The human heart, an organ of astonishing complexity and resilience, functions as the central pump of the circulatory system, meticulously orchestrating the flow of blood throughout the body. Its intricate design, featuring four chambers and a network of valves, ensures that deoxygenated blood is routed to the lungs for oxygenation, while freshly oxygenated blood is propelled to the systemic circulation to nourish tissues and organs. Within this marvel of biological engineering, however, anomalies can arise, disrupting the precise flow dynamics. One such significant deviation is a “shunt,” a term that, in cardiology, refers to an abnormal connection or passage that allows blood to flow directly between two parts of the heart or between major blood vessels, bypassing the normal circulatory pathway. Understanding a cardiac shunt requires delving into the sophisticated architecture and fluid mechanics of the cardiovascular system, highlighting how even minor structural irregularities can lead to profound physiological consequences. This exploration reveals the heart not just as a pump but as a finely tuned biological machine, where deviations from its optimal design demand sophisticated diagnostic and therapeutic innovations.
The Heart as a Complex System: Understanding Its Intricacies
At its core, the heart is a four-chambered muscular organ divided by a septum into right and left sides. The right side receives deoxygenated blood from the body and pumps it to the lungs (pulmonary circulation), while the left side receives oxygenated blood from the lungs and pumps it to the rest of the body (systemic circulation). This dual-pump system operates in a precise sequence, driven by electrical impulses and regulated by an array of hormones and neural signals. The atria collect blood, and the ventricles pump it out, with one-way valves ensuring unidirectional flow and preventing regurgitation. The pressure gradients within the heart chambers and major vessels are critical; the left side of the heart typically operates at significantly higher pressures than the right, a differential essential for efficient systemic perfusion. Any disruption to this delicate balance, whether structural or functional, can have cascading effects, underscoring the cardiovascular system as a prime example of biological innovation where redundancy and precise control mechanisms are paramount.
The Fundamental Role of Cardiac Circulation
Normal cardiac circulation is a marvel of efficiency. Deoxygenated blood enters the right atrium from the superior and inferior vena cava, passes through the tricuspid valve into the right ventricle, and is then pumped through the pulmonary valve into the pulmonary artery, leading to the lungs. In the lungs, carbon dioxide is exchanged for oxygen. The now oxygenated blood returns via the pulmonary veins to the left atrium, moves through the mitral valve into the left ventricle, and is finally ejected through the aortic valve into the aorta, distributing oxygen-rich blood throughout the body. This continuous, rhythmic cycle is fundamental to sustaining life, and any deviation, such as a shunt, represents a significant challenge to the system’s ability to maintain homeostasis.
Defining a Cardiac Shunt: An Anomaly in Flow Dynamics
A cardiac shunt fundamentally represents an aberrant pathway that allows blood to diverge from its intended course. This typically involves blood flowing from a high-pressure area to a lower-pressure area. The most common shunts involve abnormal openings between the heart’s chambers or between the great arteries connected to the heart. The physiological impact of a shunt largely depends on its size, location, and the direction of blood flow, which in turn is dictated by the pressure differences between the communicating areas and the relative resistances of the pulmonary and systemic circulations. Shunts are broadly categorized by the direction of blood flow relative to the systemic circulation: left-to-right, right-to-left, or bidirectional.
