The term “CPAM” might sound alarming, especially when associated with infants. However, understanding this medical condition is crucial for parents and caregivers. CPAM, an acronym for Congenital Pulmonary Airway Malformation, is a congenital lung abnormality that affects the development of the fetal lungs. It’s a relatively rare condition, occurring in approximately 1 in 8,000 to 1 in 30,000 live births. While the name itself can be daunting, modern medical advancements allow for accurate diagnosis, monitoring, and effective management of CPAM, ensuring the best possible outcomes for affected newborns.
Understanding Congenital Pulmonary Airway Malformation (CPAM)
CPAM is a spectrum of lesions within the lung that arise during fetal development. These malformations are characterized by the abnormal formation of the airways and lung tissue. The key feature of CPAM is the presence of cysts or abnormal air-filled spaces within the lung. These cysts can vary in size and number, and their location can also differ, affecting different parts of the lung lobe. Historically, CPAM was also referred to as congenital cystic adenomatoid malformation (CCAM), but the term CPAM is now preferred as it more accurately describes the heterogeneous nature of these lesions.
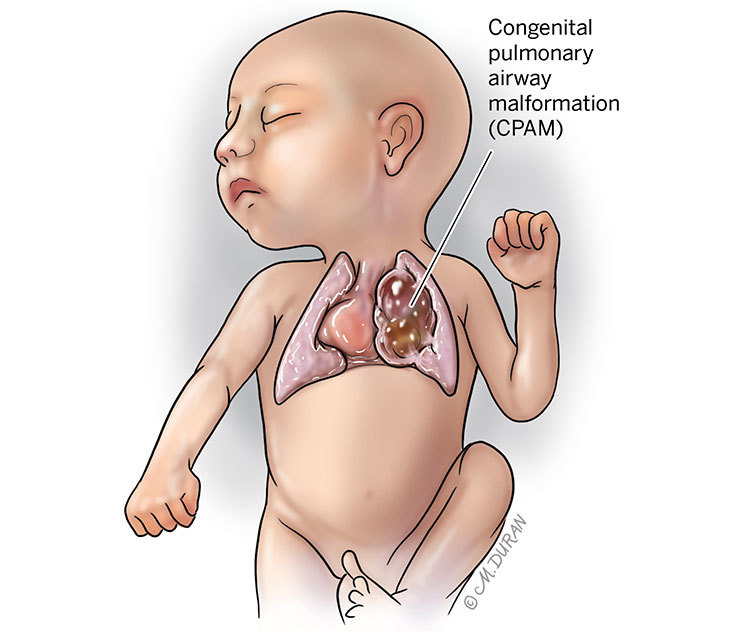
The exact cause of CPAM is not fully understood, but it is believed to be a developmental anomaly that occurs early in gestation. During the formation of the lungs, typically between the 4th and 8th week of pregnancy, the primitive airways develop into a complex branching structure. If this process is disrupted, it can lead to the formation of CPAM. Genetic factors are thought to play a role, although specific genes have not been definitively identified as the sole cause. Environmental factors during pregnancy have also been considered, but evidence remains inconclusive. It’s important to emphasize that CPAM is not caused by anything the mother did or didn’t do during pregnancy.
Types and Classification of CPAM
CPAM is classified into different types based on the microscopic appearance of the lung tissue and the size and arrangement of the cystic structures. This classification helps in predicting the potential severity and management strategies. The most commonly used classification is the one proposed by Stocker, which categorizes CPAM into five types:
Type 0: Tracheobronchomalacia
This is the rarest and most severe form, characterized by abnormalities of the trachea and bronchi, leading to airway collapse. Infants with Type 0 often experience significant breathing difficulties immediately after birth and have a poor prognosis.
Type I: Macrocystic CPAM
This is the most common type, accounting for approximately 50-60% of cases. It is characterized by one or more large cysts, typically measuring more than 2 cm in diameter. These cysts may occupy a significant portion of a lung lobe.
Type II: Microcystic CPAM
This type is characterized by multiple small cysts, usually less than 1 cm in diameter, that diffusely involve a lung lobe. Type II CPAM is often associated with other congenital anomalies, such as renal, cardiac, and gastrointestinal malformations.
Type III: Mixed Microcystic and Macrocystic CPAM
This type presents with a combination of both large and small cysts. The distribution and size of these cysts can vary widely.
Type IV: Pleuro-pulmonary Blastoma (PPB)
While technically a distinct entity, PPB was historically grouped under CCAM. It is a rare and aggressive tumor that can arise from the lung. PPB has a higher risk of malignancy.
The classification is often determined by pathological examination after surgical removal of the affected lung tissue. However, prenatal imaging techniques can provide strong indications of the type of CPAM present.
