Immunomodulators represent a fascinating and rapidly evolving class of therapeutic agents that hold immense potential for treating a wide spectrum of diseases. Unlike traditional drugs that directly target a specific pathogen or biochemical pathway, immunomodulators work by influencing the body’s own immune system, either by suppressing an overactive immune response or by enhancing a weakened one. This nuanced approach allows for the treatment of conditions where the immune system itself is the culprit, such as autoimmune diseases, or where a compromised immune system leaves the body vulnerable to infections or cancer.
The term “immunomodulator” is broad, encompassing a diverse range of substances and strategies. At their core, these agents aim to restore balance to the immune system, a complex network of cells, tissues, and organs that work together to defend the body against foreign invaders like bacteria, viruses, and parasites. When this intricate system malfunctions, it can lead to a variety of health problems, making immunomodulators a critical area of medical research and development. Understanding the mechanisms, applications, and future potential of these powerful agents is crucial for appreciating their significance in modern medicine.

Understanding the Immune System: A Foundation for Immunomodulation
Before delving into the specifics of immunomodulators, it’s essential to have a fundamental understanding of how the immune system operates. This intricate biological defense mechanism is a marvel of evolutionary design, capable of distinguishing between self and non-self, and mounting a targeted response to threats while maintaining tolerance to the body’s own tissues. The immune system can be broadly divided into two interconnected branches: the innate immune system and the adaptive immune system.
The Innate Immune System: The First Line of Defense
The innate immune system is the body’s immediate, non-specific defense mechanism. It’s always on standby and reacts rapidly to a wide range of pathogens and cellular damage. Key components of the innate immune system include physical barriers like the skin and mucous membranes, as well as cellular players such as phagocytes (like macrophages and neutrophils) that engulf and destroy foreign particles, and natural killer (NK) cells that can kill infected or cancerous cells. It also involves inflammatory responses, characterized by redness, swelling, heat, and pain, which help to isolate and eliminate pathogens. The innate system doesn’t have “memory” in the same way the adaptive system does, meaning it responds to threats in a similar fashion each time.
The Adaptive Immune System: The Targeted and Remembered Response
In contrast, the adaptive immune system is highly specific and develops over time in response to exposure to particular antigens – molecules found on pathogens or foreign substances. This system has a remarkable ability to “remember” past encounters, leading to a faster and more potent response upon subsequent exposures. The central players in the adaptive immune system are lymphocytes, specifically B cells and T cells.
B cells are responsible for producing antibodies, proteins that bind to specific antigens, marking them for destruction or neutralizing their effects. T cells, on the other hand, have diverse roles. Helper T cells (CD4+) orchestrate the immune response by activating other immune cells. Cytotoxic T cells (CD8+) directly kill infected or cancerous cells. Regulatory T cells (Tregs) play a crucial role in preventing the immune system from attacking the body’s own tissues, thereby maintaining self-tolerance. The intricate interplay between these cells and their signaling molecules, known as cytokines, forms the basis of effective immune surveillance and protection.
Mechanisms of Action: How Immunomodulators Work
Immunomodulators achieve their therapeutic effects by precisely influencing the complex machinery of the immune system. Their mechanisms of action are diverse and depend on the specific agent and the condition being treated. Broadly, these agents can either dampen an overactive immune response or boost a deficient one, thereby restoring immune homeostasis.