Left-to-Right Shunts: Overloading the Pulmonary Circuit
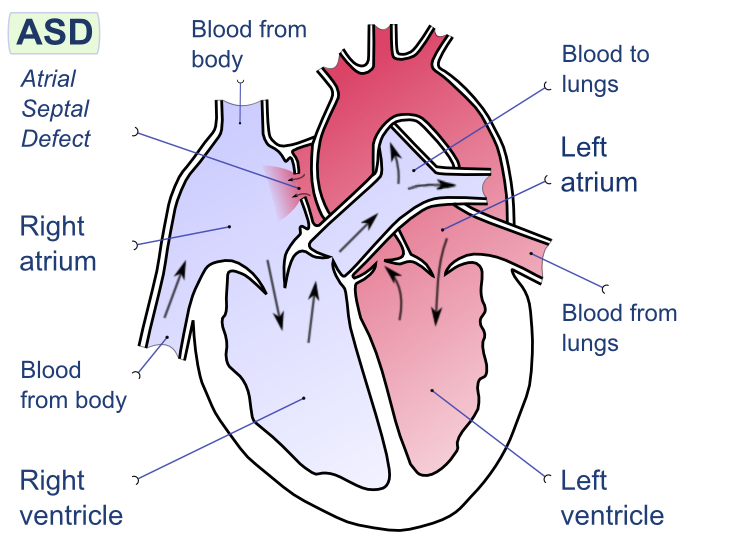
Left-to-right shunts occur when oxygenated blood from the left side of the heart (or aorta) flows into the right side of the heart (or pulmonary artery). This is the most common type of cardiac shunt. Examples include atrial septal defects (ASDs), ventricular septal defects (VSDs), and patent ductus arteriosus (PDA). Because the left side of the heart and systemic circulation operate at higher pressures than the right side and pulmonary circulation, blood naturally flows from left to right.
The primary consequence of a left-to-right shunt is an increase in blood flow to the lungs. While initially well-tolerated, a large and long-standing left-to-right shunt can lead to “pulmonary overcirculation,” causing increased pressure in the pulmonary arteries (pulmonary hypertension) and, eventually, irreversible changes in the lung blood vessels. The increased workload on the right ventricle can also lead to right heart failure. The body’s sophisticated homeostatic mechanisms attempt to compensate, but often these compensations are insufficient to prevent long-term damage without intervention.
Right-to-Left Shunts: The Challenge of Cyanosis
Right-to-left shunts occur when deoxygenated blood from the right side of the heart bypasses the lungs and flows directly into the left side of the heart or systemic circulation. This results in the systemic delivery of inadequately oxygenated blood, leading to cyanosis—a bluish discoloration of the skin and mucous membranes. Right-to-left shunts are typically more serious and often manifest earlier in life. Classic examples include Tetralogy of Fallot, transposition of the great arteries, and Eisenmenger syndrome (a late-stage complication of uncorrected large left-to-right shunts where pulmonary hypertension becomes so severe that pressures in the pulmonary circulation exceed systemic pressures, reversing the shunt direction).
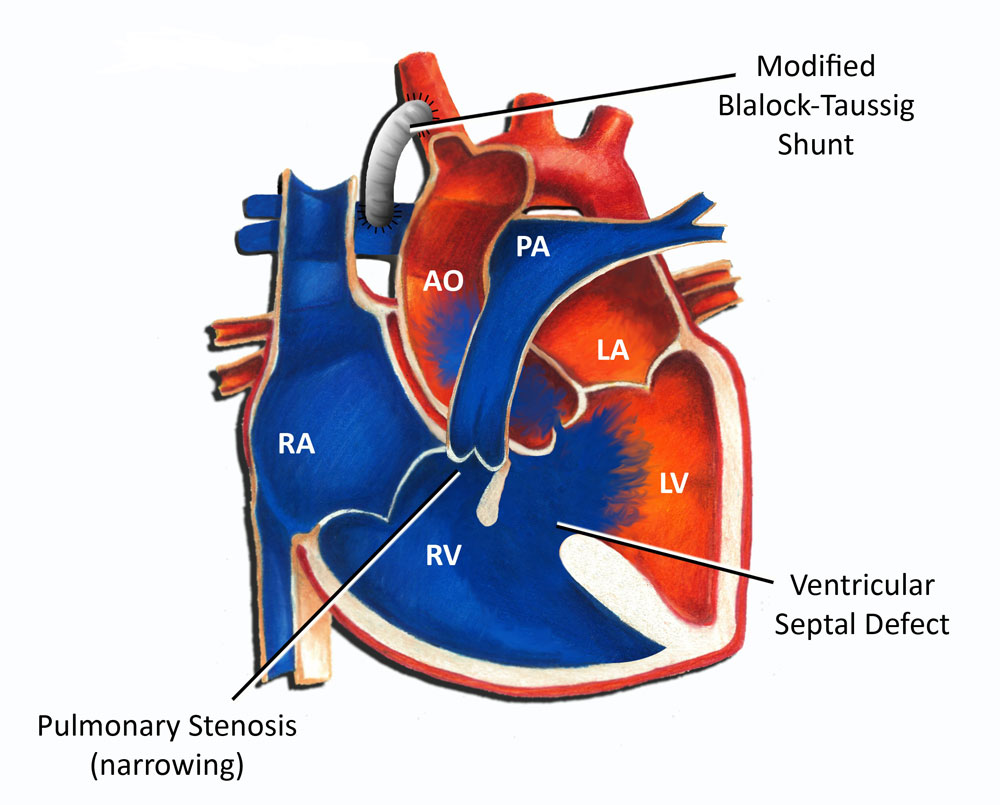
The mechanism usually involves a combination of a structural defect (e.g., a large VSD) and an obstruction to pulmonary blood flow (e.g., pulmonary stenosis). The increased resistance to flow into the lungs diverts deoxygenated blood across the defect into the systemic circulation, directly impairing tissue oxygenation. The challenge in these conditions is not merely volume overload but profound systemic hypoxia, necessitating urgent and often complex medical and surgical interventions.
Bidirectional Shunts: A Dynamic Imbalance
