Giardiasis, a common intestinal illness caused by the parasite Giardia duodenalis (also known as Giardia lamblia or Giardia intestinalis), can cause significant discomfort and gastrointestinal distress. Prompt and accurate diagnosis is crucial for effective treatment and preventing further spread. A range of diagnostic tests are available, each with its own strengths and limitations, designed to detect the presence of Giardia in stool or other biological samples. This article will delve into the various testing methodologies employed for giardiasis, exploring their principles, applications, and the considerations that guide their selection in clinical practice.
Microscopic Examination of Stool
One of the foundational methods for diagnosing giardiasis involves the direct examination of stool samples under a microscope. This technique, while sometimes labor-intensive and dependent on the skill of the microscopist, remains a widely accessible and often cost-effective diagnostic tool.
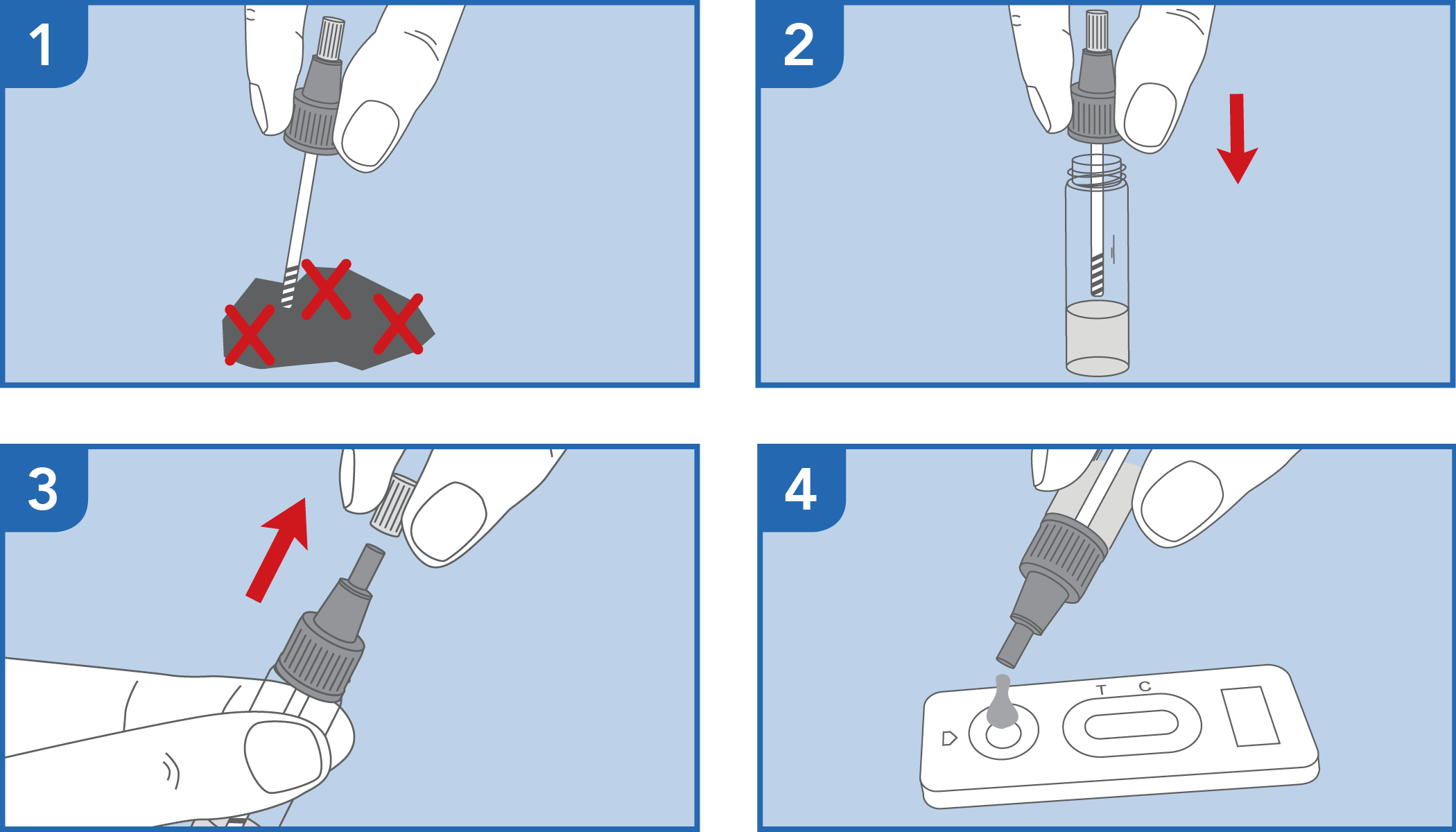
Direct Wet Mounts
In a direct wet mount, a small amount of fresh stool is mixed with a saline solution or a staining agent on a glass slide, covered with a coverslip, and examined under a microscope. This method aims to visualize the characteristic pear-shaped trophozoites (the motile form of the parasite) or the oval-shaped cysts (the infective, environmentally resistant form). Trophozoites are typically seen actively moving, with flagella and a distinctive “falling leaf” motility. Cysts, on the other hand, are more commonly found in formed stools and are characterized by their tough outer wall and the presence of internal structures, such as median bodies and flagella.
