The landscape of weight management and type 2 diabetes treatment has been dramatically reshaped by the advent of novel GLP-1 receptor agonists and dual GIP/GLP-1 receptor agonists. Among the most talked-about medications in this class are semaglutide (marketed as Ozempic, Wegovy, and Rybelsus) and tirzepatide (marketed as Mounjaro and Zepbound). Both have demonstrated remarkable efficacy in improving glycemic control and facilitating significant weight loss, leading many to ponder which offers superior benefits. While both drugs represent significant advancements, understanding their distinct mechanisms, clinical trial data, and potential side effects is crucial for discerning their comparative advantages.

Understanding the Mechanisms of Action
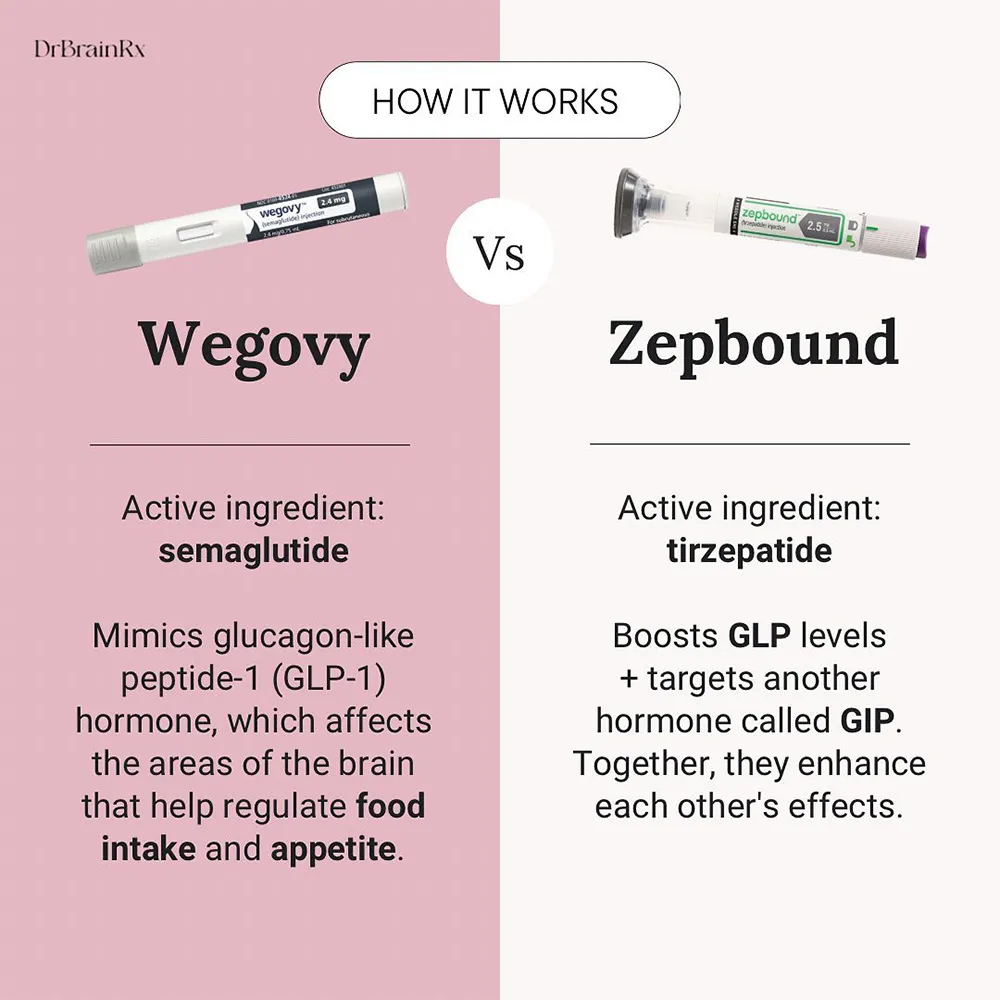
At the core of the difference between tirzepatide and semaglutide lies their unique molecular targets and mechanisms of action. This distinction is fundamental to understanding their differing levels of efficacy and therapeutic profiles.
Semaglutide: A Potent GLP-1 Receptor Agonist
Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist. GLP-1 is an incretin hormone naturally produced in the gut in response to food intake. It plays a vital role in glucose homeostasis by:
- Stimulating Insulin Secretion: GLP-1 enhances the release of insulin from pancreatic beta cells in a glucose-dependent manner, meaning it only stimulates insulin release when blood glucose levels are high, thereby reducing the risk of hypoglycemia.