A bidirectional shunt occurs when blood flows in both directions across an abnormal communication, sometimes simultaneously, or with the predominant direction shifting over time. This dynamic state often arises when there is a large defect and the pressure differences between the communicating chambers or vessels are finely balanced or fluctuate, frequently due to changes in pulmonary vascular resistance. Eisenmenger syndrome often begins with a large left-to-right shunt that, over years, leads to such severe pulmonary hypertension that the shunt reverses or becomes bidirectional. Bidirectional shunts represent a critical point in disease progression, indicating significant hemodynamic instability and a complex clinical picture.
Etiology and Manifestation: When Shunts Occur
Cardiac shunts can be broadly categorized based on their origin: whether they are present at birth (congenital) or develop later in life (acquired). The understanding of their etiology is crucial for both prevention, where possible, and effective management strategies.
Congenital Shunts: Developmental Anomalies
The vast majority of cardiac shunts are congenital, meaning they are present at birth. These defects arise during fetal development of the heart and great vessels, typically within the first trimester of pregnancy. The precise causes are often multifactorial, involving a complex interplay of genetic predisposition and environmental factors. Some common congenital shunts include:
- Atrial Septal Defect (ASD): An opening in the septum between the two upper chambers (atria).
- Ventricular Septal Defect (VSD): An opening in the septum between the two lower chambers (ventricles). This is the most common congenital heart defect.
- Patent Ductus Arteriosus (PDA): A persistence of the ductus arteriosus, a normal fetal blood vessel that connects the aorta and pulmonary artery, which should close shortly after birth.
- Tetralogy of Fallot: A complex condition involving four distinct defects: a VSD, pulmonary stenosis (narrowing of the pulmonary outflow tract), an overriding aorta (aorta positioned over the VSD), and right ventricular hypertrophy.
Advances in embryology and genetics are continually shedding light on the intricate cellular and molecular pathways that govern cardiac development, offering new avenues for understanding and potentially mitigating these developmental anomalies.
Acquired Shunts: Secondary to Disease or Injury
While less common than congenital shunts, acquired shunts can develop later in life due to various medical conditions or traumatic injuries. These typically result from damage to heart tissue or vessels, disrupting normal structural integrity. Examples include:
- Myocardial Infarction (Heart Attack): A severe heart attack can lead to necrosis of heart tissue, potentially causing a rupture of the ventricular septum and creating an acquired VSD.
- Trauma: Penetrating chest injuries can directly damage the heart or great vessels, leading to fistulas or abnormal connections.
- Iatrogenic Causes: Medical procedures, though rare, can sometimes inadvertently create shunts, for instance, during catheterizations or surgeries.
