The term “hypertonic solution” might sound complex, conjuring images of laboratories and intricate scientific processes. However, at its core, understanding hypertonic solutions reveals fascinating principles that underpin advancements in diverse technological fields, from sophisticated medical treatments to cutting-edge material science. While originating in biological and chemical contexts, the concept of osmotic pressure and the behavior of solutions with differing solute concentrations has profound implications for innovation, particularly in areas where precise fluid management and cellular interaction are critical. This article will explore what hypertonic solutions are, the fundamental science behind them, and their burgeoning role within the realm of Tech & Innovation.
The Fundamental Science of Osmosis and Tonicity
To grasp the essence of a hypertonic solution, we must first delve into the fundamental concepts of osmosis and tonicity. These phenomena are driven by the inherent behavior of water molecules across selectively permeable membranes.
Osmosis: The Movement of Water
Osmosis is a specific type of diffusion that refers to the net movement of solvent molecules, typically water, through a selectively permeable membrane from a region of higher solvent concentration (lower solute concentration) to a region of lower solvent concentration (higher solute concentration). Imagine a semi-permeable barrier, like a fine sieve, that allows water molecules to pass through but restricts the movement of larger solute particles (like salts, sugars, or proteins). In this scenario, water will naturally migrate from the side where it is more abundant and less “crowded” by solutes, towards the side where it is less abundant and more “crowded” by solutes. This movement continues until equilibrium is reached, meaning the concentration of solutes is equal on both sides, or until the pressure exerted by the opposing forces balances the osmotic pressure.
Tonicity: Defining Solution Strength
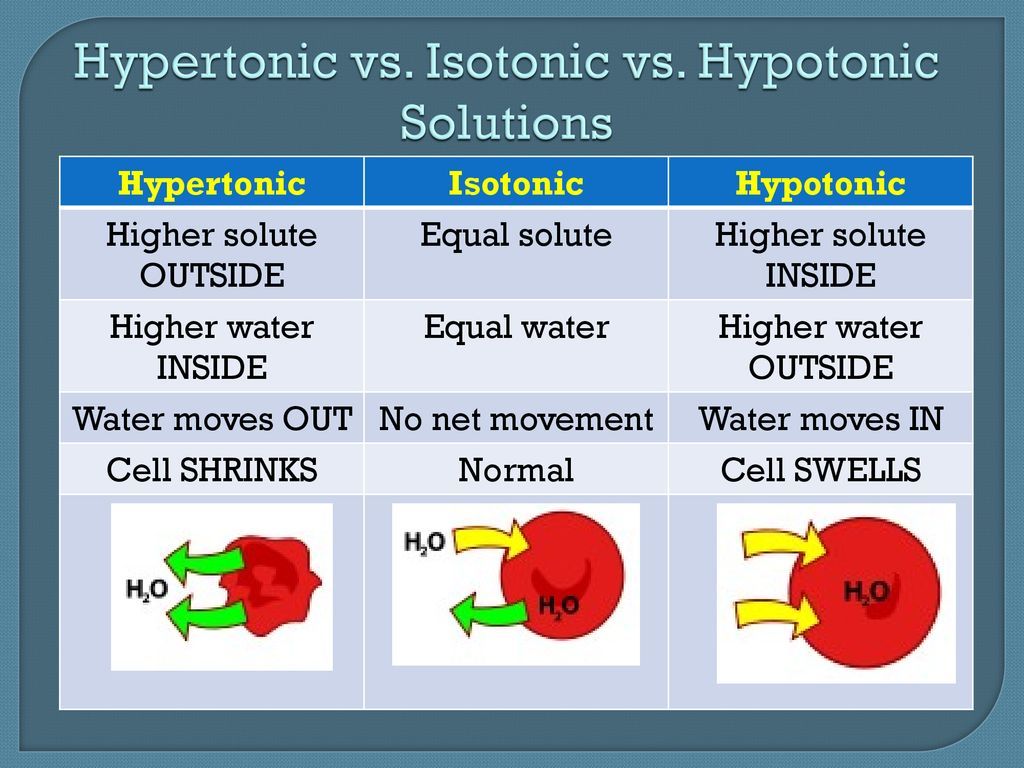
Tonicity, a term closely related to osmosis, describes the relative concentration of solutes dissolved in a solution compared to another solution, separated by a semipermeable membrane. It is a concept used to predict the direction and extent of water movement across that membrane. Tonicity is particularly important in biological systems, as it dictates how cells will respond to their surrounding environment. When we talk about solutions in relation to tonicity, we typically compare them to the intracellular fluid of a cell. There are three primary classifications of solutions based on their tonicity: isotonic, hypotonic, and hypertonic.