Limitations: The sensitivity of direct wet mounts can be variable. Giardia parasites may not be uniformly distributed throughout the stool sample, leading to false-negative results, especially in individuals with low parasite burdens. The presence of other fecal material can also obscure the parasites, making identification challenging.
Stained Smears
To improve visualization and aid in identification, stool samples can be stained. Common stains used in giardiasis diagnosis include:
-
Iodine Stains: Iodine preparations, such as Lugol’s iodine, are used to enhance the visibility of cyst structures. Iodine stains the glycogen reserves within the cysts a brown color, making their outlines and internal components more apparent. However, iodine does not stain trophozoites effectively.
-
Trichrome Stains: The modified trichrome stain is a more sensitive and specific staining method that stains both trophozoites and cysts. Trophozoites typically appear as bluish-green structures with darker blue nuclei and internal organelles. Cysts stain a purplish-red color. Trichrome staining allows for better differentiation of Giardia from other protozoa and fecal debris.
-
Acid-Fast Stains: While not the primary stain for Giardia, acid-fast stains can sometimes be used, particularly in conjunction with other diagnostic methods. Giardia cysts may appear as round or oval structures with a characteristic pinkish hue.
Advantages of Staining: Staining techniques generally increase the sensitivity and specificity of microscopic examination, making it easier to detect even small numbers of parasites and differentiate them from other microorganisms.
Antigen Detection Tests
Antigen detection tests offer a more rapid and often more sensitive alternative to microscopic examination for detecting Giardia infection. These assays are designed to identify specific proteins (antigens) produced by the parasite within a stool sample.
Enzyme Immunoassays (EIAs)
Enzyme immunoassays, also known as enzyme-linked immunosorbent assays (ELISAs), are widely used for Giardia antigen detection. In these tests, antibodies specific to Giardia antigens are immobilized on a solid surface (e.g., a microplate or a dipstick). When a stool sample is added, any Giardia antigens present will bind to these antibodies. A second antibody, linked to an enzyme, is then added. This enzyme-antibody complex binds to the immobilized antigen-antibody complex. Finally, a substrate is added, which the enzyme converts into a detectable signal, such as a color change. The intensity of the color change is usually proportional to the amount of antigen present.
Advantages of EIAs: EIAs are generally more sensitive than direct microscopic examination, can detect infections even when parasite shedding is intermittent, and provide relatively quick results. They are also less dependent on the expertise of the microscopist.
Immunochromatographic Assays (ICAs) or Lateral Flow Assays

Immunochromatographic assays, often referred to as rapid antigen tests or lateral flow assays, are similar in principle to EIAs but are designed for faster, point-of-care testing. These tests typically involve a strip with immobilized antibodies. A processed stool sample is applied to a designated area, and the sample flows along the strip. If Giardia antigens are present, they bind to the antibodies, forming a visible line or band, analogous to a pregnancy test.
Advantages of ICAs: ICAs offer the significant advantage of speed, providing results within minutes. They are user-friendly, require minimal equipment, and are ideal for use in resource-limited settings or for rapid screening.
