The realm of chromatography, a powerful set of analytical techniques used to separate and analyze complex mixtures, hinges on a fundamental concept: the mobile phase. This often-overlooked component is the driving force behind the separation process, carrying the sample components through the stationary phase. Understanding its nature, properties, and selection is paramount to achieving accurate and meaningful analytical results. In essence, the mobile phase acts as the “runner” in a race, escorting different “runners” (analytes) at varying speeds based on their interactions with the “track” (stationary phase). Its choice profoundly impacts resolution, sensitivity, and the overall efficiency of the chromatographic separation.
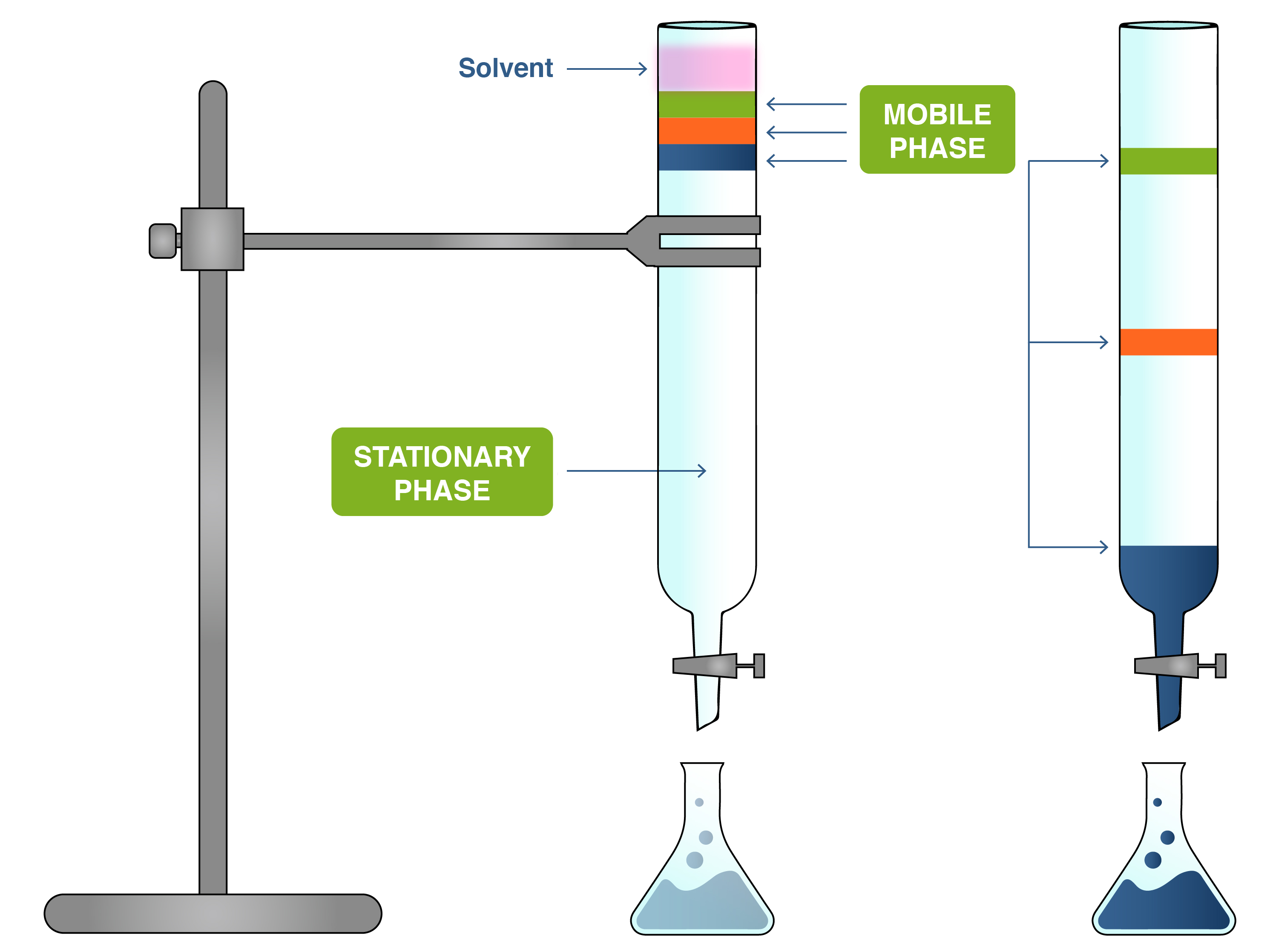
The Role of the Mobile Phase in Separation
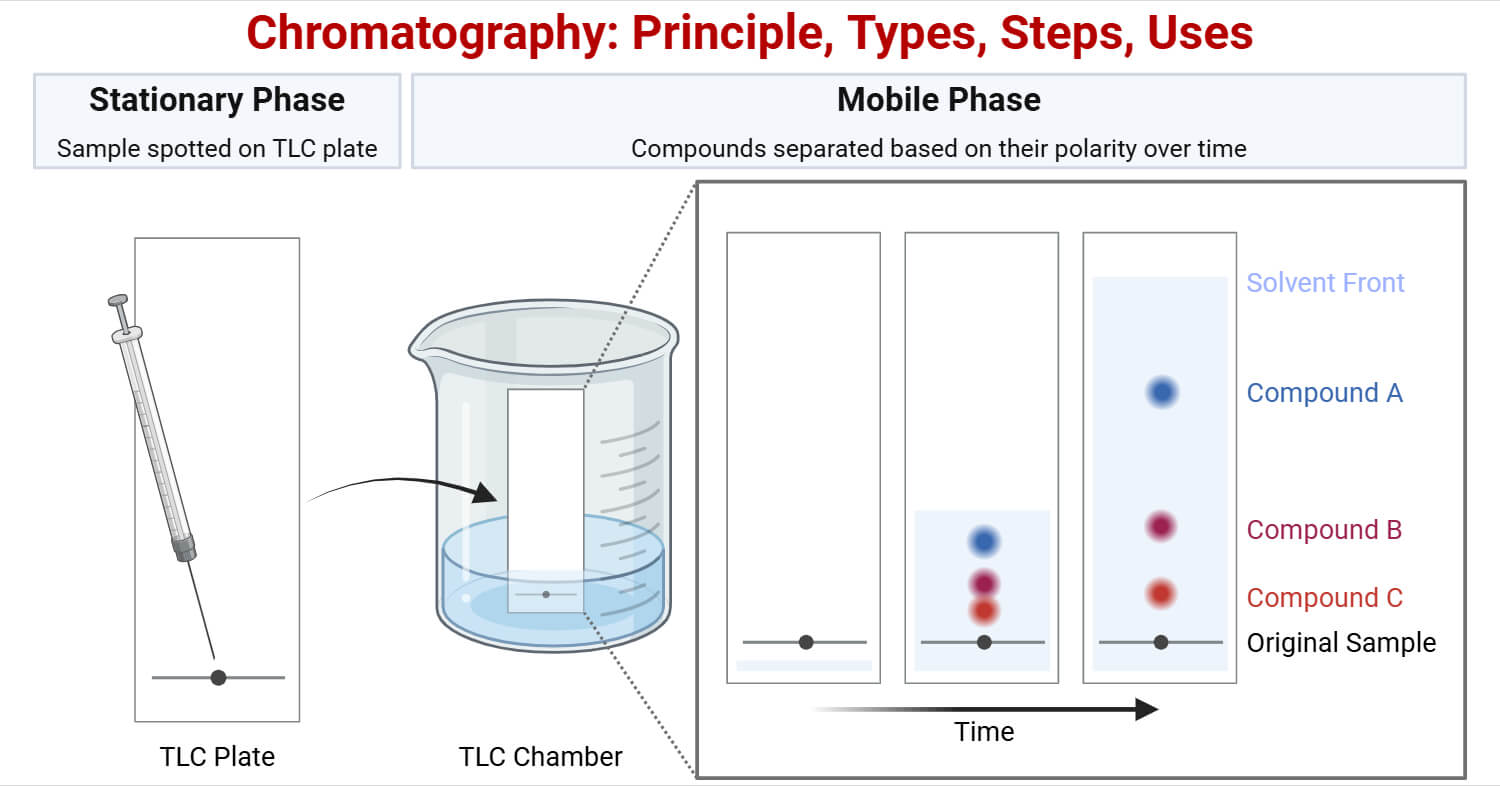
At its core, chromatography operates on the principle of differential partitioning. When a sample is introduced into a chromatographic system, it interacts with both the mobile phase and the stationary phase. The stationary phase is a solid or a liquid coated on a solid support, packed within a column or spread as a thin layer. The mobile phase, which can be a liquid or a gas, flows continuously over or through the stationary phase.