-
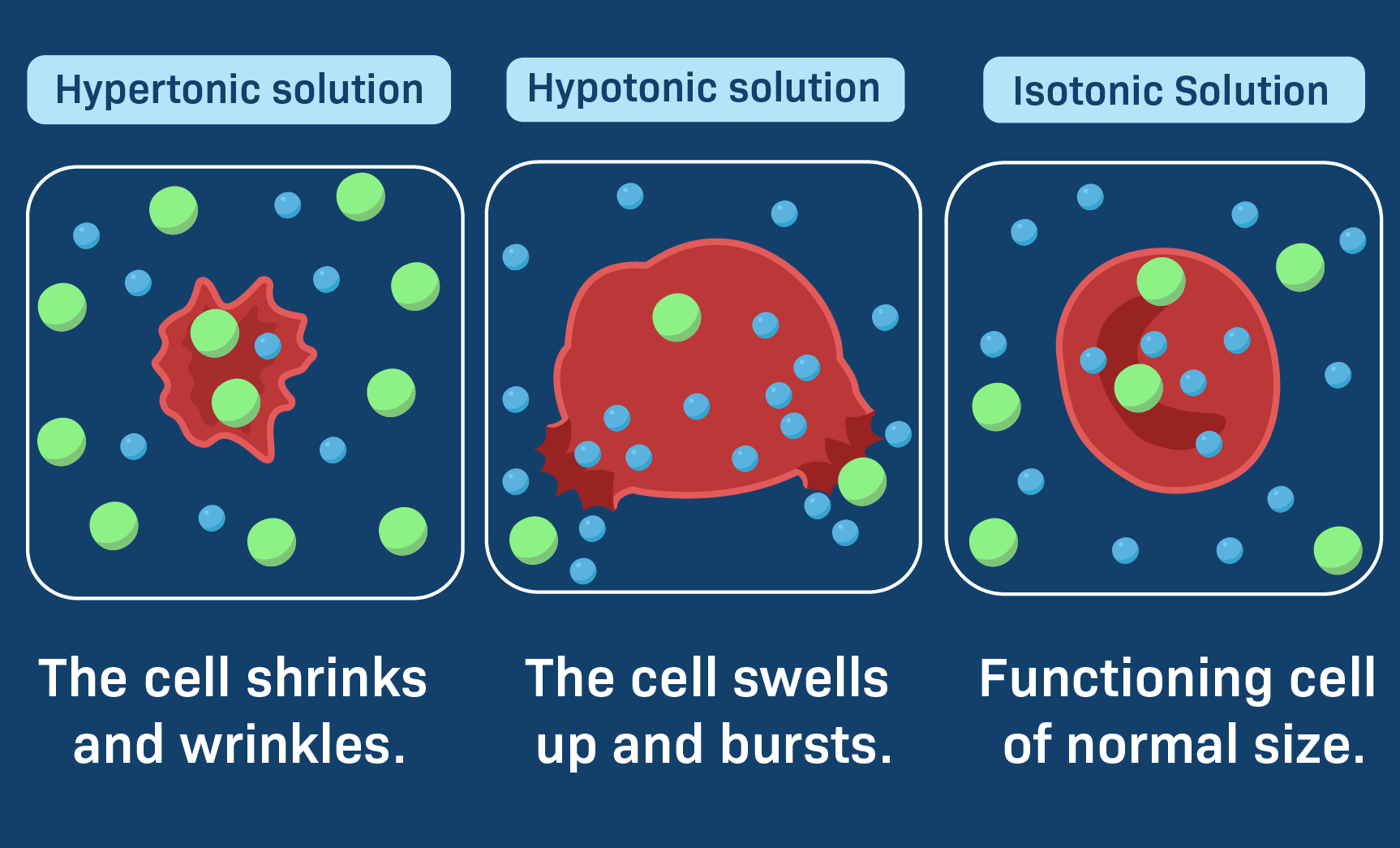
Isotonic Solutions: In an isotonic solution, the solute concentration is the same both inside and outside the cell. This means there is no net movement of water across the cell membrane, and the cell maintains its normal shape and volume. For example, physiological saline (0.9% sodium chloride) is isotonic to human blood.
