In the vast and intricate landscape of biological systems, efficiency, precision, and robust energy management are not merely advantageous; they are existential imperatives. From the simplest bacterium to the most complex multicellular organism, every living cell operates as a sophisticated, self-sustaining machine, meticulously processing resources and converting energy to power its myriad functions. At the heart of this universal biological “tech stack” lies a molecule that, while often overshadowed by its more energetic counterpart, Adenosine Triphosphate (ATP), serves as an indispensable linchpin in the continuous cycle of energy transformation: Adenosine Diphosphate, or ADP.
To truly appreciate the elegance and innovation embedded within natural systems, one must understand the foundational protocols that govern their operations. ADP represents a critical component in the cellular energy grid, acting as both a receiver of spent energy currency and a precursor for recharging. Its role is so fundamental that it can be likened to a universal battery state or a key intermediate in a highly optimized energy conversion algorithm, continuously being recycled and regenerated to maintain dynamic equilibrium within the cell. This article will deconstruct ADP, exploring its molecular architecture, its pivotal role in energy dynamics, its broader signaling functions, and its ubiquitous importance across all known life forms, showcasing it as a testament to nature’s unparalleled engineering ingenuity.
The Molecular Architecture of ADP: A Blueprint for Bio-Energetic Systems
Understanding ADP begins with dissecting its molecular structure, a design perfected over billions of years of evolution. This structure is not merely a random assembly of atoms but a highly functional blueprint that enables its critical roles in cellular processes.
Defining Adenosine Diphosphate: The Charged State’s Precursor
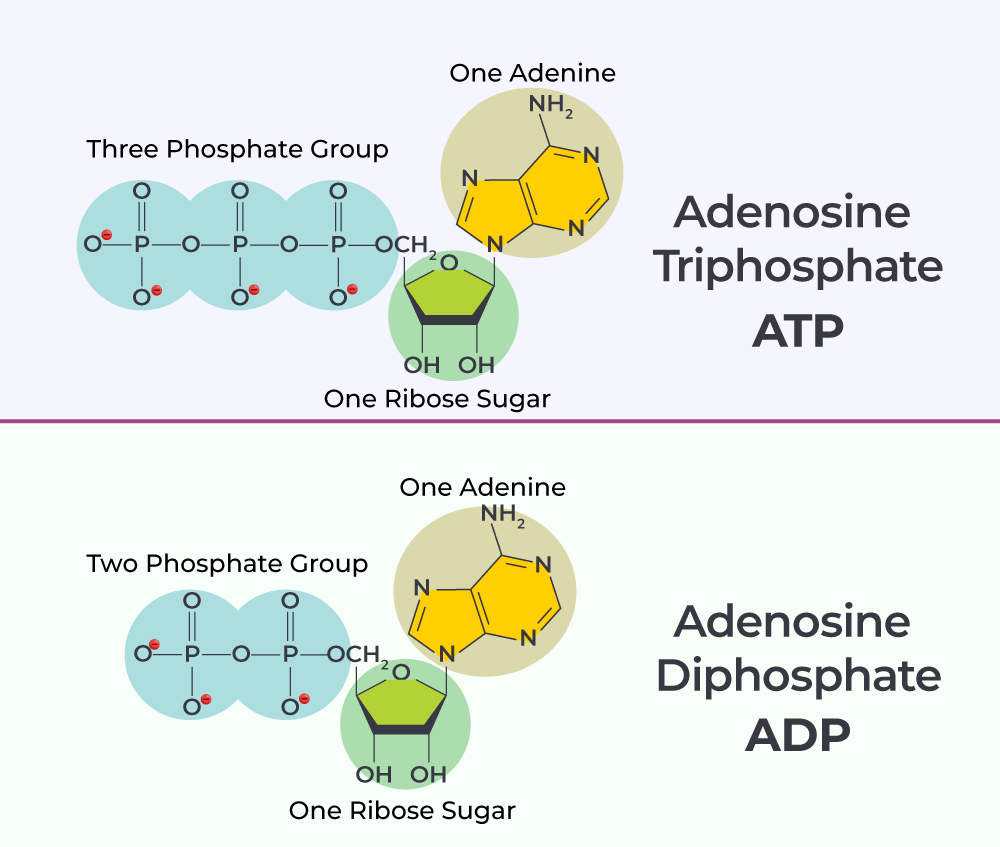
Adenosine Diphosphate is an organic compound crucial for metabolism and energy storage in living organisms. As its name suggests, “diphosphate” indicates the presence of two phosphate groups. It stands in direct contrast to Adenosine Triphosphate (ATP), which possesses three phosphate groups, and Adenosine Monophosphate (AMP), with a single phosphate group. ADP is effectively the “discharged” or “partially charged” state of the cell’s primary energy currency, poised for rapid conversion back into its high-energy ATP form. In a technological analogy, if ATP is a fully charged power cell, ADP is the momentarily depleted version, ready for an immediate recharge cycle.
