The human brain, the command center of our existence, is exquisitely protected by one of nature’s most sophisticated biological innovations: the blood-brain barrier (BBB). Far more than a simple physical partition, the BBB is a dynamic, highly selective interface that meticulously regulates the passage of substances from the bloodstream into the central nervous system (CNS). Its intricate architecture is a testament to biological engineering, designed to safeguard neuronal function from circulating toxins, pathogens, and drastic fluctuations in blood composition, while selectively allowing essential nutrients to pass through. Understanding “what the blood-brain barrier is made of” is not merely an anatomical exercise; it’s an exploration into a complex system that challenges current biotechnologies and drives continuous innovation in neuroscience, drug delivery, and disease treatment.

At its core, the BBB is formed by a specialized network of brain endothelial cells lining the capillaries within the brain. However, its functional integrity and remarkable selectivity are not solely dependent on these cells. It is an elaborate, multi-cellular structure, a true innovation of biological design, involving a constellation of cellular and extracellular components that collectively establish this vital protective shield. The exploration of these components, often requiring cutting-edge imaging and molecular technologies, reveals a biological marvel that continues to inspire novel research and therapeutic strategies.
The Core Components: A Biological Marvel Under the Microscope
The blood-brain barrier is a microanatomical masterpiece, a finely tuned biological system whose components interact synergistically to maintain the brain’s delicate internal environment. Unveiling its construction requires advanced microscopic techniques and molecular analyses, treating this biological construct as a complex piece of engineering.
Endothelial Cells: The Tight Junction Guardians
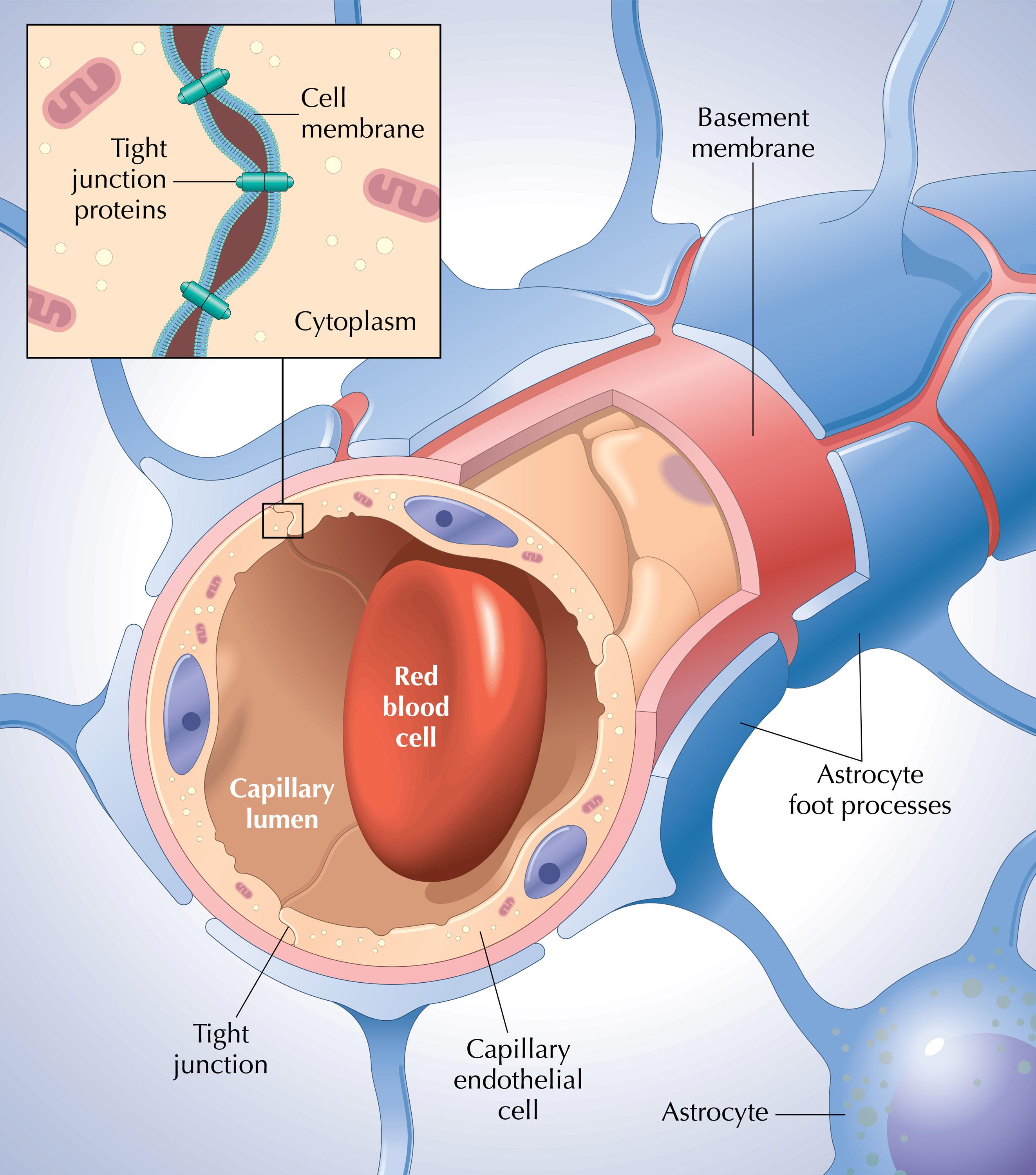
The primary structural and functional components of the BBB are the specialized endothelial cells that line the cerebral microvessels. Unlike endothelial cells in other parts of the body, those in the brain possess unique characteristics that form the actual barrier. Their most distinguishing feature, and a key innovation in their design, are the tight junctions. These junctions are multiprotein complexes that literally “seal” the paracellular pathway between adjacent endothelial cells, preventing the non-selective passage of molecules.