- Suppressing Glucagon Release: Glucagon is a hormone that raises blood glucose levels. GLP-1 inhibits glucagon secretion, further contributing to lower blood sugar.
- Slowing Gastric Emptying: By delaying the rate at which food leaves the stomach, GLP-1 promotes a feeling of fullness and satiety, which can lead to reduced food intake and subsequent weight loss.
- Acting on the Brain: GLP-1 also affects appetite-regulating centers in the brain, reducing hunger and increasing feelings of fullness.
Semaglutide is designed to mimic the action of native GLP-1 but with a significantly longer duration of action, allowing for once-weekly subcutaneous injections (Ozempic and Wegovy) or a daily oral tablet (Rybelsus).
Tirzepatide: A Dual GIP and GLP-1 Receptor Agonist
Tirzepatide takes a more comprehensive approach by acting as a dual agonist for two distinct incretin hormones: glucose-dependent insulinotropic polypeptide (GIP) and GLP-1.
- GIP Receptor Agonism: GIP is another incretin hormone that, like GLP-1, stimulates insulin release in a glucose-dependent manner and suppresses glucagon secretion. However, GIP’s effects on gastric emptying and appetite are generally considered less pronounced than those of GLP-1. Importantly, GIP also plays a role in fat metabolism and insulin sensitivity.
- GLP-1 Receptor Agonism: Tirzepatide also activates the GLP-1 receptor, conferring the benefits associated with GLP-1 agonism, including enhanced insulin secretion, reduced glucagon, slowed gastric emptying, and appetite suppression.
By targeting both GIP and GLP-1 receptors, tirzepatide leverages the synergistic effects of these two hormones. This dual action is hypothesized to contribute to its enhanced efficacy in both glucose control and weight reduction compared to GLP-1 receptor agonists alone.
Clinical Trial Evidence: Comparing Efficacy and Outcomes
The comparative superiority of tirzepatide over semaglutide in clinical outcomes is largely supported by robust clinical trial data. Head-to-head studies and meta-analyses have provided compelling evidence for tirzepatide’s greater impact on both HbA1c reduction and weight loss.
SURPASS Trials: Demonstrating Tirzepatide’s Superiority
The pivotal Phase 3 clinical trial program for tirzepatide, known as the SURPASS trials, consistently showed significant advantages over comparators, including semaglutide in some instances.
- SURPASS-2: This landmark trial directly compared tirzepatide (at doses of 5 mg, 10 mg, and 15 mg weekly) to semaglutide 1 mg weekly in adults with type 2 diabetes. The results were striking:
- HbA1c Reduction: Tirzepatide achieved significantly greater reductions in HbA1c compared to semaglutide across all tested doses. For example, the 15 mg dose of tirzepatide resulted in an average HbA1c reduction of 2.46%, compared to 1.84% for semaglutide.
- Weight Loss: Tirzepatide also induced substantially more weight loss. The 15 mg dose of tirzepatide led to an average weight reduction of 22.5%, whereas semaglutide resulted in an average weight loss of 15.0%.
- Other SURPASS Trials: Across various comparator arms in the SURPASS program, tirzepatide consistently demonstrated superior efficacy in improving glycemic control and promoting weight loss compared to placebo and other active treatments, further solidifying its position as a highly potent agent.
Efficacy in Weight Management: Wegovy vs. Zepbound
When considering weight management specifically, the marketed formulations of semaglutide (Wegovy) and tirzepatide (Zepbound) have also been compared, albeit often through extrapolations from their respective trial data.
- SELECT Trial (Semaglutide): The SELECT trial, which investigated the cardiovascular outcomes of semaglutide in individuals with overweight or obesity and established cardiovascular disease, also provided significant data on weight loss. Participants on semaglutide 2.4 mg weekly achieved an average weight loss of approximately 15%.
- SURMOUNT-1 Trial (Tirzepatide): This trial, designed for individuals with obesity or overweight and at least one comorbidity (excluding diabetes), demonstrated even more substantial weight loss with tirzepatide. Participants on the highest dose of tirzepatide (15 mg weekly) achieved an average weight reduction of 22.5%.

These figures suggest that, on average, tirzepatide elicits a greater degree of weight loss compared to semaglutide when used at their respective maximal indicated doses for weight management.