The Fundamental Structure of ADP: Nucleotide Engineering
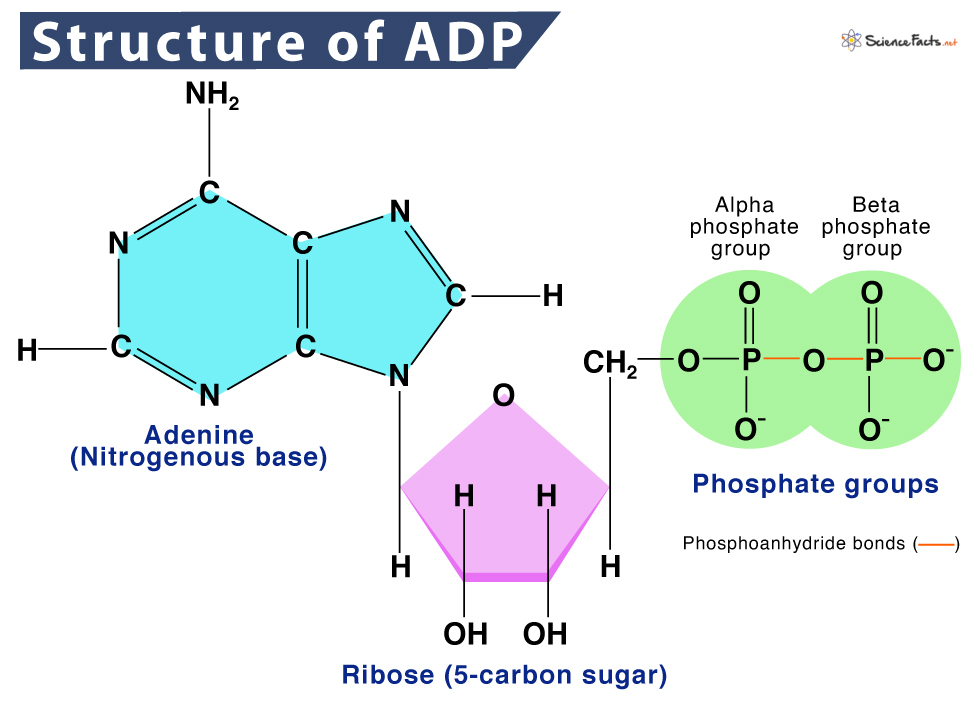
The molecular framework of ADP is a classic example of nucleotide engineering. It is comprised of three distinct but interconnected components:
- Adenine: A nitrogenous base, specifically a purine, which is also a fundamental building block of DNA and RNA. This aromatic ring structure provides stability and is crucial for specific recognition and binding within enzymes.
- Ribose: A five-carbon sugar molecule (a pentose). Ribose acts as the central scaffold, linking the adenine base to the phosphate groups. Its specific configuration dictates the orientation and reactivity of the molecule.
- Two Phosphate Groups: These are the most critical functional components for energy transfer. They are linked sequentially to the ribose sugar. The bond between the first and second phosphate group, often referred to as a “high-energy phosphate bond,” is where a significant amount of cellular energy is stored and released. While the term “high-energy” can be misleading (the bond itself isn’t intrinsically strong, but its hydrolysis releases a substantial amount of usable energy due to favorable thermodynamic conditions and stabilization of products), it signifies its crucial role in energy transactions.
This intricate yet robust design allows ADP to seamlessly integrate into complex enzymatic reactions, facilitating its rapid interconversion with ATP and enabling the continuous flow of energy that underpins all life.
ADP’s Pivotal Role in Cellular Energy Dynamics: The Bio-Reactor’s Cycle
The true genius of ADP lies not just in its structure, but in its dynamic interplay within the cellular energy infrastructure, particularly through its symbiotic relationship with ATP. This cycle represents one of nature’s most efficient and finely tuned energy management systems.