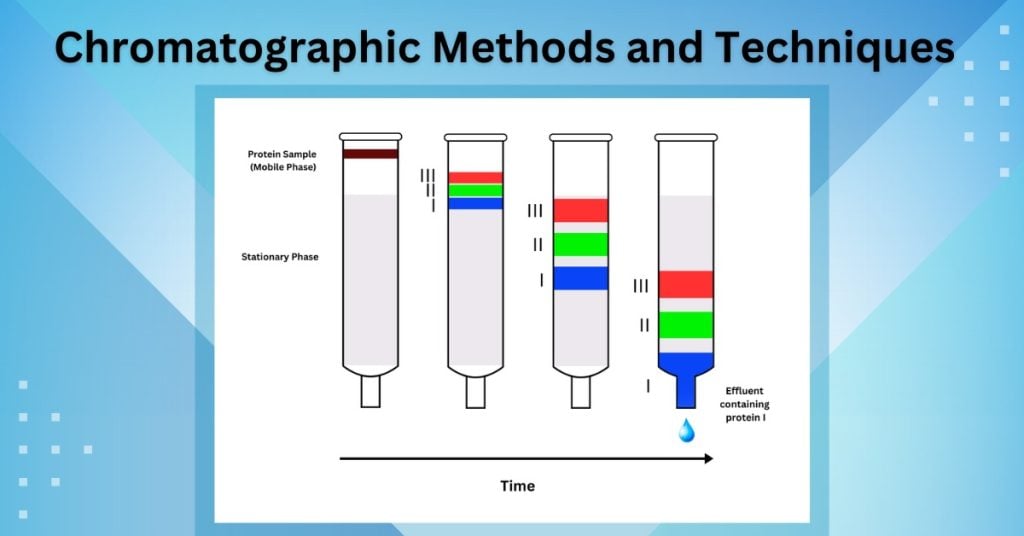
The separation of components within the sample is driven by the varying affinities each component has for the mobile and stationary phases. Components that have a higher affinity for the stationary phase will spend more time interacting with it, effectively slowing their progress through the system. Conversely, components with a greater affinity for the mobile phase will be swept along more rapidly. This difference in migration rates, or retention times, is what allows for the separation and subsequent identification or quantification of individual analytes.
Mechanism of Separation
The mobile phase’s primary function is to transport the sample mixture. However, its chemical and physical properties also directly influence the separation mechanism. In liquid chromatography (LC), the mobile phase is typically a liquid solvent or a mixture of solvents. The polarity, viscosity, and pH of the mobile phase play critical roles. For instance, in reversed-phase liquid chromatography (RPLC), a common technique, the stationary phase is non-polar, and the mobile phase is polar. Analytes that are more polar will elute faster because they are more soluble in the polar mobile phase and interact less with the non-polar stationary phase. Conversely, non-polar analytes will be retained longer, as they interact more strongly with the stationary phase.
In gas chromatography (GC), the mobile phase, known as the carrier gas, is an inert gas such as helium, nitrogen, or hydrogen. Unlike in LC, the carrier gas in GC generally does not interact chemically with the analytes. Its role is solely to transport the vaporized sample components through the column. The separation in GC is primarily governed by the differences in the volatility of the analytes and their interactions with the stationary phase. However, the flow rate of the carrier gas is a crucial parameter that affects the efficiency of the separation.
