Aryl groups are fundamental building blocks in organic chemistry, playing a pivotal role in the structure and function of countless molecules, from pharmaceuticals and agrochemicals to polymers and advanced materials. Understanding aryl groups is essential for anyone delving into the complexities of organic synthesis, reaction mechanisms, and molecular design. At their core, aryl groups are derived from aromatic hydrocarbons, characterized by their unique cyclic, planar structures and delocalized pi electron systems.
The Essence of Aromaticity and Aryl Groups
The defining characteristic of an aryl group is its origin from an aromatic ring system. The most common and archetypal aromatic hydrocarbon is benzene ($text{C}6text{H}6$). Benzene’s structure is a six-membered ring of carbon atoms, each bonded to one hydrogen atom. However, the simple representation of alternating single and double bonds in benzene is an oversimplification. In reality, the pi electrons in benzene are delocalized, meaning they are spread evenly across the entire ring. This delocalization imparts significant stability to the molecule, a phenomenon known as aromaticity.
Aromatic Hydrocarbons: The Parent Compounds
Aromatic hydrocarbons, often called arenes, are organic compounds that contain one or more benzene rings. The simplest arenes are single-ring systems, such as benzene itself, toluene (methylbenzene), and xylene (dimethylbenzene). More complex arenes feature fused ring systems, like naphthalene (two fused benzene rings) and anthracene (three linearly fused benzene rings). These structures, with their characteristic electron delocalization, are the precursors from which aryl groups are derived.
Derivation of Aryl Groups: Removing a Hydrogen Atom
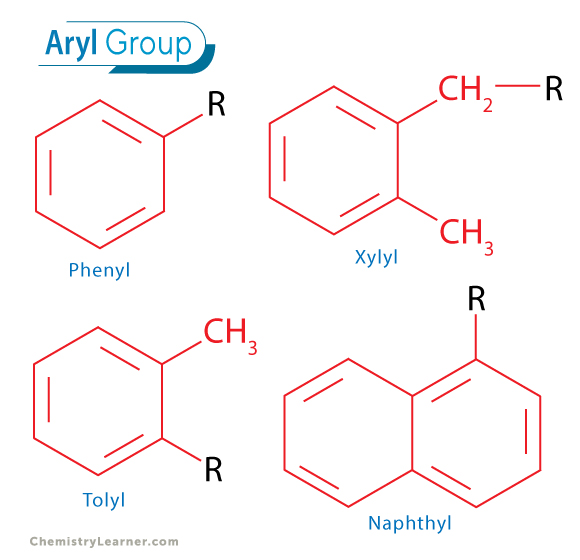
An aryl group is formed when a hydrogen atom is removed from an aromatic hydrocarbon. This creates a reactive site on the aromatic ring, allowing it to bond with other atoms or molecular fragments. For instance, when a hydrogen atom is removed from benzene, the resulting group is called a phenyl group. The phenyl group has the chemical formula $-text{C}6text{H}5$. It’s crucial to note that the remaining five hydrogen atoms on the phenyl group are still attached to the carbon atoms of the ring, and the aromaticity of the ring is preserved.
Nomenclature and Common Aryl Groups
The nomenclature of aryl groups follows systematic rules. The phenyl group, derived from benzene, is the most ubiquitous. Other common aryl groups include:
- Toluyl groups: Derived from toluene ($text{C}6text{H}5text{CH}3$). Removing a hydrogen from the methyl group yields the benzyl group ($-text{CH}2text{C}6text{H}5$), while removing a hydrogen from the aromatic ring produces toluyl groups (e.g., o-toluyl, m-toluyl, p-toluyl, depending on the position of the methyl group relative to the point of attachment).
- Naphthyl groups: Derived from naphthalene. There are two possible points of attachment on the naphthalene ring system, leading to $alpha$-naphthyl and $beta$-naphthyl groups.
- Anisyl group: Derived from anisole (methoxybenzene).
- Xylyl groups: Derived from xylene, with various isomers ($o$-, $m$-, $p$-).
The suffix “-yl” typically denotes a radical or a univalent radical, indicating that the group is capable of forming a bond.
The Significance of Aryl Groups in Chemistry
The unique electronic properties conferred by the aromatic ring make aryl groups exceptionally important in organic chemistry. Their influence extends to reactivity, stability, and the physical properties of molecules.