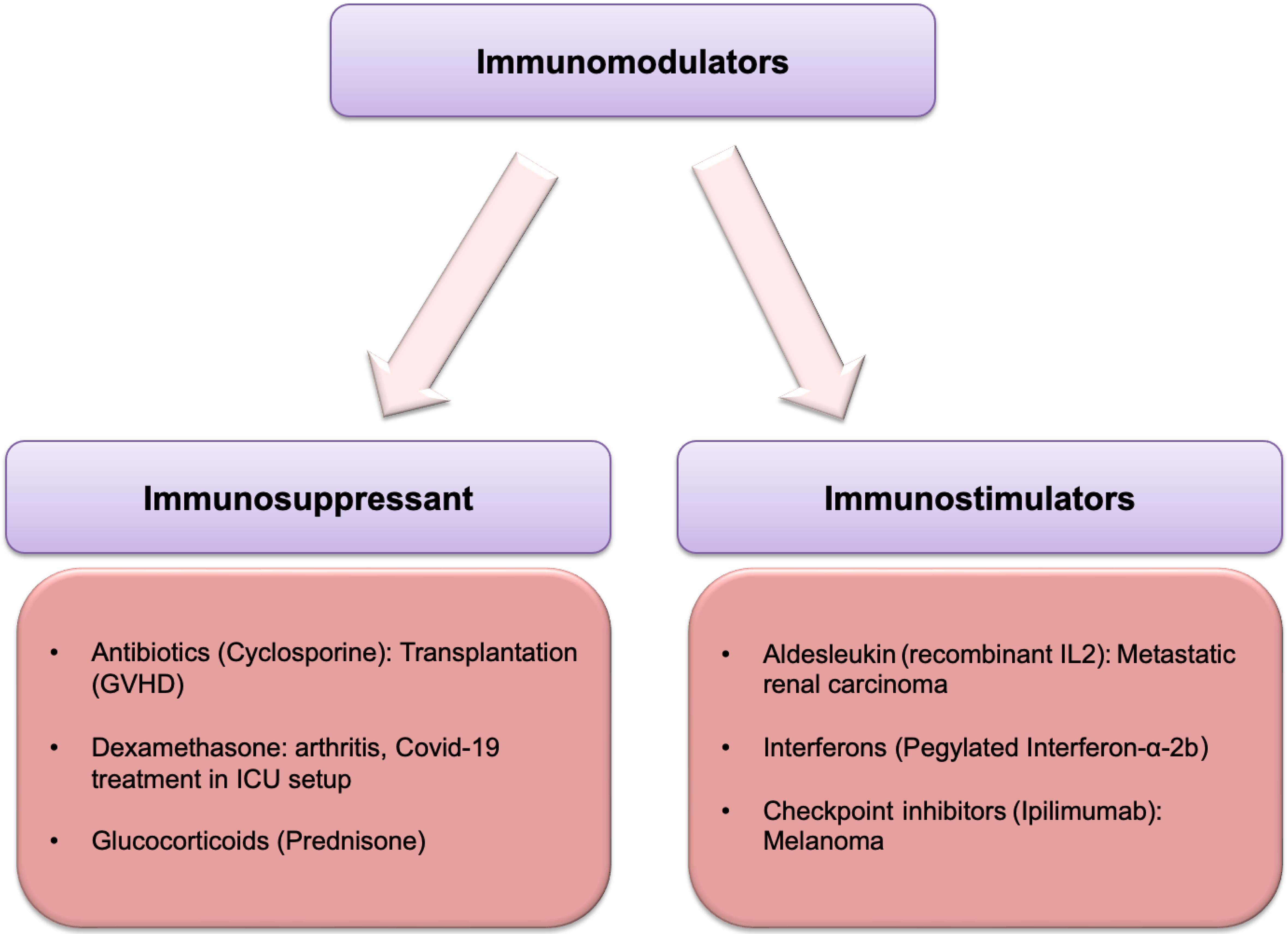
Immunosuppression: Calming an Overactive Immune System
In conditions where the immune system mistakenly attacks the body’s own tissues, such as in autoimmune diseases like rheumatoid arthritis, lupus, or multiple sclerosis, immunosuppressive immunomodulators are employed. These agents work by dampening the activity of immune cells that are responsible for causing damage. For instance, some immunosuppressants interfere with the proliferation of lymphocytes, reducing the number of cells that can mount an attack. Others target specific signaling pathways within immune cells, preventing them from releasing inflammatory cytokines or from becoming activated.
Corticosteroids, for example, are potent immunosuppressants that broadly inhibit the inflammatory response and reduce the activity of many immune cells. More targeted therapies include biologics that block specific cytokines, such as tumor necrosis factor-alpha (TNF-α) or interleukins, which are key drivers of inflammation in many autoimmune diseases. Another approach involves depleting specific types of immune cells, such as B cells or T cells, that are implicated in the disease process. The goal of immunosuppression is not to abolish the immune system entirely, but rather to reduce its harmful activity to a level where it no longer causes significant damage to the body.
Immunostimulation: Bolstering a Weakened Immune System
Conversely, in situations where the immune system is compromised, such as in certain immunodeficiency disorders, during chemotherapy-induced immunosuppression, or in some types of cancer, immunostimulatory immunomodulators can be beneficial. These agents aim to enhance the body’s ability to fight off infections or cancer. This can be achieved through various mechanisms, such as promoting the proliferation and maturation of immune cells, enhancing the activity of existing immune cells, or improving their ability to recognize and attack targets.

Examples of immunostimulatory agents include interferons, a group of naturally occurring proteins that help the immune system fight viral infections and some cancers. Colony-stimulating factors (CSFs) are another class of drugs that stimulate the production of white blood cells, which is particularly useful for patients undergoing chemotherapy, as it helps to restore their immune defenses more quickly. Cytokines themselves, when administered therapeutically, can act as immunostimulants. Research is also actively exploring therapies that enhance the anti-tumor immune response, such as checkpoint inhibitors that release the brakes on T cells, allowing them to more effectively target cancer cells.
