The advent of drone technology has revolutionized numerous scientific and industrial fields, particularly in remote sensing and environmental monitoring. Modern unmanned aerial vehicles (UAVs) are equipped with sophisticated sensors capable of gathering vast amounts of data, from high-resolution imagery to intricate spectral analyses. Understanding fundamental atomic properties, specifically atomic mass and atomic number, is critical for interpreting the data captured by these advanced drone systems, especially when analyzing material composition from a distance. These two concepts, while related, describe distinct characteristics of an atom and play unique roles in how drones gather and process information about the world around us.

The Foundation of Elemental Identification: Atomic Number in Drone Sensing
The atomic number (Z) is arguably the most fundamental property of a chemical element. It unequivocally defines an element by representing the precise number of protons found in the nucleus of an atom. This number is unique to each element on the periodic table; for instance, all atoms of carbon have an atomic number of 6, and all atoms of oxygen have an atomic number of 8. This singular characteristic makes the atomic number the “fingerprint” for identifying an element.
Protons as the Defining Characteristic
In the context of drone-based remote sensing, the atomic number is crucial because it dictates an element’s chemical behavior and its characteristic spectral signature. When a drone carries a hyperspectral or multispectral sensor, it measures the electromagnetic radiation (light) reflected or emitted by surfaces below. Different elements, due to their unique electron configurations (which are determined by the number of protons/atomic number), interact with light in specific ways, absorbing or emitting energy at distinct wavelengths. These unique spectral patterns allow for the qualitative identification of elements and, by extension, the compounds they form.
Applications in Spectroscopic Analysis for Drones
Drone-mounted spectrometers, for example, can detect the presence of specific elements in soil, water, or vegetation. By analyzing the absorption or emission lines in the collected spectra, scientists can deduce the elemental composition of an area. For instance, detecting specific nitrogen or phosphorus signatures in crops helps assess nutrient deficiencies, while identifying heavy metal signatures in contaminated sites pinpoints pollutants. The underlying principle for these detections is the distinct atomic number of each element, which dictates how its electrons behave and thus its unique spectral response. Without knowing the atomic number, identifying these elements remotely would be impossible, severely limiting the analytical power of drone-based sensing platforms.
Unveiling Nuances: Atomic Mass and Isotopic Signatures in Aerial Data
While the atomic number identifies the element, the atomic mass provides a more detailed understanding of its physical composition and, importantly, its isotopic variations. Atomic mass is the total mass of an atom, primarily determined by the sum of its protons and neutrons in the nucleus, with a small contribution from electrons. Unlike the atomic number, which is fixed for an element, atomic mass can vary slightly among atoms of the same element due to differing numbers of neutrons. These variations are known as isotopes.

The Role of Neutrons and Electrons
The presence of varying numbers of neutrons for a given element leads to different isotopes (e.g., Carbon-12 vs. Carbon-14). While isotopes of an element have the same chemical properties (due to the same number of protons and electrons), their differing masses can be exploited in advanced remote sensing. The average atomic mass of an element, as typically listed on the periodic table, is a weighted average of the masses of all its naturally occurring isotopes. In drone sensing, subtle differences in atomic mass can indicate a range of phenomena, from the origin of materials to the processes they have undergone.
Isotope Analysis for Geological and Environmental Insights
Advanced drone sensors, particularly those employing technologies like laser-induced breakdown spectroscopy (LIBS) or gamma-ray spectroscopy, can differentiate between isotopes. This capability opens up a powerful dimension for remote sensing. For example, isotopic ratios of elements like carbon, oxygen, or nitrogen can act as powerful tracers in environmental studies. Drones equipped with such technology can map variations in these ratios across landscapes. This could reveal:
- Water sources and movement: Deuterium and Oxygen-18 isotopes in water provide clues about its origin and hydrological pathways.
- Pollution source tracking: Specific isotopic signatures of pollutants can help identify their industrial source.
- Geological mapping: Isotopic ratios in rocks and minerals aid in understanding geological formations, ore deposits, and the history of planetary surfaces (relevant for astrobiology-focused drone missions).
- Vegetation stress and carbon cycling: Changes in carbon isotopic ratios in plants can indicate drought stress or changes in photosynthetic pathways, critical for climate change research.
Therefore, while the atomic number tells us what element is present, the atomic mass, particularly through isotopic analysis, can tell us where it came from, how old it is, or what processes it has been involved in.
Practical Implications for Drone-Based Research
The distinction and interplay between atomic number and atomic mass have profound practical implications for the design, deployment, and data interpretation of drone-based remote sensing platforms. Leveraging these fundamental properties allows for more precise and informative data collection, moving beyond simple visual inspection to detailed material characterization.