- Arteriovenous Malformations (AVMs): Although not strictly cardiac, AVMs are abnormal connections between arteries and veins that can cause significant shunting of blood, increasing cardiac workload if large.
The development of acquired shunts underscores the heart’s vulnerability to external stressors and internal pathologies, highlighting the need for robust therapeutic strategies in emergency and critical care medicine.
Physiological Impact and Clinical Significance
The presence of a cardiac shunt, regardless of its type or origin, alters the normal hemodynamics of the circulatory system. The body’s physiological response to these altered flow patterns dictates the clinical manifestations and the long-term prognosis.
Hemodynamic Consequences
The primary hemodynamic consequence of a shunt is the redistribution of blood flow. In left-to-right shunts, the increased volume of blood traversing the pulmonary circulation leads to pulmonary hypertension over time. This sustained high pressure can remodel the pulmonary vasculature, increasing resistance to blood flow and eventually causing the pulmonary pressures to exceed systemic pressures, leading to a shunt reversal (Eisenmenger syndrome). This progression is a testament to the dynamic interplay between structural defects and physiological adaptations, which ultimately can become maladaptive.
In right-to-left shunts, the bypass of the pulmonary circuit results in hypoxemia, meaning insufficient oxygen in the arterial blood. Chronic hypoxemia triggers compensatory mechanisms such as polycythemia (an increase in red blood cell count) to enhance oxygen-carrying capacity. However, polycythemia can increase blood viscosity, raising the risk of thrombosis and stroke. The body’s attempt to compensate, while vital in the short term, often introduces new challenges.
Symptomatology and Disease Progression
The symptoms associated with a cardiac shunt vary widely based on the size of the shunt, its direction, the patient’s age, and the presence of any associated conditions. Small shunts may be asymptomatic and discovered incidentally. Larger shunts, however, can manifest with a range of symptoms:
- In infants and children: Poor feeding, failure to thrive, recurrent respiratory infections, rapid breathing (tachypnea), shortness of breath (dyspnea), and cyanosis (bluish skin, especially in right-to-left shunts).
- In adults: Exercise intolerance, fatigue, dyspnea on exertion, palpitations, and symptoms of heart failure (swelling in the legs, fluid retention).
The progression of shunt-related heart disease highlights the critical importance of early diagnosis and intervention. Without appropriate management, severe shunts can lead to irreversible pulmonary vascular disease, right heart failure, arrhythmias, and other life-threatening complications, underscoring the urgent need for innovative diagnostic and therapeutic approaches.

The Role of Advanced Diagnostics and Management
Diagnosing a cardiac shunt typically involves a combination of clinical evaluation, non-invasive imaging, and sometimes invasive procedures. A thorough physical examination may reveal characteristic heart murmurs or signs of heart failure or cyanosis. Electrocardiograms (ECGs) can detect signs of chamber enlargement or strain. Chest X-rays can show cardiomegaly (enlarged heart) or increased pulmonary vascular markings.
However, definitive diagnosis and detailed anatomical and physiological assessment primarily rely on advanced imaging technologies. Echocardiography (ultrasound of the heart) is the cornerstone, providing real-time images of the heart’s structure, blood flow patterns, and chamber pressures. More advanced techniques like cardiac MRI and CT scans offer high-resolution anatomical details, particularly useful for complex defects or pre-surgical planning. Cardiac catheterization, an invasive procedure, allows for precise measurement of pressures and oxygen saturations within heart chambers and vessels, and can even be therapeutic for closing certain shunts.
Management strategies range from watchful waiting for small, asymptomatic shunts to medical management of symptoms (e.g., diuretics for heart failure, oxygen therapy for hypoxemia) and, crucially, interventional closure. Surgical repair or transcatheter device closure (using catheters to deliver a device to close the defect) are common approaches for significant shunts. These interventions are a testament to the continuous innovation in medical technology, allowing for the correction of complex cardiac anomalies and vastly improving patient outcomes. The ongoing advancements in imaging, interventional cardiology, and surgical techniques exemplify how technological innovation continues to revolutionize the understanding and treatment of intricate biological systems like the human heart.
