In the realm of advanced diagnostics and biomedical research, the question “what is b cell lymphoma cancer” is not solely a clinical inquiry but also a profound challenge addressed by cutting-edge cameras and imaging technologies. To truly understand a complex cellular anomaly like B-cell lymphoma—a type of cancer affecting B lymphocytes, a critical component of the immune system—one must first be able to visualize it, characterize its microscopic features, assess its metabolic activity, and map its distribution within the body. This understanding, foundational to diagnosis and treatment, is made possible through an intricate array of sophisticated imaging modalities and high-resolution camera systems, forming the bedrock of modern medical insight.

The journey from a vague clinical suspicion to a definitive diagnosis of conditions like B-cell lymphoma is heavily reliant on technological prowess in capturing, processing, and interpreting visual data. From the minute cellular architecture revealed by advanced microscopy to the macroscopic landscape mapped by medical imaging scans, cameras and imaging systems are not merely tools but essential lenses through which the invisible becomes visible, the abstract becomes concrete, and ultimately, the unknown becomes understood. This exploration delves into the pivotal role of these technologies, illustrating how they collaboratively answer the fundamental “what is it?” question concerning intricate biological challenges.
The Imperative Role of Advanced Imaging in Biological Diagnostics
The initial step in dissecting any biological anomaly, including complex conditions like B-cell lymphoma, is visualization. Traditional methods, while foundational, have been significantly augmented by a suite of advanced imaging technologies that provide unprecedented levels of detail and insight, bridging the gap between clinical observation and cellular reality. These technologies form the bedrock upon which diagnostic clarity is built, enabling clinicians and researchers to “see” what was once beyond human perception.
Visualizing the Microscopic: From Light Microscopy to Electron Tomography
At the cellular level, the journey of discovery begins with microscopy. Conventional light microscopes, equipped with high-fidelity digital cameras, allow pathologists to examine stained tissue samples (biopsies) for morphological changes characteristic of cancerous cells. These digital cameras, often featuring high pixel counts and dynamic range, capture intricate details of cell nuclei, cytoplasm, and overall tissue architecture. For instance, abnormal B lymphocytes in lymphoma often exhibit specific nuclear patterns or cellular enlargement that can be identified and documented with digital pathology systems.
Beyond light microscopy, electron microscopy (EM) pushes the boundaries of resolution, employing beams of electrons instead of light to visualize subcellular structures with astounding detail. Transmission electron microscopy (TEM) can reveal the ultrastructure of B-cells, including mitochondrial abnormalities or ribosomal densities, which can be critical for subtyping certain lymphomas. Scanning electron microscopy (SEM) provides three-dimensional surface views of cells and tissues. Integrating these EM systems with specialized cameras and detectors capable of sensing electron signals allows for the capture of images at nanometer resolution, offering insights into the minute components that define cellular health or disease. Electron tomography, an advanced EM technique, allows for the reconstruction of 3D cellular volumes, offering a comprehensive spatial understanding of cellular pathology relevant to B-cell lymphoma.
Non-Invasive Insights: The Power of Radiography and CT Scans
Moving to a broader anatomical scale, radiography (X-rays) and Computed Tomography (CT) scans offer non-invasive methods to assess the presence and extent of disease within the body. While not directly visualizing B-cells, these techniques are crucial for identifying enlarged lymph nodes, organ involvement (spleen, liver), or bone lesions often associated with B-cell lymphoma. Modern CT scanners utilize sophisticated X-ray detectors and fast-processing cameras that capture multiple cross-sectional images in rapid succession. These images are then computationally reconstructed into detailed 3D anatomical views. The quality of these reconstructions hinges on the sensitivity and speed of the detectors and the algorithms used for image processing, providing high-resolution anatomical maps that guide further diagnostic steps and treatment planning. The ability to precisely localize disease burden is a critical answer to “what is it?” in terms of its physical manifestation and spread.
Specialized Imaging Techniques for Cellular and Molecular Pathology
While anatomical imaging provides localization, a deeper understanding of cellular and molecular pathology requires more specialized techniques that can differentiate tissues based on their intrinsic properties, metabolic activity, or specific molecular markers. These advanced modalities leverage distinct physical principles and sophisticated detector systems to paint a more nuanced picture of disease.
