In recent years, terms like “climate change,” “global warming,” and “carbon footprint” have become part of our daily vocabulary. At the heart of these discussions lies a specific group of compounds known as greenhouse gases. But what exactly is the definition of greenhouse gases, and why are they so critical to our planet’s survival—and its current crisis?
1. Defining Greenhouse Gases (GHGs)
By definition, greenhouse gases (GHGs) are gas molecules in the Earth’s atmosphere that have the specific physical property of absorbing and emitting radiant energy within the thermal infrared range.
In simpler terms, these gases act like a thermal blanket or the glass walls of a greenhouse. They allow sunlight to pass through the atmosphere to warm the Earth’s surface but prevent a significant portion of that heat from escaping back into space.
2. How the Greenhouse Effect Works
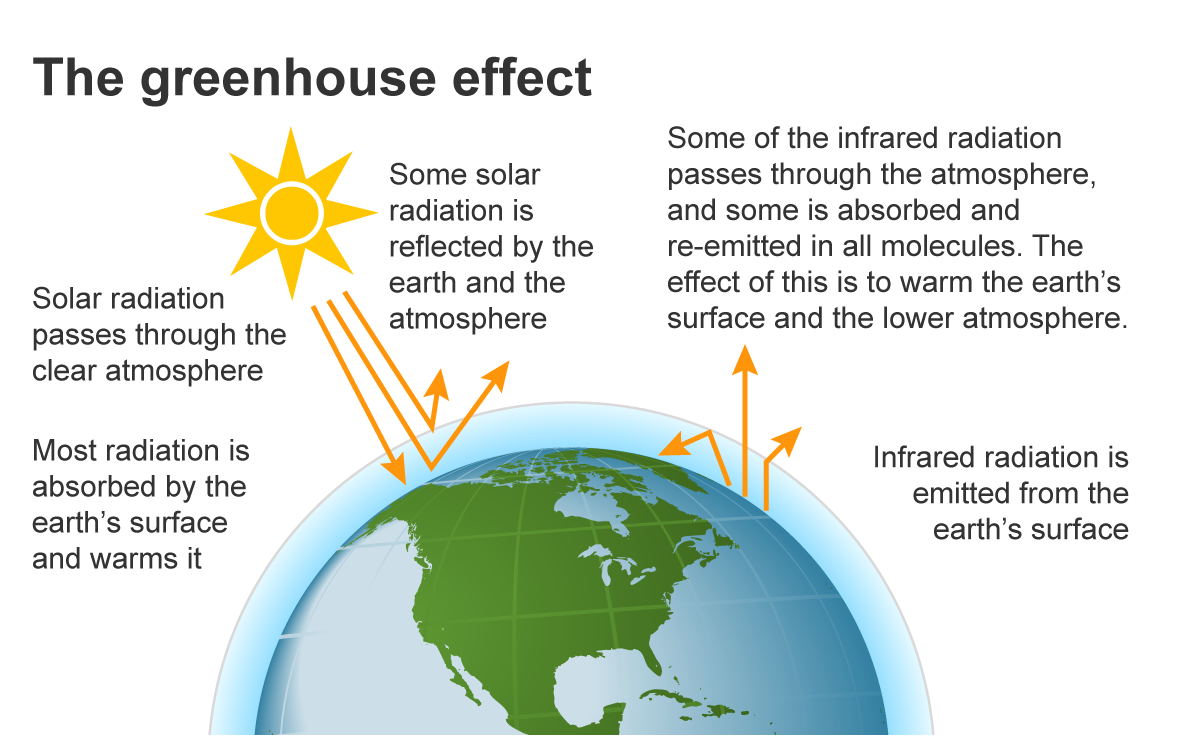
To understand the gases, we must understand the process. The “Greenhouse Effect” is a natural phenomenon:
- Solar Radiation: The sun sends energy to Earth in the form of light.
- Absorption: The Earth’s surface absorbs this energy and warms up.
- Re-radiation: The Earth then radiates this energy back toward space as infrared radiation (heat).
- Trapping Heat: Instead of escaping into the vacuum of space, greenhouse gases absorb this infrared energy and radiate it back toward the surface, keeping the planet warm enough to sustain life.
Without any greenhouse gases, the Earth’s average temperature would be roughly -18°C (0°F), rather than the comfortable 15°C (59°F) we enjoy today.
3. The Main Types of Greenhouse Gases
Not all gases in our atmosphere are greenhouse gases. Nitrogen (78%) and Oxygen (21%) make up the bulk of our air but do not trap heat. The primary GHGs include:
Carbon Dioxide ($CO_2$)
The most well-known GHG, $CO_2$ is released through natural processes like respiration and volcanic eruptions, but it is primarily driven by human activities such as burning fossil fuels (coal, oil, and natural gas) and deforestation.
Methane ($CH_4$)
Methane is far more effective at trapping heat than $CO_2$ (over 25 times more potent over a 100-year period). It is emitted during the production of coal, natural gas, and oil, as well as by livestock and the decay of organic waste in landfills.
Nitrous Oxide ($N_2O$)
Generated by agricultural and industrial activities, as well as the combustion of fossil fuels and solid waste. It is a powerful GHG that also contributes to the depletion of the ozone layer.
Fluorinated Gases (F-Gases)
These are synthetic, powerful gases emitted from a variety of industrial processes (like refrigeration and electronics manufacturing). While they are present in smaller concentrations, they have a “Global Warming Potential” (GWP) thousands of times higher than $CO_2$.
Water Vapor
Technically the most abundant greenhouse gas. However, it acts as a “feedback loop.” As the air warms due to other GHGs, it holds more water vapor, which then traps even more heat.
4. Why are Greenhouse Gases a Problem Now?
If the greenhouse effect is natural, why are we worried? The issue is concentration.
Since the Industrial Revolution, human activity has increased the concentration of these gases in the atmosphere at an unprecedented rate. This “enhanced greenhouse effect” is trapping too much heat, leading to:
- Global Warming: A steady rise in the Earth’s average temperature.
- Climate Change: Shifts in weather patterns, increased frequency of extreme storms, and melting polar ice caps.
- Ocean Acidification: Oceans absorb excess $CO_2$, which harms marine life.

5. Conclusion
The definition of greenhouse gases goes beyond simple chemistry; it describes the regulatory system for our planet’s temperature. While they are essential for making Earth habitable, the current imbalance caused by human industry is the primary driver of the global climate crisis.
Understanding what these gases are and where they come from is the first step in taking action to reduce our emissions and protect the environment for future generations.
Summary Table: Primary Greenhouse Gases
| Gas | Main Source | Longevity in Atmosphere |
|---|---|---|
| Carbon Dioxide | Fossil Fuel Combustion | 300 – 1,000 years |
| Methane | Agriculture / Landfills | ~12 years |
| Nitrous Oxide | Fertilizers / Industry | ~114 years |
| F-Gases | Refrigerants / Industrial | Weeks to Thousands of years |
