The fundamental building blocks of everything we see and interact with are atoms. They are the smallest units of an element that retain the chemical properties of that element. Imagine them as the individual LEGO bricks of the universe. Molecules, on the other hand, are formed when two or more atoms bond together. They are like the structures you build with those LEGO bricks. While the terms “atom” and “molecule” are often used interchangeably in casual conversation, they represent distinct levels of matter and play crucial roles in understanding chemistry and physics.
The Atom: The Undivided Core
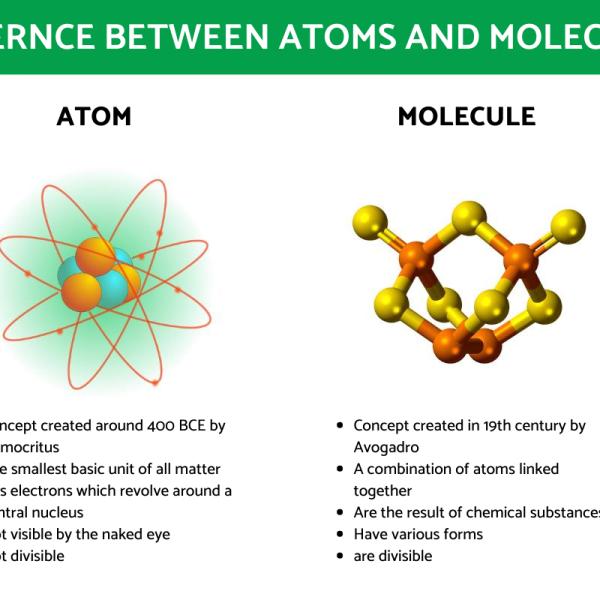
At the heart of all matter lies the atom. The word “atom” itself comes from the Greek word “atomos,” meaning “indivisible.” While we now know that atoms are composed of even smaller subatomic particles, they are the smallest particles that can exist independently and still exhibit the properties of a chemical element.
Structure of an Atom
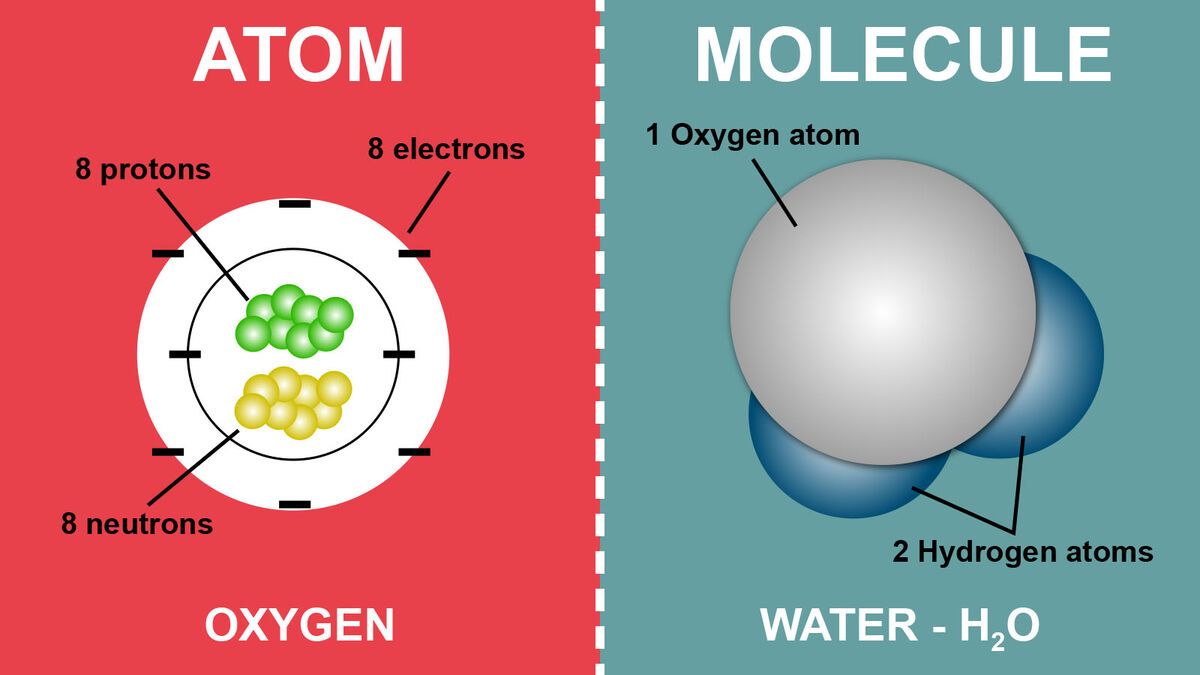
Every atom consists of a central nucleus, which contains positively charged protons and neutrally charged neutrons. Surrounding the nucleus are negatively charged electrons that orbit the nucleus in specific energy levels or shells. The number of protons in an atom’s nucleus determines which element it is. For instance, an atom with one proton is always hydrogen, an atom with six protons is carbon, and an atom with eight protons is oxygen. This number is known as the atomic number.
The number of neutrons in the nucleus can vary for a given element, leading to different isotopes. While isotopes of an element have the same chemical properties (due to the same number of protons and electrons), they can differ in mass and nuclear stability. The electrons are crucial for chemical bonding. Their arrangement and number in the outermost shell dictate how an atom will interact with other atoms.
Elements and Their Properties
The periodic table is a systematic organization of all known elements, arranged by their atomic number and electron configurations. Each element possesses unique physical and chemical properties, such as melting point, boiling point, reactivity, and conductivity. These properties are a direct consequence of the atom’s structure, particularly the number of protons and the electron configuration. For example, elements in the same group (column) of the periodic table tend to have similar chemical behaviors because they have the same number of valence electrons – the electrons in the outermost shell, which are involved in bonding.