Magnetic Resonance Imaging (MRI): Delving into Soft Tissue Anomalies
Magnetic Resonance Imaging (MRI) stands out for its exceptional ability to visualize soft tissues without ionizing radiation. MRI scanners use powerful magnetic fields and radio waves to generate detailed images of organs, soft tissues, bone, and virtually all other internal body structures. The signals emitted by water molecules in different tissues are detected by highly sensitive radiofrequency coils (acting as specialized “cameras”), which then transmit this data to powerful computers for image reconstruction. The contrast between normal and diseased tissue (e.g., enlarged lymph nodes or lesions in the brain or spinal cord in cases of CNS lymphoma) is often superior with MRI compared to CT. Different MRI sequences (T1-weighted, T2-weighted, FLAIR, DWI) act like different “filters” or “lenses,” highlighting distinct tissue characteristics, providing a multi-parametric view of the disease. This detailed soft tissue contrast is invaluable for defining the extent of disease and monitoring treatment response, offering critical insights into the physical nature of the condition.
Positron Emission Tomography (PET) Scans: Unveiling Metabolic Activity
PET scans offer a functional perspective, revealing the metabolic activity of cells rather than just their structure. This is particularly crucial for aggressive cancers like many B-cell lymphomas, which often exhibit high metabolic rates. A small amount of a radioactive tracer, typically a glucose analog like FDG, is injected into the patient. Cancer cells, being highly metabolically active, tend to absorb more of this tracer. PET scanners feature an array of detectors (often scintillation crystals coupled with photomultiplier tubes or silicon photomultipliers) that detect the gamma rays emitted when the tracer decays. These detectors effectively “image” the distribution of metabolic activity throughout the body. The resulting images, often fused with CT scans (PET-CT) for anatomical correlation, highlight metabolically active areas, indicating sites of disease activity, even before significant structural changes occur. This functional imaging capability is an indispensable tool for staging, assessing treatment response, and detecting recurrence, providing an answer to “what is it?” in terms of its biological aggression and vitality.
Fluorescence and Confocal Microscopy: Precision at the Cellular Level
For highly precise cellular characterization, especially in research or detailed diagnostic work on biopsy samples, fluorescence and confocal microscopy are paramount. These techniques utilize specialized cameras – often high-quantum efficiency CCD or sCMOS cameras – to capture light emitted from fluorescent probes attached to specific cellular components or molecular markers. In the context of B-cell lymphoma, these probes can target specific surface proteins (e.g., CD20, CD45) that help identify B-cells and differentiate cancerous B-cells from healthy ones. Confocal microscopes enhance this by using pinholes to eliminate out-of-focus light, producing sharp optical sections that can be stacked to create high-resolution 3D reconstructions of cells and tissue architecture. This provides an exquisite level of detail regarding cellular morphology, protein expression patterns, and the spatial relationships within a tissue sample, offering definitive answers at the molecular level of “what is it?”.
