome continues to expand, the diagnostic and therapeutic potential of stool analysis is set to grow even further, promising new avenues for preventing, diagnosing, and managing a wide spectrum of diseases.