Stability and Resonance
The delocalized pi electron system in aromatic rings results in a lower energy state compared to analogous non-aromatic systems. This enhanced stability is a key feature of aryl groups. This stability means that aryl groups tend to retain their aromatic character even when incorporated into larger molecules, influencing the overall electronic distribution and reactivity of the compound. The phenomenon of resonance, where electron density is shared across multiple atoms, is the underlying principle behind this enhanced stability.
Reactivity: Electrophilic Aromatic Substitution
Aryl groups are known for their characteristic reactivity, particularly through a class of reactions called electrophilic aromatic substitution (EAS). In EAS, an electrophile (an electron-seeking species) replaces a hydrogen atom on the aromatic ring. Despite the inherent stability of the aromatic ring, the pi electrons can be attacked by strong electrophiles. Common examples of EAS include nitration, halogenation, sulfonation, Friedel-Crafts alkylation, and Friedel-Crafts acylation. The presence of substituents on the aromatic ring can significantly influence the rate and regioselectivity of these reactions, directing the incoming electrophile to specific positions on the ring.
Electronic Effects of Aryl Groups

Aryl groups can act as both electron-donating and electron-withdrawing groups, depending on the nature of substituents attached to them and their position relative to the point of attachment.
- Electron-donating groups (EDGs) on the aromatic ring, such as alkyl or alkoxy groups, push electron density into the ring, activating it towards electrophilic attack and directing substituents to ortho and para positions.
- Electron-withdrawing groups (EWGs), such as nitro or carbonyl groups, pull electron density out of the ring, deactivating it towards electrophilic attack and directing substituents to the meta position.
When an aryl group itself is attached to another functional group, it can exert inductive effects (electron withdrawal through sigma bonds) and resonance effects (electron donation or withdrawal through pi systems). The phenyl group, for instance, is generally considered to be weakly activating or deactivating depending on the specific reaction conditions and the nature of the group it’s attached to, often through a combination of inductive and resonance effects.
Applications and Importance Across Disciplines
The pervasive presence and unique properties of aryl groups make them indispensable in a wide array of scientific and industrial applications.
Pharmaceuticals and Medicinal Chemistry
Aryl groups are ubiquitous in pharmaceutical compounds. Their planar structure and ability to engage in pi-pi stacking interactions are crucial for drug-receptor binding. Many active pharmaceutical ingredients (APIs) contain phenyl or other aryl moieties. For example, aspirin contains a phenyl group, as do many anti-inflammatory drugs, analgesics, and antibiotics. The lipophilicity conferred by aryl groups can also influence a drug’s absorption, distribution, metabolism, and excretion (ADME) properties.
Agrochemicals
Similar to pharmaceuticals, aryl groups are prevalent in herbicides, insecticides, and fungicides. They contribute to the efficacy and stability of these agricultural chemicals. For instance, many triazine herbicides and pyrethroid insecticides incorporate aromatic rings.
Polymers and Materials Science
Aryl groups are essential components in the synthesis of many high-performance polymers. The rigid, planar nature of aromatic rings imparts thermal stability and mechanical strength to polymer chains. Examples include polystyrene, polyethylene terephthalate (PET), and polycarbonates, all of which contain aryl units. These materials find applications in packaging, textiles, electronics, and structural components.
Dyes and Pigments
The extended pi electron systems in aromatic compounds are responsible for their ability to absorb and emit light in the visible spectrum. Many dyes and pigments are based on conjugated aromatic systems, often featuring multiple fused or connected aryl rings. The phenyl group is a common structural element in many synthetic dyes.
Organic Electronics and Advanced Materials
In the realm of advanced materials, aryl groups are fundamental to organic semiconductors, organic light-emitting diodes (OLEDs), and solar cells. The delocalized electrons in conjugated aryl systems allow for efficient charge transport and light emission or absorption. Molecules like poly(p-phenylene vinylene) (PPV) are prime examples where aryl units are crucial for electronic properties.
Fragrances and Flavors
Many natural and synthetic flavor and fragrance compounds contain aryl groups. Their aromatic nature contributes to their characteristic scents and tastes. For example, vanillin, the primary component of vanilla flavor, contains a phenyl ring.
Conclusion
In essence, aryl groups represent a cornerstone of organic chemistry. Their derivation from stable aromatic hydrocarbons, characterized by delocalized pi electron systems, endows them with unique electronic properties and reactivity. From their fundamental role in electrophilic aromatic substitution to their critical contributions to the stability, functionality, and application of molecules across pharmaceuticals, materials science, and beyond, aryl groups are integral to the molecular world. A thorough understanding of their structure, properties, and behavior is indispensable for advancing chemical synthesis, molecular design, and the development of innovative technologies.