-
Hypotonic Solutions: A hypotonic solution has a lower solute concentration and therefore a higher water concentration than the cell’s interior. When a cell is placed in a hypotonic solution, water will move into the cell via osmosis. This influx of water can cause the cell to swell and, in some cases, burst (lysis), particularly in animal cells which lack a rigid cell wall. Plant cells, with their rigid cell walls, will become turgid, which is a healthy state for them.
-
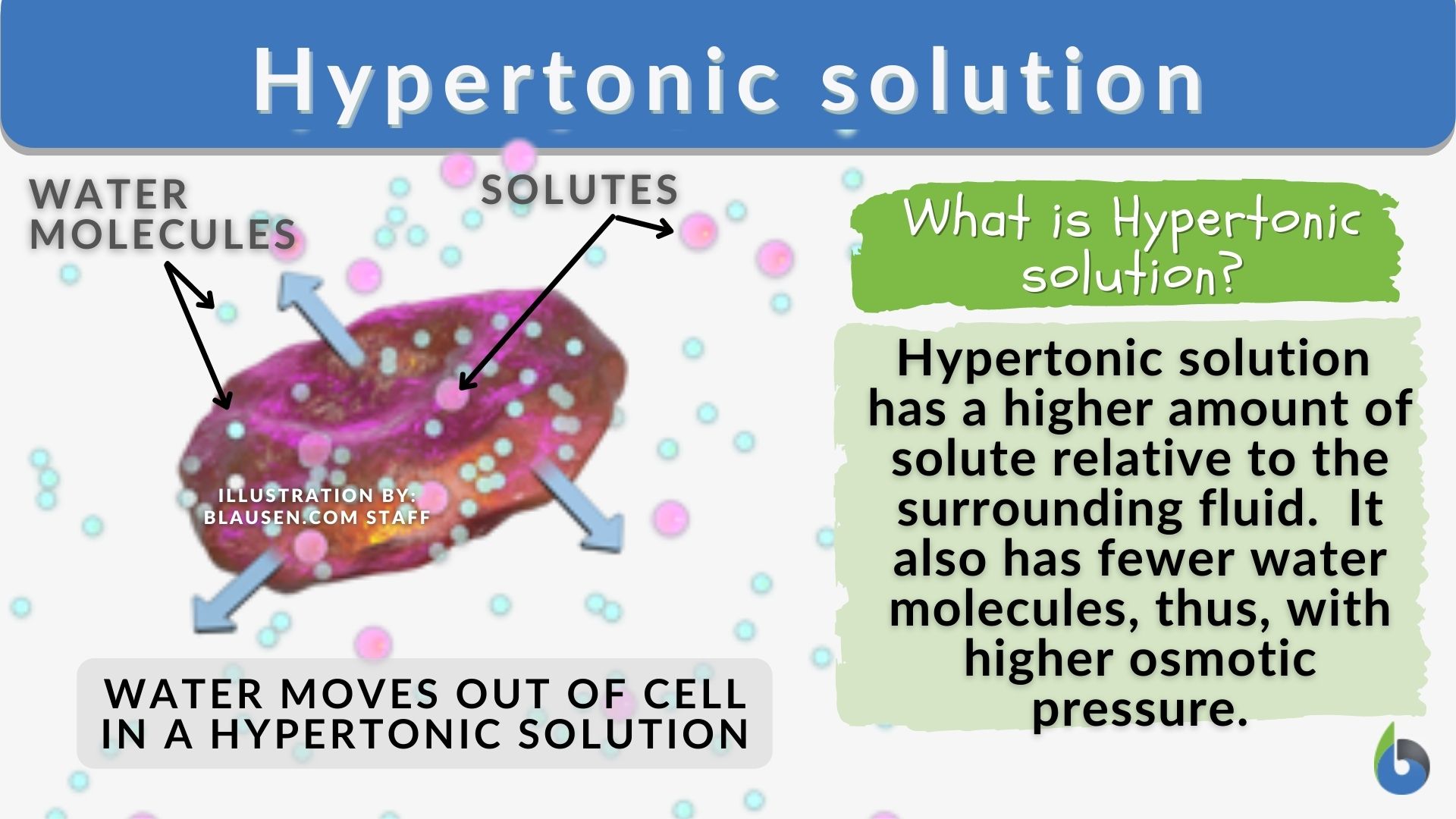
Hypertonic Solutions: This is our focus. A hypertonic solution is characterized by having a higher solute concentration and, consequently, a lower water concentration compared to the cell’s interior. When a cell is immersed in a hypertonic solution, the osmotic gradient drives water to move out of the cell and into the surrounding solution. This outward movement of water leads to the cell shrinking or becoming crenated. In plant cells, this process is known as plasmolysis, where the cell membrane pulls away from the cell wall.
