The Carnot cycle, a theoretical thermodynamic concept, serves as a foundational principle in understanding the efficiency limits of heat engines. While not directly applicable to the mechanics of drone flight or onboard camera systems, its principles offer valuable insights into the broader landscape of energy conversion and efficiency that underpins many technological advancements, including those found in sophisticated unmanned aerial vehicles (UAVs). Understanding the Carnot cycle is akin to grasping the fundamental laws of thermodynamics that govern how energy is transformed, a critical consideration in the development of power-efficient and high-performance technologies.
The cycle was first described by French physicist Sadi Carnot in 1824, and it represents the most efficient possible thermodynamic cycle operating between two given temperature reservoirs. It is an idealized, reversible cycle consisting of four distinct thermodynamic processes: two isothermal processes and two adiabatic processes. Its significance lies not in its practical implementability – as perfect reversibility and infinite heat reservoirs are unattainable in reality – but in establishing an upper bound for the efficiency of any heat engine operating between those same two temperatures. This theoretical ceiling is crucial for engineers and scientists aiming to optimize energy conversion in various applications.

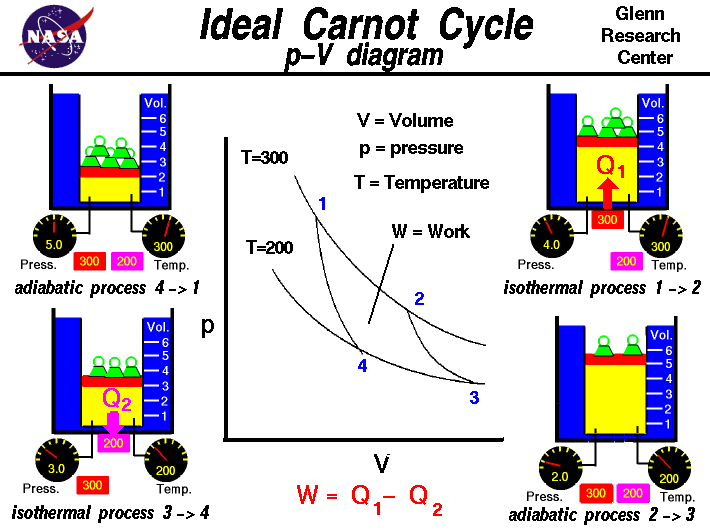
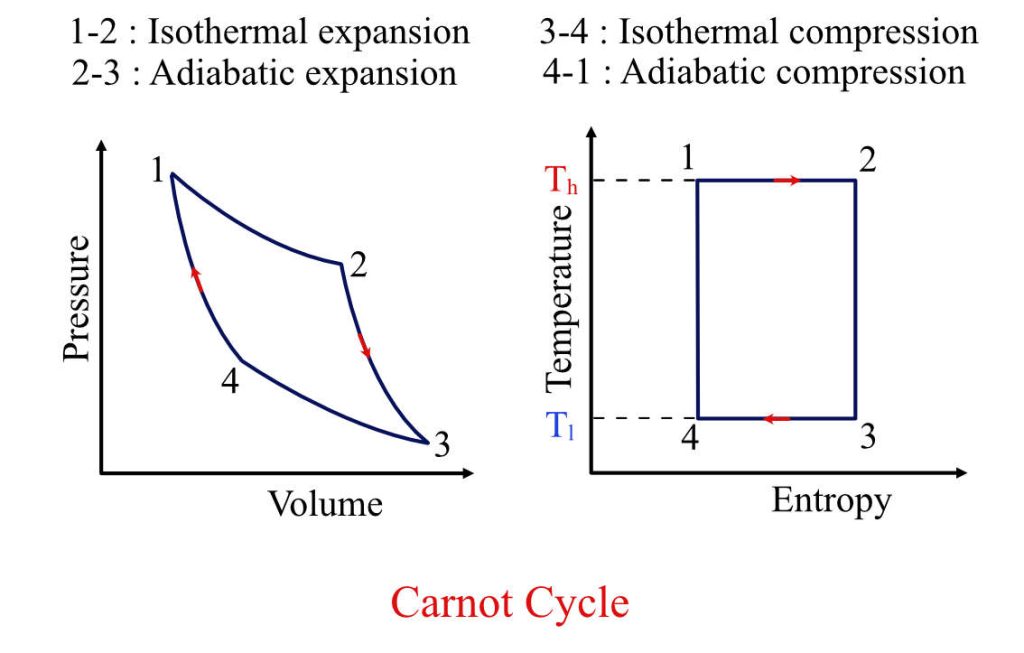
The Four Stages of the Carnot Cycle
The Carnot cycle’s elegance lies in its simplicity and the clear delineation of its four reversible processes. Each stage plays a specific role in the overall transformation of heat energy into mechanical work.
Isothermal Expansion
The cycle begins with an isothermal expansion of an ideal working substance, typically a gas. At the start of this phase, the working substance is in thermal contact with a high-temperature heat reservoir (Thot). Heat (Qhot) is absorbed by the working substance from this reservoir, causing it to expand. Crucially, this expansion occurs at a constant temperature, meaning the internal energy of the gas remains constant. According to the first law of thermodynamics ($Delta U = Q – W$), since the internal energy change is zero, the work done by the gas ($W$) is equal to the heat absorbed ($Q_{hot}$). This absorbed heat is what drives the mechanical work performed by the engine. In the context of theoretical efficiency, this process highlights the direct conversion of heat into work under ideal conditions.
Adiabatic Expansion
Following the isothermal expansion, the working substance undergoes an adiabatic expansion. In this phase, the working substance is thermally isolated from its surroundings, meaning no heat is exchanged with either the hot or cold reservoir ($Q = 0$). As the gas continues to expand, it does work on its surroundings. Since no heat is entering or leaving the system, the expansion causes the internal energy of the gas to decrease, leading to a drop in its temperature. This adiabatic process is vital for lowering the temperature of the working substance, preparing it for the next stage of the cycle where heat will be rejected. The work done during this adiabatic expansion further contributes to the net mechanical output of the engine.