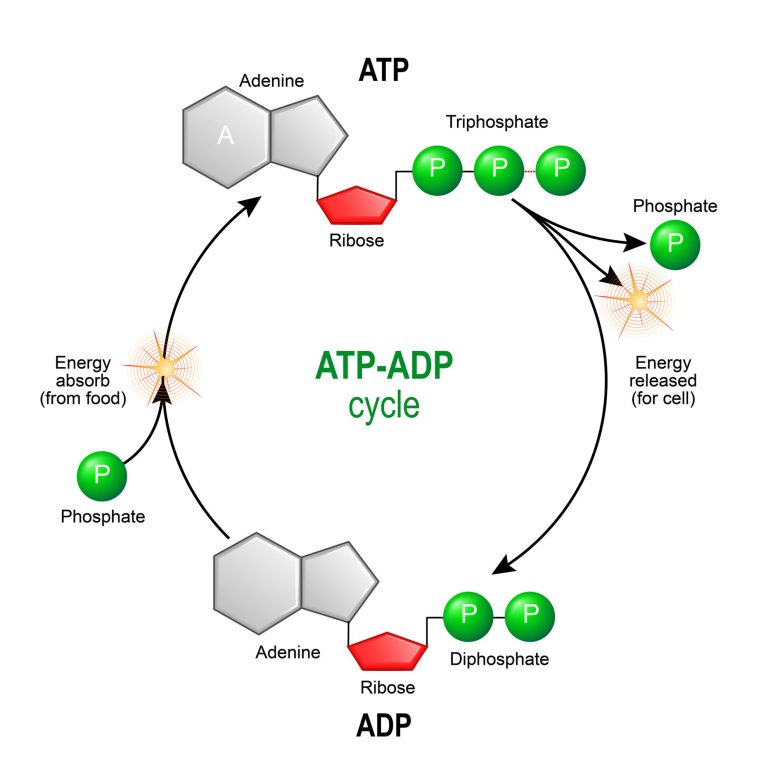
The ADP-ATP Cycle: A Biological Power Plant
The ADP-ATP cycle is the central mechanism by which cells manage their energy budget. It functions much like a rechargeable battery system within a highly advanced autonomous drone. When a cell needs energy for processes such as muscle contraction, active transport across membranes, or the synthesis of complex molecules, ATP is hydrolyzed (a water molecule is used to break a bond) into ADP and an inorganic phosphate (Pi). This reaction releases a burst of energy that can be harnessed to power cellular work.
Conversely, when energy is abundant, typically from the breakdown of fuel molecules (like glucose during cellular respiration or sunlight during photosynthesis), ADP and Pi are reunited to synthesize ATP. This process, known as phosphorylation, involves the formation of a new high-energy phosphate bond, effectively “recharging the battery.” This continuous interconversion—ATP breaking down to ADP to release energy, and ADP being rebuilt into ATP to store energy—ensures a constant and readily available supply of power for all cellular functions.
Mechanism of Energy Capture and Release: Optimized Bio-Conversion
The efficiency of energy capture and release via the ADP-ATP system is a marvel of optimized bio-conversion. The hydrolysis of ATP to ADP is an exergonic reaction, meaning it releases free energy. This energy release is not wasteful but is precisely coupled to endergonic (energy-requiring) cellular processes. This “energy coupling” is a cornerstone of metabolic regulation, ensuring that energy is expended only when and where it is needed, minimizing loss.
The synthesis of ATP from ADP, conversely, is an endergonic process that requires an input of energy. This energy comes primarily from two major cellular “power generators”:
- Cellular Respiration: In mitochondria, the chemical energy stored in glucose and other organic molecules is gradually released through a series of redox reactions. This energy is ultimately used to pump protons across a membrane, creating an electrochemical gradient. The flow of these protons back across the membrane through an enzyme called ATP synthase drives the phosphorylation of ADP to ATP. This process, known as oxidative phosphorylation, is highly analogous to a high-efficiency combustion engine powering a generator.
- Photosynthesis: In chloroplasts of plants and algae, light energy is captured and converted into chemical energy. This energy is used to power the phosphorylation of ADP to ATP (photophosphorylation) and reduce NADP+ to NADPH, providing both energy and reducing power for the synthesis of sugars. This is akin to a solar panel system directly converting radiant energy into usable power.