These tight junctions are not static; they are dynamic structures, regulated by a sophisticated network of signaling pathways. Key proteins involved in these junctions include occludin, claudins (especially claudin-3 and claudin-5), and junctional adhesion molecules (JAMs). These proteins form a continuous, impermeable seal, making the brain capillaries far less permeable than capillaries elsewhere. This biological “technology” ensures that only substances that can traverse the endothelial cells themselves – either via highly specific transporters or through lipid solubility – can enter the brain parenchyma. The selective transport systems embedded within these endothelial cells, such as efflux pumps like P-glycoprotein, actively remove undesirable substances, representing another layer of sophisticated biological filtration.
Astrocytes and Pericytes: Support Systems and Regulators
Beyond the endothelial cells, the integrity and function of the BBB are profoundly influenced by two other crucial cell types: astrocytes and pericytes. These cells form a critical part of what is known as the “neurovascular unit” (NVU), an integrated system where neurons, glial cells, and vascular cells interact.
Pericytes are mural cells embedded within the basal lamina of capillaries. They are strategically positioned to directly interact with endothelial cells, playing a critical role in inducing and maintaining BBB properties during development and in adulthood. Research, often utilizing single-cell sequencing and advanced cellular models, has revealed that pericytes regulate endothelial tight junction formation, restrict transcellular permeability, and control vessel diameter and blood flow. Their absence or dysfunction can lead to BBB breakdown, highlighting their innovative role as essential structural and regulatory components.
Astrocytes, a type of glial cell, extend specialized processes called “end-feet” that completely ensheathe the brain capillaries. While they do not form direct tight junctions, their interactions with endothelial cells are vital. Astrocytes release factors that promote and maintain the tight junction integrity of the endothelial cells, modulate transporter expression, and regulate local blood flow. Advanced imaging techniques have allowed scientists to observe these intricate cellular conversations, revealing how astrocytes act as vital communicators and regulators in the sophisticated BBB system. The interplay between endothelial cells, pericytes, and astrocytes is a prime example of biological synergy, a multi-component “system” where each part contributes to the overall stability and functionality.