Isothermal Compression
The third stage of the Carnot cycle is an isothermal compression. Here, the working substance is brought into thermal contact with a low-temperature heat reservoir (Tcold). Heat ($Q{cold}$) is rejected from the working substance to this reservoir as it is compressed. Similar to the isothermal expansion, this compression occurs at a constant temperature. The work done on the gas during compression is equal to the heat rejected to the cold reservoir. This process effectively removes heat from the working substance, bringing it back to its initial state of temperature and pressure before the next phase.
Adiabatic Compression
The final stage involves an adiabatic compression. Again, the working substance is thermally isolated, and no heat is exchanged. Work is done on the gas to compress it. This compression increases the internal energy of the gas, causing its temperature to rise back to its initial high temperature. By the end of this adiabatic compression, the working substance has returned to its original state, ready to begin the cycle anew. The work done during this compression is essential for completing the cycle and ensuring that the net work output over the entire cycle is positive.
The Carnot Efficiency Formula
The efficiency of any heat engine is defined as the ratio of the net work output to the heat input. For the Carnot cycle, this efficiency ($eta_{Carnot}$) is remarkably simple and depends solely on the temperatures of the hot and cold reservoirs.
$eta{Carnot} = frac{W{net}}{Q{hot}} = 1 – frac{T{cold}}{T_{hot}}$
Where:
- $W_{net}$ is the net work done by the engine in one cycle.
- $Q_{hot}$ is the heat absorbed from the hot reservoir.
- $T_{cold}$ is the absolute temperature of the cold reservoir (in Kelvin).
- $T_{hot}$ is the absolute temperature of the hot reservoir (in Kelvin).
This formula highlights a fundamental principle: the greater the temperature difference between the hot and cold reservoirs, the higher the theoretical maximum efficiency of the heat engine. Conversely, if $T{cold}$ approaches $T{hot}$, the efficiency approaches zero. If $T_{cold}$ is absolute zero (0 Kelvin), the efficiency approaches 100%. However, achieving absolute zero is physically impossible, thus a 100% efficient heat engine is unobtainable.
The net work ($W{net}$) in the Carnot cycle is the difference between the work done during the isothermal expansion and the work done on the gas during the isothermal compression: $W{net} = W{expansion} – W{compression}$. Since $Q{hot} = W{expansion}$ and $Q{cold} = W{compression}$ (in magnitude), the net work can also be expressed as $W{net} = Q{hot} – Q_{cold}$. Substituting this into the efficiency formula gives:
$eta{Carnot} = frac{Q{hot} – Q{cold}}{Q{hot}} = 1 – frac{Q{cold}}{Q{hot}}$
And since for isothermal processes of an ideal gas, $Q/T$ is constant, we have $Q{cold}/T{cold} = Q{hot}/T{hot}$, which leads back to the temperature-dependent efficiency formula.
Implications for Technology and Innovation
While the Carnot cycle itself is an idealized construct, its implications for technological advancement are profound and far-reaching. It establishes a benchmark against which the performance of all real-world heat engines can be compared. Engineers striving to design more efficient engines, whether for power generation, transportation, or even the power systems of advanced drones, must always acknowledge that their designs cannot surpass the Carnot efficiency limit for the operating temperatures involved.
Understanding Efficiency Limits
The Carnot cycle provides a theoretical framework for understanding why certain energy conversion processes are inherently limited. This understanding is crucial in fields that rely heavily on efficient energy utilization. For instance, in the development of advanced battery technologies for drones, understanding energy storage and discharge efficiencies, which are analogous to heat engine efficiencies in terms of energy conversion, is paramount. While not a direct application of heat engines, the thermodynamic principles of energy loss and conversion efficiency are universally applicable.
Driving Research and Development
The pursuit of efficiencies closer to the Carnot limit fuels innovation. Engineers are constantly exploring new working substances, novel engine designs, and advanced materials to minimize irreversible losses such as friction, heat leaks, and incomplete combustion that plague real-world engines. This relentless drive to improve efficiency leads to breakthroughs in areas like lightweight materials, advanced cooling systems, and more sophisticated control algorithms – all of which can find their way into the design and operation of cutting-edge technological systems, including those in the aviation and imaging sectors.
The Pursuit of Higher Temperatures
The Carnot efficiency formula reveals that increasing $T{hot}$ or decreasing $T{cold}$ can significantly boost efficiency. This principle has driven research into high-temperature materials capable of withstanding extreme thermal conditions, such as ceramics and superalloys. In applications where heat is generated as a byproduct, like within sophisticated onboard processing units of drones or the power systems of larger UAVs, the ability to effectively utilize or dissipate this heat while maintaining high efficiency becomes a critical design challenge.
Thermodynamics in Modern Systems
Even in electronic systems, where heat is often an unwelcome byproduct rather than a direct energy source for work, thermodynamic principles are vital. Efficient thermal management is essential for preventing overheating and ensuring the longevity and performance of sensitive components, including the cameras and sensors that are integral to many drone applications. Understanding how heat flows and dissipates, governed by thermodynamic laws, is directly related to the principles that underpin the Carnot cycle.
While the direct application of the Carnot cycle to the operational mechanics of drones, their cameras, or flight technology might be abstract, its role as a fundamental law of physics governing energy conversion provides an indispensable lens through which to view the constant quest for greater efficiency and performance in all technological domains. It stands as a testament to the power of theoretical physics in guiding practical engineering endeavors, pushing the boundaries of what is possible and inspiring future innovations.