Elution and Retention
The process by which analytes emerge from the chromatographic column is called elution. The mobile phase is responsible for this elution. As the mobile phase flows, it continuously carries the analytes along. The time it takes for an analyte to travel from the point of injection to the detector is known as its retention time. A shorter retention time indicates weaker interaction with the stationary phase and/or stronger affinity for the mobile phase, allowing it to elute faster. Conversely, a longer retention time signifies stronger interaction with the stationary phase and/or weaker affinity for the mobile phase, leading to slower elution.
The retention factor (k’), a more normalized measure of retention, is defined as the ratio of the time an analyte spends in the mobile phase to the time it spends in the stationary phase. A higher retention factor indicates stronger retention. The mobile phase’s composition directly influences the retention factor. By adjusting the mobile phase, chromatographers can manipulate the retention of analytes, optimizing their separation.
Properties and Selection of the Mobile Phase
The choice of mobile phase is one of the most critical decisions in developing a chromatographic method. It is not a one-size-fits-all scenario; the optimal mobile phase is highly dependent on the specific analytes being separated, the type of stationary phase, and the desired outcome. Several key properties are considered:
Polarity
Polarity is a fundamental property that dictates the interaction between the mobile phase, stationary phase, and analytes. In reversed-phase LC, where the stationary phase is non-polar, a polar mobile phase (e.g., water, methanol, acetonitrile) is used. Increasing the proportion of the organic solvent (less polar) in the mobile phase decreases the overall polarity, leading to increased retention of non-polar analytes. Conversely, increasing the proportion of water (more polar) decreases retention of non-polar analytes.
In normal-phase LC, the stationary phase is polar (e.g., silica), and a non-polar mobile phase (e.g., hexane, heptane) is employed. Here, increasing the polarity of the mobile phase (e.g., by adding a small percentage of isopropanol or ethyl acetate) will lead to faster elution of polar analytes.
Solvent Strength
Solvent strength refers to the ability of a mobile phase to elute compounds from the stationary phase. A stronger solvent will elute compounds more quickly. In RPLC, water is considered a weak solvent for non-polar compounds, while organic solvents like acetonitrile are strong solvents. In normal-phase LC, hexane is a weak solvent, and more polar solvents are stronger.
Viscosity
The viscosity of the mobile phase affects the backpressure generated in the column and the efficiency of the separation. Higher viscosity leads to higher backpressure, which can limit the flow rate and potentially reduce separation efficiency. Highly viscous mobile phases can also lead to longer run times. Therefore, mobile phases with moderate viscosity are generally preferred.
pH and Buffers
In LC, particularly when separating ionizable compounds (acids and bases), the pH of the mobile phase is a critical factor. The pH determines the ionization state of the analyte, which significantly impacts its interaction with both the stationary phase and the mobile phase. For example, in reversed-phase chromatography, weakly acidic analytes will be less retained at a low pH (where they are protonated and less polar) and more retained at a high pH (where they are deprotonated and more polar).
Buffers are often used in the mobile phase to maintain a stable pH, especially when dealing with aqueous mobile phases. A stable pH is essential for reproducible retention times and accurate quantification. The choice of buffer system depends on the desired pH range and the compatibility with the detector system.
Purity and Degassing
The purity of mobile phase solvents is crucial. Impurities can lead to extraneous peaks in the chromatogram, interfere with analyte detection, and degrade the stationary phase over time. Solvents should be of high purity, often designated as “HPLC grade” or “GC grade.”
Furthermore, mobile phases, especially those used in LC, should be degassed. Dissolved gases (like oxygen and carbon dioxide) can form bubbles in the system, leading to erratic baseline, fluctuating detector signals, and inconsistent flow rates. Degassing can be achieved through sonication, vacuum filtration, or sparging with an inert gas.
Types of Mobile Phases in Common Chromatographic Techniques
The mobile phase is intrinsically linked to the specific chromatographic technique being employed. Each technique has its preferred mobile phase characteristics and types.