Basal Lamina and Extracellular Matrix: The Foundation
Underlying the endothelial cells is a specialized layer of extracellular matrix called the basal lamina (or basement membrane). This acellular layer is primarily composed of laminin, collagen IV, fibronectin, and proteoglycans. It provides structural support to the endothelial cells and pericytes, acting as a scaffold for the neurovascular unit. The basal lamina is not merely a passive support structure; it plays an active role in cell signaling and adhesion, contributing to the overall integrity and regulatory functions of the BBB. Disruptions to this foundational layer, detectable through advanced immunofluorescence and electron microscopy, are often indicative of BBB dysfunction in various neurological conditions.
Unveiling the BBB’s Architecture: Innovative Research Technologies
Understanding the intricate components and dynamics of the BBB relies heavily on cutting-edge technological innovations. From visualizing its cellular makeup to dissecting its molecular pathways, technology is at the forefront of every significant discovery.
Advanced Imaging Techniques: From Electron Microscopy to fMRI
The physical composition of the BBB has been meticulously studied using a range of sophisticated imaging techniques. Electron microscopy (TEM, SEM) provides ultra-high resolution images, revealing the tight junctions between endothelial cells and the close apposition of pericytes and astrocyte end-feet to the capillary walls. This allows researchers to literally see what the barrier is made of at a subcellular level.
Beyond static images, live-cell imaging with fluorescent markers, often in conjunction with confocal microscopy or two-photon microscopy, allows scientists to observe the dynamics of BBB components in real-time, both in vitro and in vivo. For instance, researchers can track the movement of specific proteins or the integrity of tight junctions under various experimental conditions. On a larger scale, functional Magnetic Resonance Imaging (fMRI) and advanced PET scanning techniques are being innovatively applied to assess BBB permeability changes in living human brains, providing invaluable insights into its integrity in neurological diseases. These technologies are crucial for translating molecular discoveries into clinical understanding.
Omics Technologies: Decoding Molecular Composition
To truly understand “what the BBB is made of” at a molecular level, omics technologies have become indispensable. Proteomics allows for the comprehensive identification and quantification of all proteins present in BBB components (endothelial cells, pericytes, astrocytes), revealing the specific protein machinery that constitutes the tight junctions, transporters, and signaling pathways. Similarly, transcriptomics (e.g., RNA sequencing) provides insights into gene expression patterns, indicating which genes are active in different BBB cell types and how their activity might be modulated in health and disease.
Single-cell RNA sequencing (scRNA-seq) represents a particularly innovative advancement, allowing researchers to characterize the unique gene expression profiles of individual cells within the neurovascular unit. This has revolutionized our understanding of cell heterogeneity and how specific cell populations contribute to BBB function. These high-throughput technologies, combined with sophisticated bioinformatics, are unlocking the molecular blueprint of the BBB, revealing not just “what it’s made of” but also “how it works” at an unprecedented resolution.
In Vitro and In Vivo Models: Simulating the Barrier
Due to the BBB’s complexity and ethical limitations of human studies, innovative model systems are critical. In vitro BBB models range from simple transwell cultures of endothelial cells to highly sophisticated microfluidic “organ-on-a-chip” devices that mimic the precise flow dynamics and cellular interactions of the human neurovascular unit. These advanced models, often incorporating induced pluripotent stem cell (iPSC)-derived brain cells, allow for controlled studies of BBB permeability, drug transport, and the effects of various stimuli, acting as sophisticated in silico and in vitro testbeds for therapeutic innovation.
In vivo animal models (mice, rats, zebrafish) are also continually being refined. Genetically engineered models, where specific BBB components can be manipulated, offer powerful tools to study the role of individual proteins or cell types. Techniques like intravital microscopy in transparent animal models provide real-time visualization of BBB dynamics within a living organism, pushing the boundaries of what can be observed in a complex biological system.
The BBB as a Frontier of Innovation: Challenges and Opportunities
The BBB’s remarkable protective capabilities present both a formidable challenge and an immense opportunity for medical innovation. Its selective nature, while vital for brain health, notoriously hinders the delivery of therapeutic drugs to the CNS, impacting treatments for conditions ranging from brain tumors to neurodegenerative diseases.