Atoms can exist independently as noble gases, which are very stable due to having a full outer electron shell. However, most atoms are not stable on their own and tend to interact with other atoms to achieve a more stable electron configuration. This drive for stability is the fundamental impetus behind chemical reactions and the formation of molecules.
The Molecule: Atoms United
When atoms join together through chemical bonds, they form molecules. These bonds are essentially attractive forces that hold the atoms in a specific arrangement. A molecule is the smallest electrically neutral unit of a chemical compound that can exist independently. Just as a word is formed by combining letters, a molecule is formed by combining atoms.
Types of Chemical Bonds
The nature of the chemical bond dictates the properties of the resulting molecule. The two primary types of chemical bonds are:
-
Covalent Bonds: In a covalent bond, atoms share electrons to achieve a stable electron configuration. This sharing can be equal (nonpolar covalent bond) or unequal (polar covalent bond), depending on the electronegativity of the atoms involved. Water (H₂O) is a classic example of a molecule with polar covalent bonds, where oxygen shares electrons with two hydrogen atoms. The unequal sharing creates a slight positive charge on the hydrogen atoms and a slight negative charge on the oxygen atom.
-
Ionic Bonds: Ionic bonds form when one atom completely transfers one or more electrons to another atom. This transfer results in the formation of charged particles called ions – a positively charged cation and a negatively charged anion. These oppositely charged ions are then attracted to each other, forming an ionic compound. Sodium chloride (NaCl), common table salt, is an example of an ionic compound. Sodium (Na) loses an electron to become a positive ion (Na⁺), and chlorine (Cl) gains that electron to become a negative ion (Cl⁻).
Other types of bonds, like metallic bonds (found in metals where electrons are delocalized) and hydrogen bonds (a weaker attraction between molecules), also play significant roles in the behavior of matter.
Molecular Structures and Properties
The specific arrangement of atoms within a molecule, known as its molecular structure, is critical to its properties. Even molecules with the same number and type of atoms can have different properties if their atoms are arranged differently. These are called isomers. For example, ethanol (drinking alcohol) and dimethyl ether have the same chemical formula (C₂H₆O) but different structures and thus very different properties. Ethanol is a liquid at room temperature and is a common solvent and intoxicating agent, while dimethyl ether is a gas at room temperature and is used as an aerosol propellant.
The size, shape, and polarity of molecules influence their interactions with other molecules, leading to macroscopic properties like boiling point, melting point, solubility, and viscosity. For instance, water molecules are polar and form strong hydrogen bonds with each other, which is why water has a relatively high boiling point and surface tension compared to nonpolar molecules of similar size.
Key Differences Summarized
Understanding the distinctions between atoms and molecules is fundamental to grasping chemical concepts. Here’s a concise breakdown:
- Composition: Atoms are the fundamental, indivisible units of an element. Molecules are formed by the chemical bonding of two or more atoms.
- Identity: An atom’s identity is determined by its number of protons. A molecule’s identity is determined by the types and number of atoms it contains and how they are bonded.
- Independence: While some atoms (like noble gases) can exist independently, many atoms are reactive and seek to bond. Molecules, being stable combinations of atoms, can exist independently.
- Smallest Unit: The atom is the smallest unit of an element. The molecule is the smallest unit of a chemical compound that retains its chemical properties.
- Bonding: Atoms are the constituents that form bonds. Molecules are the result of these bonds.
Examples Illustrating the Difference
To solidify the understanding, let’s consider some common examples:
- Oxygen (O₂): While we often refer to “breathing oxygen,” the oxygen we breathe is actually a molecule. It consists of two oxygen atoms covalently bonded together. An individual oxygen atom (O) is highly reactive and not stable on its own.
- Water (H₂O): A water molecule is composed of two hydrogen atoms and one oxygen atom, all bonded covalently. A single hydrogen atom or a single oxygen atom cannot independently exhibit the properties of water.
- Carbon Dioxide (CO₂): This molecule consists of one carbon atom double-bonded to two oxygen atoms.
- Iron (Fe): A piece of iron is made up of a vast collection of iron atoms. In its solid metallic form, these atoms are arranged in a crystal lattice, but they are not considered a discrete molecule in the same way as water or CO₂.
The Significance in Science and Technology
The distinction between atoms and molecules is not merely an academic exercise; it underpins vast areas of science and technology.
Chemistry and Material Science
In chemistry, the study of how atoms interact to form molecules is central. Understanding molecular structures and bonding allows chemists to predict and design new substances with specific properties. This knowledge is crucial for developing new medicines, materials, fuels, and countless other products. Material scientists leverage this understanding to create advanced materials with enhanced strength, conductivity, or flexibility by controlling the atomic and molecular arrangements.
Biology
Life itself is a testament to the power of molecular interactions. DNA, proteins, carbohydrates, and lipids – the very building blocks of life – are all complex molecules. Understanding how these molecules are formed, how they interact, and how they change (chemical reactions) is the essence of biology and biochemistry. Processes like photosynthesis, cellular respiration, and metabolism are all intricate molecular dance.
Nanotechnology
At the very frontier of science, nanotechnology deals with manipulating matter at the atomic and molecular scale. Scientists and engineers are learning to build structures and devices atom by atom and molecule by molecule, leading to revolutionary advancements in areas like medicine, electronics, and energy. The ability to control individual atoms and molecules opens up possibilities that were once the realm of science fiction.
In conclusion, atoms are the fundamental units of elements, while molecules are formed when these atoms chemically bond together. This hierarchical organization, from the indivisible atom to the complex molecule, is the foundation upon which the entire material world is built, driving innovation and shaping our understanding of the universe.
