Understanding the electrical properties of molecules is fundamental to many scientific disciplines, from chemistry and physics to materials science. One of the key concepts in this area is the dipole moment, a measure of the separation of positive and negative electrical charges within a system. When a molecule possesses a non-zero dipole moment, it signifies an inherent asymmetry in its charge distribution, leading to distinct electrical behaviors. This article delves into the nature of non-zero dipole moments, explaining what they mean for molecular polarity and their implications.
The Concept of a Dipole Moment
At its core, a dipole moment arises when there is a separation of positive and negative charges. Imagine two equal and opposite charges, +q and -q, separated by a distance ‘d’. This arrangement constitutes an electric dipole. The strength of this dipole is quantified by the dipole moment vector, denoted by $mu$. Its magnitude is the product of the magnitude of the charges and the distance between them ($mu = q times d$), and its direction points from the negative charge to the positive charge.

In the context of molecules, this charge separation is not typically due to discrete point charges but rather to differences in the electronegativity of the atoms involved in chemical bonds. Electronegativity is an atom’s ability to attract shared electrons in a chemical bond. When two atoms with significantly different electronegativities form a covalent bond, the electrons in that bond are pulled more towards the more electronegative atom. This creates a polar covalent bond, where one end of the bond carries a partial negative charge ($delta^-$) and the other carries a partial positive charge ($delta^+$).
Individual Bond Dipole Moments
Every polar covalent bond within a molecule contributes to the overall electrical character of that molecule. These individual bond dipoles can be visualized as vectors, each originating from the less electronegative atom and pointing towards the more electronegative atom. The magnitude of these bond dipoles depends on the difference in electronegativity between the bonded atoms and the bond length. For instance, a bond between oxygen and hydrogen (O-H) is highly polar due to oxygen’s significantly higher electronegativity compared to hydrogen.
Molecular Geometry and Net Dipole Moment
While individual polar bonds create local charge separations, the overall polarity of a molecule is determined by the vector sum of all its individual bond dipole moments. This is where molecular geometry plays a crucial role. The three-dimensional arrangement of atoms in a molecule dictates how these individual bond dipoles combine.
If the bond dipoles are arranged symmetrically such that they cancel each other out, the molecule will have no net dipole moment, even if it contains polar bonds. Conversely, if the bond dipoles do not cancel out due to an asymmetrical arrangement, the molecule will possess a net dipole moment, indicating that it is a polar molecule.
What Constitutes a Non-Zero Dipole Moment?
A non-zero dipole moment is the defining characteristic of a polar molecule. It signifies that there is a persistent separation of positive and negative charge across the entire molecule, resulting in a net electrical asymmetry. This asymmetry arises from two primary factors working in conjunction:
Polar Bonds
The presence of polar covalent bonds is a prerequisite for a molecule to have a non-zero dipole moment. As discussed, these bonds occur when atoms with different electronegativities share electrons unequally. The greater the difference in electronegativity, the more polar the bond, and the larger the individual bond dipole moment.
Asymmetrical Molecular Geometry
Even if a molecule contains polar bonds, it will only exhibit a non-zero dipole moment if its geometry is asymmetrical. This asymmetry ensures that the vector sum of the individual bond dipoles does not result in zero. Common examples of asymmetrical molecular geometries that lead to polar molecules include:
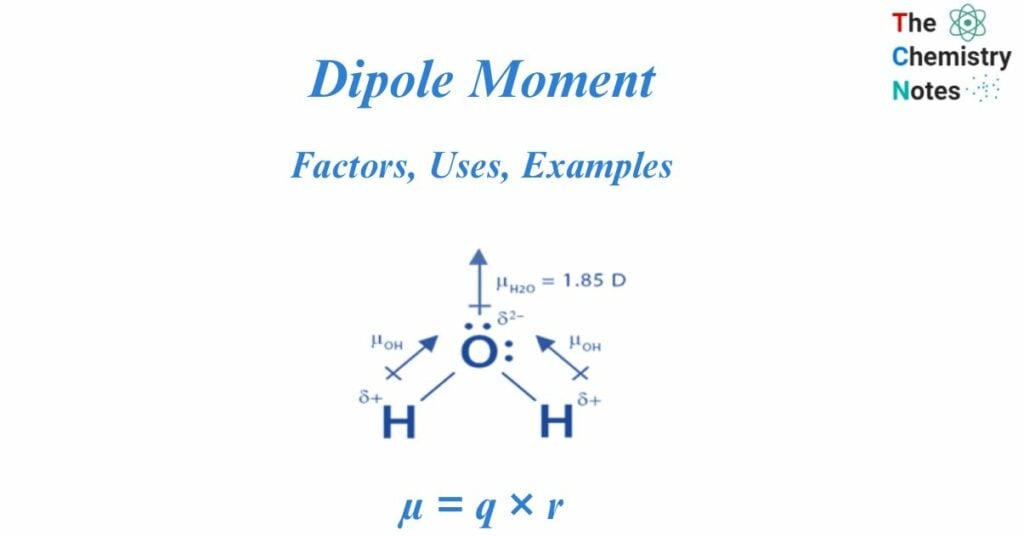
- Bent or V-shaped molecules: Water (H₂O) is a classic example. The oxygen atom is more electronegative than the hydrogen atoms, creating polar O-H bonds. The molecule has a bent geometry, with the two hydrogen atoms positioned at an angle to the oxygen. The bond dipoles of the two O-H bonds do not cancel out; instead, they add up to create a significant net dipole moment pointing towards the oxygen atom.