Navigating Drug Delivery: Overcoming the Barrier
The tight junctions and active efflux pumps of the BBB mean that over 98% of small-molecule drugs and virtually all large-molecule biotherapeutics cannot effectively reach their targets in the brain. This “drug delivery crisis” has spurred massive innovation in drug design and delivery strategies. Nanotechnology is a key player, with researchers developing nanoparticles, liposomes, and polymeric micelles designed to encapsulate drugs and either exploit existing BBB transporters or temporarily open the barrier.
Other innovative approaches include receptor-mediated transcytosis, where drugs are conjugated to ligands that bind to specific receptors on the BBB endothelial cells (e.g., transferrin receptor), tricking the barrier into internalizing and transporting the drug. Focused ultrasound technology is another groundbreaking innovation, capable of transiently and reversibly opening the BBB in targeted brain regions, allowing for localized drug delivery without affecting the entire barrier. These technological advancements aim to leverage or temporarily circumvent the BBB’s inherent design, transforming it from an obstacle into a pathway for treatment.
Understanding Neurological Disorders: Insights from BBB Research
Breakdown or dysfunction of the BBB is increasingly recognized as a critical factor in the pathogenesis and progression of numerous neurological disorders, including Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, stroke, and brain tumors. Innovations in BBB research are providing crucial insights into these conditions. By understanding how the barrier’s components are altered in disease states (e.g., loosening of tight junctions, changes in transporter expression, pericyte loss), researchers are identifying new diagnostic biomarkers and therapeutic targets.
For example, observing the leakage of contrast agents across the BBB using advanced MRI techniques provides early indicators of inflammation or damage. Developing innovative strategies to restore BBB integrity, rather than just bypass it, is a burgeoning area of therapeutic innovation, promising new avenues for disease prevention and treatment.
Future Innovations in BBB Research and Manipulation
The future of BBB research is intrinsically linked to the continued evolution of technology and innovative thinking. The complexity of the barrier demands increasingly sophisticated tools and interdisciplinary approaches.
AI and Machine Learning: Predicting Permeability and Design
Artificial intelligence (AI) and machine learning (ML) are poised to revolutionize BBB research. AI algorithms can analyze vast datasets from proteomics, transcriptomics, and drug screening experiments to identify patterns, predict drug permeability, and even suggest novel molecules likely to cross the barrier. Machine learning models can be trained on molecular features of drugs to predict their interaction with BBB transporters and tight junction proteins, accelerating the drug discovery process. This represents a paradigm shift, moving from laborious experimental screening to intelligent, data-driven design. Furthermore, AI could help model the complex interactions within the neurovascular unit, offering a deeper understanding of its dynamic regulation.

Targeted Delivery Systems: Nanotechnology and Gene Editing
The precision of drug delivery to the brain will continue to advance through nanotechnology and potentially gene editing. Future innovations in nanotechnology will focus on even more sophisticated “smart” nanoparticles that can respond to specific stimuli (e.g., pH, enzyme activity) within the brain environment, releasing their therapeutic cargo only at the target site. Gene editing technologies like CRISPR-Cas9 offer the potential to directly modify BBB components in specific disease contexts, either to enhance barrier function where it is compromised or to facilitate drug passage where it is needed. This level of precise manipulation, still largely in early research phases, represents the ultimate frontier in innovating how we interact with and utilize the blood-brain barrier for therapeutic gain.
In conclusion, the blood-brain barrier, a complex biological innovation of tight junctions, specialized cells, and intricate molecular interactions, stands as a critical protector of brain health. Its very existence has driven continuous technological advancement and innovation, from the microscopic tools required to unravel its composition to the cutting-edge strategies developed to precisely navigate or manipulate it for therapeutic purposes. Understanding “what the blood-brain barrier is made of” is not just a scientific question; it’s a gateway to unlocking novel treatments for a myriad of neurological disorders and represents a vibrant, evolving frontier of tech and innovation in biomedicine.