The precise control over these conversion mechanisms highlights the sophisticated regulatory systems inherent in biological tech.
Beyond ATP: Diverse Functions and Regulatory Significance of ADP
While its primary role in the ATP cycle is undeniable, ADP is far from a passive intermediate. It actively participates in various other cellular processes, acting as a crucial signaling molecule and a regulator of metabolic pathways.
ADP as a Signaling Molecule: Cellular Communication Protocol
Emerging research continually reveals ADP’s profound role as an extracellular signaling molecule, functioning as a vital component in intercellular communication protocols. When cells are damaged or activated, ADP can be released into the extracellular space, where it binds to specific receptors on the surface of other cells. These P2Y and P2X purinergic receptors trigger a cascade of intracellular events, leading to diverse physiological responses.
A prominent example of ADP’s signaling role is in blood clotting (hemostasis). When blood vessels are injured, platelets release ADP, which acts as a powerful activator for other platelets. This leads to platelet aggregation, forming a plug that helps seal the wound. This rapid, localized signaling mechanism is a critical safety protocol, preventing excessive blood loss, much like an automated emergency response system in a complex machine.
Regulation of Metabolic Pathways: A Feedback Mechanism
Intracellularly, ADP also functions as an allosteric regulator for several key enzymes involved in energy metabolism. High levels of ADP signal a low energy state within the cell. This signal can activate enzymes in catabolic pathways (those that break down molecules to release energy), such as glycolysis and the Krebs cycle, thereby stimulating ATP production. Conversely, high ATP levels often inhibit these pathways.
This intricate feedback mechanism allows cells to finely tune their metabolic activity in response to their energetic demands. It’s a sophisticated self-regulating system, much like an intelligent power management unit in an autonomous system, dynamically adjusting resource allocation based on real-time operational needs. For instance, in muscle cells, elevated ADP levels directly stimulate phosphofructokinase, a rate-limiting enzyme in glycolysis, ensuring a rapid supply of ATP during intense activity.
The Broader Implications of ADP in Biological Systems: Universal Engineering Principles
The omnipresence and multifunctionality of ADP underscore universal engineering principles that biological systems employ to maintain viability and adapt to changing conditions. Its roles extend into specialized cellular functions and reflect deep evolutionary conservation.
ADP in Muscle Contraction and Nerve Transmission: Driving Specific Bio-Actuators
In specialized cells, ADP is integral to their specific bio-actuator functions. In muscle contraction, the energy from ATP hydrolysis to ADP powers the movement of myosin heads along actin filaments, leading to muscle shortening. The availability of ADP and Pi is also a regulatory signal, influencing the rate of contraction. The rapid cycling between ATP and ADP in these high-demand “motors” highlights the efficiency required for biological movement.
Similarly, in nerve transmission, while ATP is directly involved in maintaining ion gradients crucial for nerve impulse generation, ADP’s role in the recycling process ensures that the energy supply remains robust for continuous signaling. The constant replenishment of ATP from ADP is vital for the sustained operation of neuronal “circuits.”
Evolutionary Conservation and Universality: A Foundational Bio-Protocol
The almost universal presence and conserved function of ADP and the ATP-ADP cycle across all domains of life—bacteria, archaea, and eukaryotes—speak volumes about its fundamental importance and evolutionary success. This indicates that the ADP-ATP system emerged very early in the history of life and has been maintained because of its unparalleled efficiency as an energy currency and regulatory molecule.
This universality underscores a core tenet of natural innovation: once an optimally efficient “protocol” or “mechanism” is developed, it is conserved and adapted across diverse systems. From a technological perspective, ADP represents a foundational bio-protocol, a low-level API (Application Programming Interface) for energy exchange that diverse biological “software” applications (cellular processes) can reliably call upon. Understanding ADP is therefore not just about biochemistry; it’s about grasping one of the most fundamental, elegant, and persistent innovations in the history of life’s complex operating systems.
In conclusion, while “what is ADP in bio” might initially seem far removed from the realms of drones and cutting-edge robotics, a deeper look reveals profound parallels. ADP is a testament to the sophisticated “tech and innovation” inherent in nature, showcasing optimized energy management, intricate signaling pathways, and robust regulatory feedback loops that continue to inspire and inform advanced technological designs. It stands as a molecular cornerstone, ensuring that the complex, dynamic machinery of life continues to operate with unparalleled precision and resilience.