The Properties and Mechanisms of Hypertonic Solutions
Understanding the defining characteristics and the underlying physical principles of hypertonic solutions is crucial for appreciating their applications.
High Solute Concentration, Low Water Potential
The defining feature of a hypertonic solution is its elevated concentration of dissolved solutes. This can be achieved by dissolving a significant amount of a substance, such as salt, sugar, or even larger molecules, in a solvent, typically water. The presence of these solute particles effectively “competes” with water molecules for space and interaction. As a result, the concentration of free water molecules is lower in a hypertonic solution compared to a solution with fewer solutes.
This difference in free water concentration translates to a difference in water potential. Water potential is a measure of the free energy of water per unit volume. Water moves from areas of higher water potential to areas of lower water potential. In a hypertonic solution, the solute molecules reduce the free energy of the water, thus lowering its water potential. Consequently, if a cell with a higher internal water potential is placed in this hypertonic environment, water will naturally flow from the cell to the solution.
Osmotic Pressure and Water Movement
The tendency for water to move across a semipermeable membrane from a region of higher water concentration to a region of lower water concentration is a powerful force known as osmotic pressure. Osmotic pressure is essentially the minimum pressure that needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane. In the context of a hypertonic solution, the high solute concentration creates a significant osmotic pressure that draws water out of adjacent cells or less concentrated environments.
This principle is not merely theoretical; it has tangible effects. For instance, if you sprinkle salt on a slug or a piece of fruit, the salt draws water out of the organism’s cells, dehydrating and effectively preserving it. This rapid dehydration is a direct consequence of the hypertonic nature of the salt solution formed by the salt dissolving in the moisture on the surface.
Factors Influencing Hypertonicity
Several factors determine whether a solution is hypertonic relative to another:
- Concentration of Solutes: This is the primary factor. A solution with more dissolved particles per unit volume is more likely to be hypertonic.
- Type of Solutes: While concentration is key, the molecular size and charge of solutes can influence their interaction with the solvent and the membrane, subtly affecting osmotic behavior. However, for general tonicity, the total molar concentration of dissolved particles is the most significant determinant.
- Temperature: Temperature affects the kinetic energy of molecules, influencing the rate of diffusion and osmosis, but it does not change the fundamental osmotic gradient determined by solute concentrations.
- Nature of the Membrane: The permeability of the membrane is critical. For osmosis to occur, the membrane must be selectively permeable, allowing the solvent (water) to pass but restricting the passage of solutes.
Applications of Hypertonic Solutions in Tech & Innovation
While the biological implications of hypertonic solutions are well-established, their principles are increasingly being leveraged and engineered within various technological domains. The ability to precisely control water movement and solute concentrations opens doors for novel solutions in medicine, materials science, and beyond.
Advanced Drug Delivery Systems
One of the most significant areas where the principles of hypertonic solutions are being applied in Tech & Innovation is in advanced drug delivery. Traditional drug delivery methods often struggle with targeted release, stability, and bioavailability. Hypertonic solutions offer innovative pathways to overcome these challenges.