Considerations for Antigen Tests: While highly sensitive and specific, antigen tests may still produce false-negative results if the parasite shedding is very low or intermittent. Conversely, cross-reactivity with other intestinal pathogens, though generally rare, is a possibility with some assays.
Molecular Diagnostic Methods
Molecular diagnostic techniques, particularly those based on the polymerase chain reaction (PCR), represent the most sensitive and specific methods for detecting Giardia infection. These methods amplify specific DNA sequences unique to Giardia.
Polymerase Chain Reaction (PCR)
PCR assays are designed to detect genetic material (DNA) of Giardia in stool samples. The process involves identifying and isolating Giardia-specific DNA sequences and then using primers to amplify these sequences exponentially. This amplification allows for the detection of even minute amounts of parasite DNA that might be missed by other methods.
Types of PCR: Various PCR formats exist, including:
- Conventional PCR: This is the foundational technique where amplified DNA is typically detected by gel electrophoresis.
- Real-time PCR (qPCR): qPCR allows for the quantification of the amplified DNA in real-time as the reaction progresses, offering greater sensitivity and providing information about the parasite load.
- Multiplex PCR: This advanced technique can simultaneously detect Giardia along with other common enteric pathogens in a single test, making it highly efficient for syndromic testing.
Advantages of PCR: PCR offers unparalleled sensitivity and specificity. It can detect infections at very early stages, even before symptoms become pronounced, and can identify specific genotypes of Giardia, which can be important for epidemiological studies and outbreak investigations. PCR can also be useful in immunocompromised individuals or those with chronic diarrhea where other methods may fail.
Limitations of PCR: PCR is generally more expensive and requires specialized laboratory equipment and trained personnel. The turnaround time for PCR results can be longer than for antigen tests, though advancements in real-time PCR are improving this. Additionally, detecting DNA does not necessarily indicate the presence of viable, infectious parasites, although it is highly indicative of infection.
Serological Testing (Antibody Detection)
Serological tests detect the presence of antibodies produced by the host’s immune system in response to a Giardia infection. These tests are typically performed on blood samples.

Enzyme-Linked Immunosorbent Assay (ELISA) for Antibodies
In ELISA-based serological tests for giardiasis, serum samples are analyzed for the presence of Giardia-specific antibodies, primarily immunoglobulin G (IgG) and immunoglobulin M (IgM). These antibodies are generated by the immune system as it fights the infection. The ELISA format is similar to that used for antigen detection, but instead of detecting parasite antigens, it detects host antibodies that bind to immobilized Giardia antigens.
Applications and Limitations: Serological testing is generally not the preferred method for the diagnosis of acute giardiasis, especially in areas where the parasite is endemic. This is because antibodies can persist in the blood for months or even years after the infection has cleared, making it difficult to distinguish between a current and a past infection. However, serology can be useful in certain situations, such as:
- Epidemiological Surveys: To assess the prevalence of past infections in a population.
- Investigating Chronic or Recurrent Symptoms: When other diagnostic methods have been repeatedly negative, but a strong clinical suspicion of giardiasis remains.
- Diagnosis in Individuals with Intermittent Shedding: In cases where parasite shedding in stool is infrequent.
Key Considerations for Choosing a Test: The selection of the most appropriate diagnostic test for giardiasis depends on several factors:
- Clinical Presentation: The severity and duration of symptoms can influence the choice of test.
- Availability and Cost: The accessibility of different tests and their associated costs are significant considerations, particularly in resource-limited settings.
- Turnaround Time: The urgency of diagnosis and the need for rapid results can favor antigen detection or even molecular methods in some laboratories.
- Sensitivity and Specificity Requirements: In situations where a definitive diagnosis is critical, molecular methods often offer the highest degree of accuracy.
- Patient Population: The likelihood of co-infections or the immune status of the patient may also guide test selection.
In conclusion, a multifaceted approach to diagnosing giardiasis is employed, ranging from traditional microscopic examination to sophisticated molecular techniques. Each testing modality plays a vital role in identifying this common parasitic infection, facilitating timely treatment, and contributing to public health efforts to control its spread. The ongoing advancements in diagnostic technologies continue to enhance our ability to accurately and efficiently detect Giardia, ultimately improving patient outcomes.