Calibrating Sensors for Precise Elemental Mapping
For drones equipped with elemental analysis capabilities, precise calibration is paramount. Sensors like LIBS, which vaporize a small amount of material with a laser and analyze the emitted light, must be calibrated against known samples with precise atomic numbers and isotopic compositions. This ensures that the spectral lines detected remotely correspond accurately to specific elements and their isotopes. Understanding the atomic number helps in targeting specific elements, while knowing the atomic mass guides the fine-tuning for isotopic differentiation, allowing for highly accurate elemental and isotopic mapping across vast or inaccessible terrains.
Differentiating Materials and Contaminants from the Air
The ability to distinguish between atomic number and atomic mass allows drone operators to perform highly nuanced environmental assessments. For instance, a drone surveying an industrial site might detect the presence of a metal (identified by its atomic number). However, by analyzing its atomic mass and isotopic ratios, researchers could potentially determine if it’s naturally occurring or a byproduct of industrial contamination. This level of detail is critical for regulatory compliance, environmental remediation efforts, and resource exploration, providing data that is both qualitative (what elements are present) and quantitative/historical (where they originated, their concentration, or their age).
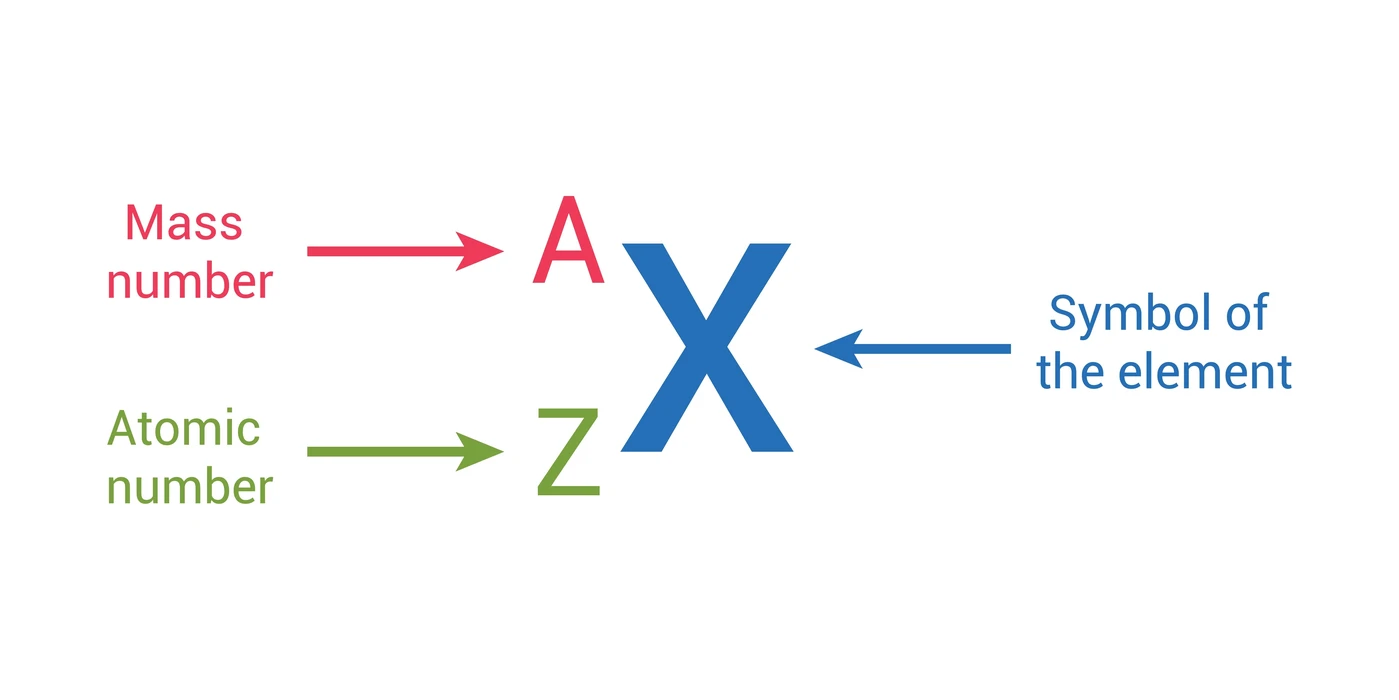
Advancing Autonomous Sensing Capabilities
As drone technology continues to evolve, the integration of AI and machine learning will further enhance the interpretation of atomic number and atomic mass data. Autonomous drones could be programmed to identify specific elemental fingerprints in real-time, adjusting their flight paths or sensor settings to gather more detailed isotopic information when anomalies are detected. This capability could lead to fully autonomous environmental monitoring systems that not only detect pollutants but also trace their origins without human intervention, or sophisticated geological surveys that map mineral deposits with unprecedented accuracy. The future of drone-based remote sensing hinges on our ability to effectively translate fundamental atomic physics into actionable insights from aerial platforms, constantly refining our understanding of the atomic numbers and masses that compose our world.