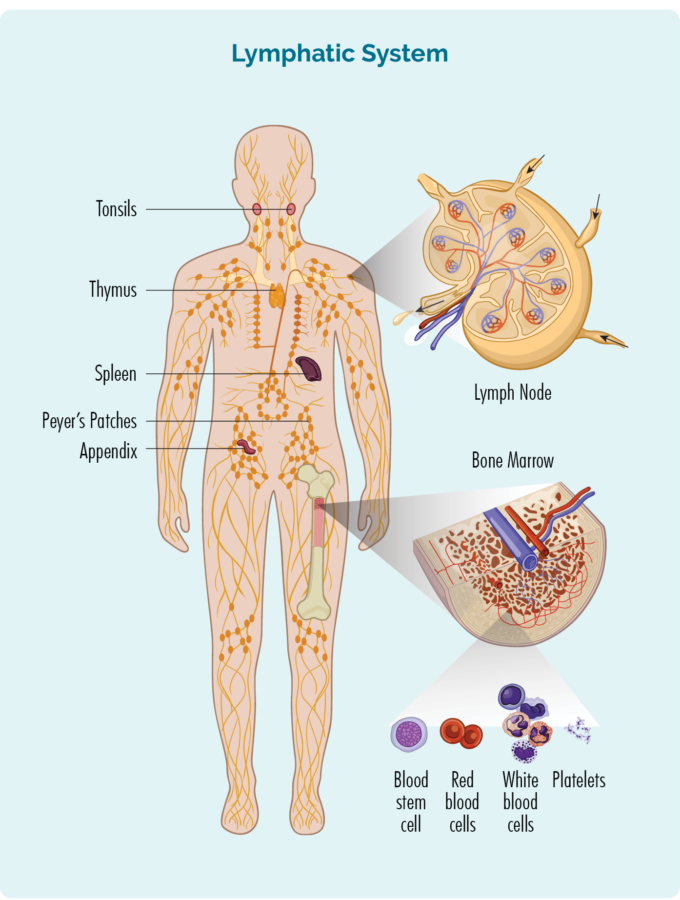
High-Resolution Cameras and Sensors: The Foundation of Diagnostic Clarity
The quality of every image, whether microscopic or macroscopic, ultimately depends on the fidelity of the camera and sensor technology employed. These components are the unsung heroes of diagnostic imaging, converting physical signals into digital data that clinicians and AI systems can interpret.
Evolving Camera Technologies in Medical Devices
The evolution of cameras in medical devices has been transformative. Early analog systems have given way to sophisticated digital cameras characterized by high resolution, increased sensitivity, and faster frame rates. In endoscopy, miniature high-definition (HD) and even 4K cameras provide incredibly detailed views of internal organs, crucial for visual inspection and guided biopsies. In pathology, digital slide scanners equipped with multi-megapixel cameras can digitize entire microscope slides at high magnification, enabling remote consultation and advanced image analysis. These cameras feature advanced sensor designs (e.g., backside-illuminated CMOS) and sophisticated optics to minimize noise and maximize light capture, ensuring that even subtle cellular abnormalities are faithfully recorded. The constant drive for higher resolution and better color accuracy directly translates to improved diagnostic potential.
The Significance of Sensor Sensitivity and Image Fidelity
Sensor sensitivity is a critical parameter in medical imaging, particularly for techniques that involve low light levels (e.g., fluorescence microscopy) or rapid signal acquisition (e.g., dynamic CT or MRI sequences). High-sensitivity sensors, often coupled with advanced noise reduction algorithms, allow for clearer images with less signal, reducing exposure times or the amount of contrast agent needed. Image fidelity, encompassing factors like signal-to-noise ratio, dynamic range, and spatial resolution, ensures that the captured image accurately represents the underlying biological reality. Manufacturers of medical imaging equipment continuously innovate in sensor design, pixel architecture, and data conversion to push these boundaries, ensuring that every pixel contributes meaningful diagnostic information. Without this foundational clarity, differentiating normal tissue from a cancerous lesion like B-cell lymphoma would be significantly more challenging.
AI and Computational Imaging: Enhancing Diagnostic Accuracy and Efficiency
The sheer volume and complexity of images generated by modern medical imaging systems necessitate advanced computational approaches, particularly Artificial Intelligence (AI) and machine learning, to extract meaningful insights and enhance diagnostic accuracy and efficiency.
Automated Image Analysis for Pathological Feature Identification
AI, especially deep learning algorithms, is revolutionizing image analysis in pathology and radiology. For conditions like B-cell lymphoma, AI can be trained on vast datasets of annotated microscopic images to automatically identify cancerous cells, classify their subtypes, and even predict prognosis based on subtle features imperceptible to the human eye. In radiology, AI algorithms can quickly scan CT or PET scans to highlight suspicious lymph nodes, measure lesion sizes, and track changes over time, improving the consistency and speed of interpretation. These AI systems act as highly sophisticated, tireless “virtual assistants” to human experts, augmenting their ability to answer “what is it?” with greater precision and consistency. The integration of AI with high-resolution camera data transforms raw visual information into actionable diagnostic intelligence.