- Trigonal pyramidal molecules: Ammonia (NH₃) is another illustration. Nitrogen is more electronegative than hydrogen, resulting in polar N-H bonds. The molecule adopts a trigonal pyramidal shape, with the nitrogen atom at the apex. The three N-H bond dipoles combine to form a net dipole moment pointing towards the nitrogen atom.
- Molecules with lone pairs: Lone pairs of electrons on a central atom can also contribute to molecular asymmetry and influence the net dipole moment.
In contrast, molecules with symmetrical geometries, such as linear molecules like carbon dioxide (CO₂) or tetrahedral molecules like carbon tetrachloride (CCl₄), can have polar bonds but will be nonpolar because their bond dipoles cancel each other out due to the symmetrical arrangement.
Polar vs. Nonpolar Molecules
The presence or absence of a non-zero dipole moment is the fundamental distinction between polar and nonpolar molecules.

Polar Molecules
Polar molecules, characterized by a net dipole moment, possess distinct positive and negative poles. This charge separation has profound implications for their physical and chemical properties.
- Intermolecular Forces: Polar molecules interact with each other through dipole-dipole forces. The positive end of one molecule is attracted to the negative end of another. These forces are stronger than London dispersion forces (which affect all molecules) but generally weaker than hydrogen bonds.
- Solubility: “Like dissolves like” is a common adage in chemistry. Polar solvents, such as water, are effective at dissolving other polar solutes because they can effectively surround and stabilize the charged regions of the solute molecules. Nonpolar substances, on the other hand, do not dissolve well in polar solvents, and vice versa.
- Boiling and Melting Points: Due to stronger intermolecular forces (dipole-dipole interactions), polar molecules generally have higher boiling and melting points compared to nonpolar molecules of similar molecular weight.
- Behavior in Electric Fields: When placed in an external electric field, polar molecules will align themselves with the field, with their positive poles pointing towards the negative electrode and their negative poles towards the positive electrode.
Nonpolar Molecules
Nonpolar molecules, by definition, have no net dipole moment. This can occur in two main scenarios:
- Molecules composed of identical atoms: Diatomic molecules like oxygen (O₂), nitrogen (N₂), or hydrogen (H₂) are nonpolar because the two atoms have identical electronegativities, resulting in nonpolar covalent bonds.
- Molecules with polar bonds arranged symmetrically: As mentioned earlier, molecules like carbon dioxide (CO₂) or methane (CH₄) have polar bonds, but their symmetrical geometries cause the individual bond dipoles to cancel out, resulting in a net dipole moment of zero.
Nonpolar molecules primarily interact through weaker London dispersion forces. Consequently, they tend to have lower boiling and melting points and are typically soluble in nonpolar solvents.
Implications of Non-Zero Dipole Moments
The existence of a non-zero dipole moment is not merely an abstract chemical concept; it underpins a vast array of phenomena and technological applications.
Chemical Reactivity
The polarity of a molecule significantly influences its reactivity. The partially positive and negative regions of a polar molecule can be attractive sites for nucleophiles (electron-rich species) and electrophiles (electron-deficient species), respectively. This dictates the types of chemical reactions a molecule can undergo. For example, the polarity of water makes it an excellent solvent for many ionic and polar compounds, facilitating a wide range of chemical reactions in aqueous solutions.
Biological Systems
Biological molecules, such as proteins, carbohydrates, and nucleic acids, are often highly polar due to the presence of functional groups containing oxygen, nitrogen, and hydroxyl (-OH) groups. This polarity is crucial for their function. For instance, the polar nature of the water molecule is essential for the formation of cell membranes, protein folding, and the transport of nutrients and waste products within organisms. The specific arrangement of polar and nonpolar regions within a protein molecule determines its three-dimensional structure and its ability to interact with other molecules.
Materials Science
The dielectric properties of materials, which are crucial for applications in electronics, capacitors, and insulators, are directly related to their molecular polarity. Materials composed of polar molecules can exhibit significant polarization when subjected to an electric field. This ability to store electrical energy makes them valuable in electronic components. The development of new polar materials with specific dielectric characteristics is an ongoing area of research.
Solvents and Separation Techniques
The ability of solvents to dissolve substances is directly linked to their polarity. Understanding the dipole moments of solvents and solutes is essential for designing effective separation processes, such as chromatography and extraction. For example, in liquid chromatography, the choice of a polar or nonpolar mobile phase can be manipulated to selectively elute compounds based on their polarity.
Industrial Applications
From detergents that work by emulsifying grease (nonpolar) in water (polar) to the formulation of paints, adhesives, and pharmaceuticals, the understanding and manipulation of molecular polarity are integral to numerous industrial processes. The ability to predict and control the interactions between molecules based on their dipole moments is a cornerstone of modern chemical engineering and product development.
In conclusion, the concept of a non-zero dipole moment is fundamental to understanding molecular behavior. It directly determines whether a molecule is polar or nonpolar, influencing its physical properties, chemical reactivity, and interactions with other molecules and external fields. This understanding is not just academic; it has far-reaching practical implications across science, technology, and everyday life.