- Encapsulation and Controlled Release: Nanoparticles and microparticles are being engineered to encapsulate therapeutic agents. The shell of these carriers can be designed to interact with the body’s fluids in specific ways. By formulating the encapsulated drug within a hypertonic core relative to the surrounding interstitial fluid, it’s possible to maintain the drug’s stability and prevent premature release. Conversely, when these carriers encounter a specific biological environment (e.g., a tumor microenvironment which may have altered tonicity), they can be triggered to release their payload.
- Osmotic Pump Systems: For continuous and precisely controlled drug administration, osmotic pumps are a sophisticated technological application. These devices utilize the principle of osmosis to deliver medication at a steady rate over extended periods. A semipermeable membrane encloses a reservoir containing the drug solution and a separate compartment containing a salt or sugar solution. Water from the body’s tissues diffuses into the salt/sugar compartment, increasing its volume and exerting pressure that forces the drug solution out through a precisely engineered valve. The rate of drug delivery is dictated by the osmotic pressure of the internal solution and the properties of the semipermeable membrane, offering a technologically controlled hypertonic mechanism.
Nanotechnology and Material Science
The precise manipulation of fluids at the nanoscale is a cornerstone of modern nanotechnology and materials science, where hypertonic principles play a vital role.
- Self-Assembly of Nanomaterials: The controlled dehydration of materials using hypertonic environments can drive the self-assembly of nanostructures. For example, certain polymers or protein aggregates might fold or arrange themselves in specific patterns as water is withdrawn from their surroundings, leading to the formation of complex architectures with tailored properties for applications in electronics, sensors, or biomaterials.
- Membrane Technologies and Desalination: Advances in reverse osmosis (RO) and other filtration technologies, crucial for desalination and water purification, rely heavily on managing osmotic pressures. While RO actively applies pressure to overcome osmotic gradients, the fundamental understanding of how solute concentration differences drive water movement in hypertonic scenarios is essential for designing efficient membranes and optimizing filtration processes. Innovations in creating membranes that selectively allow water passage while effectively blocking hypertonic solute concentrations are a testament to this understanding.
- Hydrogels and Swelling Behavior: Hydrogels, widely used in biomedical implants, drug delivery, and soft robotics, are cross-linked polymer networks that can absorb large amounts of water. Their swelling behavior is directly influenced by the tonicity of the surrounding fluid. By incorporating specific solutes into hydrogels or by placing them in hypertonic external solutions, engineers can precisely control their expansion and contraction, enabling the development of smart materials that respond to environmental cues.
Biomedical Diagnostics and Therapeutics
Beyond drug delivery, hypertonic solutions are finding diverse applications in diagnostics and therapeutic interventions within the broader tech landscape.
- Contrast Agents in Imaging: In certain medical imaging techniques, such as MRI or CT scans, contrast agents are used to enhance the visibility of tissues and structures. Some of these agents may be formulated in solutions with specific tonicities to influence their distribution within the body or their interaction with biological tissues, leveraging osmotic principles for better diagnostic resolution.
- Wound Healing Technologies: Hypertonic saline dressings have been used for decades in wound care. From a technological perspective, the development of advanced wound dressings often incorporates principles of moisture management. Hypertonic environments within a dressing can help draw excess fluid and exudate from a wound, promoting a cleaner healing environment and reducing the risk of infection, showcasing a practical application of hypertonicity in a bio-integrated technological context.
- Cell Culture and Bioprocessing: In biotechnological applications, maintaining optimal conditions for cell growth and function is paramount. The tonicity of the culture medium directly impacts cell viability and behavior. Precise control over the solute concentration in cell culture media, often through the use of buffer solutions and nutrient formulations, ensures that cells are not exposed to hypotonic or hypertonic stresses that could impede their growth or disrupt their function, a critical aspect of bioprocessing innovation.
In conclusion, while the term “hypertonic solution” originates from the fundamental science of osmosis, its implications extend far beyond the biological realm. As technology continues to advance, our ability to harness and manipulate the principles of osmotic pressure and solute concentration is leading to innovative solutions in drug delivery, nanotechnology, materials science, and biomedical applications. The careful design and application of hypertonic environments are becoming increasingly vital tools in the arsenal of technological innovation, pushing the boundaries of what is possible in precision engineering and scientific discovery.