Safety Profiles and Side Effects
While both tirzepatide and semaglutide are generally well-tolerated, they share a common class of side effects, primarily gastrointestinal in nature. Understanding these potential adverse events and their management is crucial for patient counseling and treatment decisions.
Common Gastrointestinal Adverse Events
The most frequently reported side effects for both medications include:
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Abdominal pain
- Decreased appetite
These side effects are often dose-dependent and tend to be most pronounced when initiating treatment or increasing the dosage. They are typically mild to moderate in severity and often resolve over time as the body adjusts to the medication. Gradual dose escalation, as recommended by prescribing guidelines, is a key strategy to mitigate these gastrointestinal disturbances.
Other Potential Side Effects
Beyond gastrointestinal issues, other potential side effects to be aware of for both drugs include:
- Pancreatitis: Although rare, there have been reports of acute pancreatitis in individuals taking GLP-1 receptor agonists and dual GIP/GLP-1 receptor agonists. Patients with a history of pancreatitis should be monitored closely.
- Gallbladder Disease: Rapid weight loss, a common outcome with these medications, can increase the risk of gallstones and gallbladder disease.
- Diabetic Retinopathy Complications: In patients with type 2 diabetes, rapid improvements in glycemic control may, in rare instances, lead to a transient worsening of diabetic retinopathy.
- Kidney Problems: Dehydration, often due to severe vomiting or diarrhea, can exacerbate pre-existing kidney issues.
- Hypoglycemia: While both drugs are glucose-dependent, the risk of hypoglycemia can increase when used in combination with other diabetes medications, such as sulfonylureas or insulin.
Differentiating Safety Concerns
While the overall safety profiles are similar, subtle differences in patient populations and specific trial findings might warrant consideration. For instance, the SURPASS trials, which directly compared tirzepatide and semaglutide, showed a slightly higher incidence of gastrointestinal side effects with tirzepatide, particularly at higher doses. However, the magnitude of these differences was generally modest, and the severity of these events did not typically lead to discontinuation from the trials.
Patient Selection and Future Directions
The choice between tirzepatide and semaglutide hinges on several factors, including the primary treatment goal, patient characteristics, and response to therapy. Both medications represent significant advancements, but tirzepatide’s enhanced efficacy suggests it may be the preferred agent for individuals seeking the most aggressive reduction in HbA1c and weight.
When Might Tirzepatide Be Preferred?
- Type 2 Diabetes with Significant Weight Loss Goals: For patients with type 2 diabetes who also struggle with significant overweight or obesity and desire substantial weight loss to improve metabolic control, tirzepatide’s demonstrated superior weight loss potential makes it a compelling choice.
- Suboptimal Glycemic Control on Other Therapies: In individuals with type 2 diabetes whose HbA1c remains elevated despite optimal lifestyle interventions and other diabetes medications, tirzepatide’s potent glucose-lowering effects can be particularly beneficial.
- Seeking Maximum Weight Reduction: For individuals focused primarily on achieving significant weight loss for health benefits, and who do not have contraindications, tirzepatide has shown greater average weight loss in clinical trials.
When Might Semaglutide Be a Strong Option?
- Established Treatment with Good Response: For patients already on semaglutide who are achieving their treatment goals and tolerating the medication well, there may be no need to switch.
- Preference for GLP-1 Agonism Alone: Some individuals or clinicians may prefer to target GLP-1 receptors exclusively, perhaps due to specific concerns or experiences.
- Availability and Cost Considerations: Access, insurance coverage, and cost can play a significant role in medication selection. While both are expensive, regional availability and formulary coverage can differ.
- Oral Formulation Preference: For individuals who cannot or prefer not to administer weekly injections, the oral formulation of semaglutide (Rybelsus) offers an alternative, though its efficacy is generally lower than the injectable forms.
Future Outlook
The advent of tirzepatide has undoubtedly raised the bar for efficacy in the treatment of type 2 diabetes and obesity. Ongoing research continues to explore the long-term benefits and safety profiles of both tirzepatide and semaglutide, as well as potential new agents in this therapeutic class. The development of more targeted therapies and combinations, potentially with fewer side effects or improved patient adherence, remains a key focus for pharmaceutical innovation. Ultimately, the “better” medication is the one that best meets the individual patient’s needs, considering their specific health status, treatment goals, and tolerance. A thorough discussion with a healthcare provider is essential to make an informed decision.