Applications in Disease Management: From Autoimmunity to Cancer
The ability of immunomodulators to fine-tune the immune system makes them invaluable tools for managing a wide range of challenging diseases. Their application spans from chronic inflammatory conditions to life-threatening cancers, offering new hope and improved outcomes for patients. The precise application depends on whether the immune system is overactive or underactive, and on the specific disease pathology.
Treating Autoimmune Diseases and Inflammatory Disorders
Autoimmune diseases are characterized by the immune system attacking the body’s own healthy tissues. Conditions like rheumatoid arthritis, where the immune system attacks the joints; type 1 diabetes, where it attacks insulin-producing cells in the pancreas; and inflammatory bowel disease (IBD), which includes Crohn’s disease and ulcerative colitis, are prime examples. Immunomodulators, primarily immunosuppressive agents, are central to their management. They aim to reduce the chronic inflammation and tissue damage caused by the misguided immune response, thereby alleviating symptoms and preventing disease progression. Biologic therapies targeting specific inflammatory pathways have revolutionized the treatment of many of these conditions, offering more targeted and often more effective treatments than older, broader immunosuppressants.
The Role in Cancer Therapy and Prevention
Immunomodulators are increasingly playing a pivotal role in oncology. The immune system’s ability to recognize and eliminate abnormal cells makes it a natural defense against cancer. However, cancer cells can evolve mechanisms to evade immune detection and destruction. Immunomodulatory therapies in cancer treatment aim to re-engage the immune system’s power against tumors. Immunotherapy, a broad term that includes various immunomodulatory strategies, has shown remarkable success in treating several types of cancer, including melanoma, lung cancer, and certain leukemias and lymphomas. This includes checkpoint inhibitors that unleash T cells to attack cancer cells, and adoptive cell therapies where immune cells are collected, enhanced, and reinfused into the patient. Beyond treatment, research is also exploring the potential of immunomodulators in cancer prevention, by bolstering the immune system’s surveillance capabilities.
Addressing Immunodeficiency and Infectious Diseases
In cases of immunodeficiency, where the immune system is not functioning effectively, immunostimulatory immunomodulators are vital. This can include congenital immunodeficiencies, where individuals are born with a weakened immune system, or acquired immunodeficiencies, such as those caused by HIV infection or treatments like chemotherapy. Immunostimulants can help to boost the production and function of immune cells, making individuals less susceptible to opportunistic infections. Furthermore, certain immunomodulators can be used to enhance the immune response to vaccines or to combat chronic viral infections where the immune system struggles to clear the pathogen. The strategic use of these agents aims to restore or augment the body’s natural defenses, improving quality of life and prognosis.
The Future of Immunomodulation: Precision and Personalization
The field of immunomodulation is continuously advancing, driven by a deeper understanding of immune system complexity and the development of innovative therapeutic approaches. The future promises more precise, personalized, and effective treatments that harness the power of the immune system with greater accuracy and fewer side effects.
Precision Medicine and Targeted Therapies
The trend towards precision medicine is profoundly impacting immunomodulation. Instead of broad-acting immunosuppressants, the focus is shifting towards therapies that target specific immune cells, signaling pathways, or inflammatory mediators implicated in a particular disease in an individual patient. Genetic profiling and advanced diagnostics will enable clinicians to identify the precise immune dysregulation at play, allowing for the selection of the most appropriate immunomodulatory agent. This personalized approach aims to maximize therapeutic efficacy while minimizing off-target effects and toxicity, leading to better patient outcomes and improved quality of life.
Novel Delivery Systems and Combination Therapies
Innovations in drug delivery systems are also enhancing the potential of immunomodulators. Researchers are exploring novel ways to deliver these agents directly to the site of disease, reducing systemic exposure and side effects. This could include nano-delivery systems that encapsulate drugs and release them gradually or target specific immune cells. Furthermore, combination therapies, where different immunomodulatory agents are used together, or in conjunction with other treatment modalities like chemotherapy or radiation, are showing promising results. The synergistic effects of such combinations can overcome resistance mechanisms and achieve more robust therapeutic responses, particularly in complex diseases like cancer.
The Expanding Frontier: Microbiome and Beyond
Emerging research is uncovering fascinating connections between the gut microbiome and immune system function. The trillions of microorganisms residing in our gut play a significant role in shaping and educating our immune system. Future immunomodulatory strategies may involve manipulating the gut microbiome through probiotics, prebiotics, or fecal microbiota transplantation to influence immune responses beneficially. Beyond the microbiome, ongoing research is exploring new frontiers in understanding immune cell plasticity, epigenetic regulation of immune responses, and the development of novel cell-based therapies, all of which hold the potential to further expand the scope and effectiveness of immunomodulators in the years to come.