Liquid Chromatography (LC) Mobile Phases
As discussed, LC mobile phases are liquids. The most common types include:
- Water: A highly polar solvent, often used as the weak component in RPLC.
- Methanol: A polar organic solvent, commonly used in RPLC, offering a good balance of elution strength and low UV cutoff.
- Acetonitrile: Another polar organic solvent, widely used in RPLC. It generally provides better peak shapes and lower viscosity than methanol, but has a higher UV cutoff.
- Isopropanol: A more polar organic solvent than methanol or acetonitrile, often used to increase elution strength in RPLC or as a mobile phase component in normal-phase LC.
- Hexane/Heptane: Non-polar solvents used as the weak mobile phase component in normal-phase LC.
- Dichloromethane/Chloroform: Moderately polar organic solvents used in normal-phase LC to increase elution strength.
- Ethyl Acetate: A moderately polar solvent used in normal-phase LC.
Often, mobile phases in LC are mixtures of these solvents to fine-tune polarity and solvent strength. Gradient elution, where the mobile phase composition is changed over time, is frequently employed to separate mixtures with a wide range of analyte polarities.
Gas Chromatography (GC) Carrier Gases
In GC, the mobile phase is an inert gas, also known as the carrier gas. The key requirement is that it does not react with the sample or the stationary phase.
- Helium (He): The most widely used carrier gas in GC. It is inert, readily available, and provides good separation efficiency over a wide range of flow rates. However, it is non-renewable and can be expensive.
- Nitrogen (N₂): Another common inert carrier gas. It is cheaper and more abundant than helium. However, it generally provides lower separation efficiency compared to helium at optimal flow rates.
- Hydrogen (H₂): Offers excellent separation efficiency and can lead to faster analyses. It is also cost-effective. However, it is flammable, posing a safety hazard, and can potentially react with certain stationary phases or analytes under specific conditions.
The choice of carrier gas in GC is often dictated by the detector being used and the desired speed and efficiency of the separation.
Optimizing Mobile Phase for Enhanced Separation
The development of a robust and effective chromatographic method often involves significant optimization of the mobile phase. This process aims to achieve optimal resolution between closely eluting peaks, improve peak shape, reduce run times, and enhance sensitivity.
Method Development Strategies
Several strategies are employed during method development to find the ideal mobile phase:
- Screening of Solvents: Initially, different solvents or solvent mixtures are tested to observe their impact on analyte retention and separation.
- Gradient Optimization: For complex mixtures, gradient elution is often necessary. This involves systematically varying the slope and starting/ending compositions of the gradient to achieve the best separation.
- pH and Buffer Optimization: When separating ionizable compounds, the pH of the mobile phase is carefully adjusted, and different buffer systems are evaluated to find the optimal pH for resolution.
- Temperature Control: While not directly a mobile phase component, the temperature of the column and the mobile phase can significantly influence separation and is often optimized alongside mobile phase composition.
- Flow Rate Adjustment: In GC, optimizing the carrier gas flow rate is crucial for achieving the Van Deemter curve optimum, which represents the most efficient separation at a given flow rate. In LC, flow rate affects backpressure and analysis time.
The Impact of Mobile Phase on Data Quality
The mobile phase directly influences the quality of the chromatographic data. An improperly chosen or prepared mobile phase can lead to:
- Poor Resolution: Peaks overlap, making it impossible to distinguish between different analytes.
- Peak Tailing or Fronting: Asymmetrical peaks, which can distort peak areas and affect quantification.
- Baseline Noise and Drift: Irregular detector signals, hindering accurate baseline determination and peak integration.
- Inconsistent Retention Times: Poor reproducibility, making it difficult to identify analytes.
- Column Degradation: Use of inappropriate solvents or impure mobile phases can shorten the lifespan of the chromatographic column.
Therefore, meticulous attention to the selection, preparation, and maintenance of the mobile phase is fundamental to obtaining reliable and accurate chromatographic results, forming the bedrock of analytical chemistry and related scientific disciplines.