Prenatal Diagnosis and Monitoring
The diagnosis of CPAM often begins during routine prenatal ultrasounds. While many CPAMs are small and asymptomatic, larger lesions can be detected as early as the second trimester of pregnancy. These malformations typically appear as echogenic masses within the fetal chest, often with visible cystic components. The size of the lesion, its relationship to the surrounding lung tissue, and the presence of any associated abnormalities are carefully assessed.
If a CPAM is suspected on ultrasound, further investigations may be recommended. Fetal Magnetic Resonance Imaging (MRI) is often used to provide more detailed imaging of the lesion. MRI offers excellent visualization of soft tissues and can help delineate the extent of the CPAM, its relationship to major blood vessels and airways, and can also help identify other potential congenital anomalies.
Monitoring of the CPAM throughout the pregnancy is crucial. This involves serial ultrasounds and MRIs to track any changes in the size of the lesion. In some cases, CPAMs can grow during pregnancy, potentially leading to complications such as polyhydramnios (excess amniotic fluid) or hydrops fetalis (fluid accumulation in fetal tissues). Conversely, some CPAMs may remain stable or even decrease in size, offering a more optimistic outlook. This close monitoring allows the medical team to prepare for the delivery and the baby’s immediate postnatal care.
Clinical Presentation and Potential Complications
The clinical presentation of CPAM in newborns can vary widely, ranging from asymptomatic to severe respiratory distress. The severity of symptoms depends on several factors, including the size and location of the CPAM, the presence of any associated congenital anomalies, and whether the CPAM becomes infected or causes complications.
Symptoms and Signs
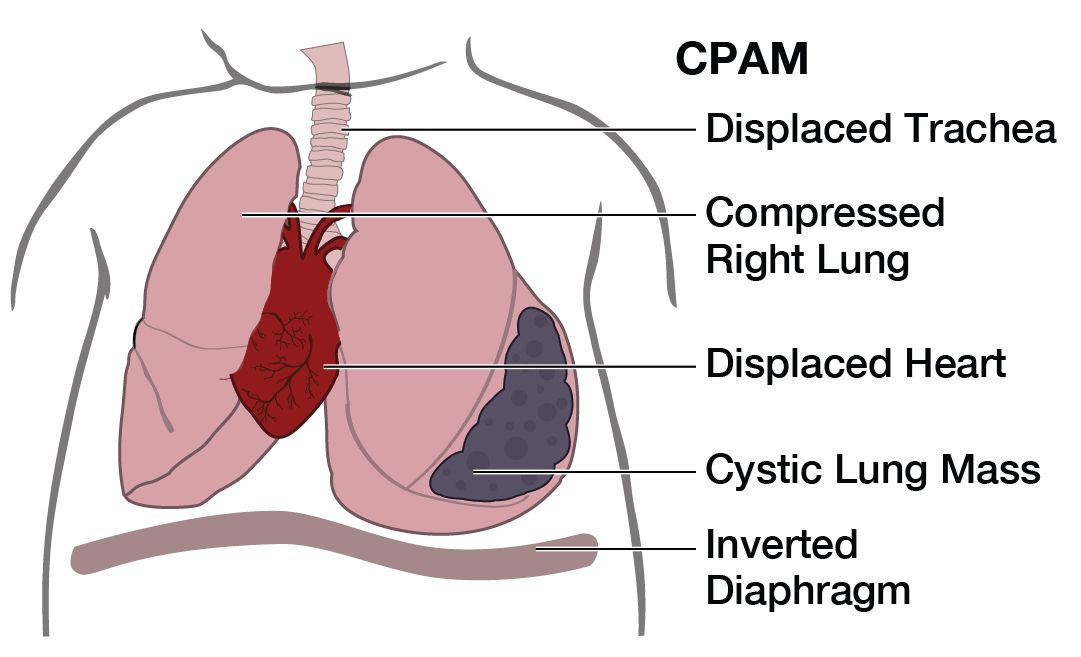
Many infants with small or non-compressing CPAMs may have no symptoms at birth and are diagnosed incidentally later in life through imaging for other reasons. However, for those with larger or more symptomatic lesions, the following signs and symptoms may be observed shortly after birth:
- Respiratory Distress: This is the most common and significant symptom. Infants may exhibit rapid breathing (tachypnea), grunting, nasal flaring, retractions (inward pulling of the chest muscles during breathing), and cyanosis (bluish discoloration of the skin due to low oxygen levels).
- Coughing and Choking: The CPAM can sometimes compress the airways, leading to a persistent cough or episodes of choking.
- Poor Feeding: Difficulty breathing can make feeding challenging, leading to poor weight gain.
- Recurrent Pneumonia: The cystic nature of CPAM can make the affected lung tissue more susceptible to infections, leading to recurrent bouts of pneumonia.
- Chest Masses: In some cases, a CPAM can present as a palpable mass in the chest.