Integrating Multi-Modal Imaging Data for Comprehensive Assessment
One of the most powerful applications of computational imaging is the integration and fusion of data from multiple imaging modalities. By combining the anatomical precision of CT or MRI with the functional insights of PET, or by correlating microscopic details with macroscopic scans, a holistic view of the disease can be constructed. AI algorithms are particularly adept at performing this multi-modal fusion, identifying subtle correlations and patterns across different data types that might be missed by manual review. This integrated assessment provides a more comprehensive answer to “what is b cell lymphoma cancer” by presenting its structural, metabolic, and cellular characteristics in a unified framework, guiding personalized treatment strategies.
Future Directions in Medical Imaging for Complex Conditions
The relentless pace of innovation in cameras and imaging technology continues to open new frontiers in understanding and combating diseases like B-cell lymphoma. The future promises even greater resolution, real-time capabilities, and more accessible diagnostic tools.
Ultra-High Resolution Systems and Real-time Visualization
The quest for higher resolution will continue, with the development of ultra-high-resolution microscopy systems that can visualize molecular interactions in living cells, potentially offering real-time insights into cellular processes relevant to cancer initiation and progression. In macroscopic imaging, faster and more detailed dynamic imaging sequences will allow for real-time monitoring of physiological processes, such as blood flow or drug distribution, providing immediate feedback on disease activity and treatment effectiveness. This will involve breakthroughs in sensor technology, data transfer rates, and computational power, pushing the boundaries of what can be captured and processed in real-time.
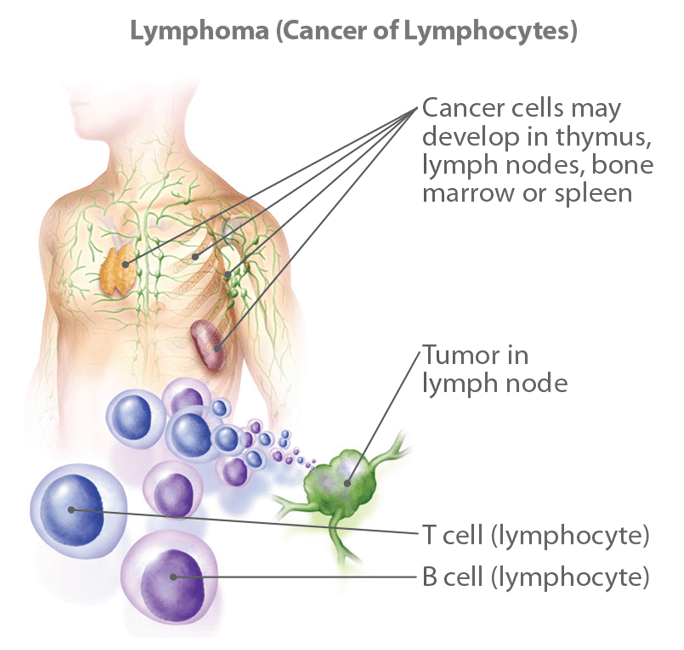
Miniaturization and Point-of-Care Imaging Solutions
Another significant trend is the miniaturization of imaging technology. Developments in micro-cameras and compact sensor arrays are paving the way for point-of-care diagnostic tools. Imagine handheld devices that can perform rapid optical biopsies, or ingestible cameras that provide highly detailed views of the gastrointestinal tract with minimal invasiveness. These portable solutions would democratize access to advanced diagnostics, enabling earlier detection and monitoring of conditions in diverse settings. The ability to bring sophisticated imaging capabilities directly to the patient, rather than requiring them to visit large imaging centers, represents a revolutionary step in making the answer to “what is it?” more readily available.
In conclusion, the question “what is b cell lymphoma cancer” is intricately tied to the capabilities of modern cameras and imaging technologies. These systems, from the microscopic precision of electron tomography to the comprehensive overview provided by PET-CT and MRI, are the indispensable tools that allow us to visualize, characterize, and ultimately understand complex biological conditions. Coupled with the transformative power of AI and computational analysis, these technologies continue to evolve, promising even more insightful and accessible diagnostics in the future, continually refining our answer to the fundamental questions of health and disease.