Potential Complications
While many infants with CPAM do well with appropriate management, there are several potential complications that can arise:
- Respiratory Insufficiency: Large CPAMs can compress healthy lung tissue and the airways, leading to significant breathing problems that may require mechanical ventilation.
- Infection: The cysts within the CPAM can become infected, leading to pneumonia or abscess formation. This can be a serious complication that requires prompt antibiotic treatment.
- Hemoptysis: In older children and adults, CPAM can sometimes lead to coughing up blood, although this is rare in newborns.
- Malignancy: While extremely rare in infancy, there is a slightly increased risk of developing certain types of tumors, particularly in cases of Type IV CPAM (PPB).
- Hydrops Fetalis: In severe cases, CPAM can lead to fluid accumulation in the fetus, a condition known as hydrops fetalis, which can be life-threatening.
Management and Treatment Strategies
The management of CPAM is tailored to the individual infant, taking into account the size of the lesion, the presence of symptoms, and the overall health of the baby. The primary goal is to ensure adequate breathing and to prevent complications.
Postnatal Assessment
Upon birth, infants suspected of having CPAM undergo a thorough postnatal assessment. This typically includes a physical examination, chest X-ray, and often a chest CT scan. A CT scan provides more detailed imaging of the lung and can help delineate the extent and characteristics of the CPAM. Echocardiography (ultrasound of the heart) may also be performed to rule out any associated cardiac anomalies.
Surgical Intervention
For symptomatic infants or those with large CPAMs that pose a risk of complications, surgical removal of the affected lung lobe (lobectomy) is the definitive treatment. This procedure is usually performed shortly after birth, ideally within the first few months of life. The timing of surgery depends on the infant’s stability and the size of the lesion.
The surgical approach can be either open thoracotomy (a traditional surgical incision in the chest) or video-assisted thoracoscopic surgery (VATS). VATS is a minimally invasive technique that uses small incisions and a camera, often resulting in a faster recovery time and less scarring for the infant.
In cases where the CPAM is bilateral or involves a significant portion of both lungs, a more complex surgical approach or a staged procedure may be necessary.
Non-Surgical Management
Many infants with asymptomatic or small CPAMs may not require immediate surgical intervention. In these cases, a strategy of watchful waiting and close monitoring is often employed. Regular follow-up appointments with imaging can help detect any changes in the lesion. Some small CPAMs may resolve on their own over time.
Antibiotics may be prescribed if an infection is suspected or confirmed. In cases of severe respiratory distress, supportive care, including oxygen therapy and mechanical ventilation, may be necessary until the infant stabilizes or surgery can be performed.
Long-Term Follow-Up
Even after successful surgical removal or if the CPAM is managed non-surgically, long-term follow-up is often recommended. This allows healthcare providers to monitor for any potential late complications, such as the development of asthma-like symptoms or recurrent respiratory infections in the remaining lung tissue. Regular check-ups and occasional imaging can help ensure the child’s continued well-being.
Prognosis and Outcomes
The prognosis for infants with CPAM is generally good, especially with modern medical care. The outcome is largely dependent on the type, size, and location of the CPAM, as well as the presence of any associated congenital anomalies.
Factors Influencing Prognosis
- Size and Location of CPAM: Larger CPAMs that compress vital structures are associated with a poorer prognosis. Lesions located in a way that significantly impairs lung function or blood flow can also lead to more severe outcomes.
- Presence of Associated Anomalies: Infants with other congenital conditions, such as cardiac defects or chromosomal abnormalities, may have a more complex clinical course and a less favorable prognosis.
- Symptomatic Presentation: Infants who present with severe respiratory distress at birth are at higher risk for complications and may require more intensive management.
- Development of Infection: Recurrent or severe infections of the CPAM can lead to long-term lung damage and affect the overall prognosis.
- Type of CPAM: Type 0 CPAM has the poorest prognosis due to its association with severe airway malformations.
Long-Term Health
For the majority of infants treated for CPAM, especially those who undergo successful surgical resection, the long-term outlook is positive. They can lead normal, healthy lives. However, some individuals may experience residual effects, such as:
- Asthma-like Symptoms: Some children may develop a history of wheezing or recurrent respiratory infections, particularly if a significant portion of lung tissue was removed.
- Reduced Lung Function: Depending on the extent of the CPAM and the amount of lung tissue removed, there might be a mild reduction in overall lung function, though this is often compensated for by the remaining healthy lung.
- Psychological Impact: While the physical recovery is often excellent, parents may experience anxiety during the pregnancy and recovery period. Ongoing emotional support is important.
The medical community continues to advance its understanding and treatment of CPAM. Early diagnosis through prenatal screening, advanced imaging techniques, and prompt, individualized management strategies have significantly improved the outcomes for infants born with this congenital lung abnormality. With appropriate care and follow-up, most children affected by CPAM can thrive and develop into healthy adults.
