Transamination, at its core, is the fundamental biological process responsible for the transfer of an amino group from one molecule to another. This enzymatic reaction is absolutely critical for the synthesis of amino acids and, by extension, for the intricate metabolic pathways that sustain life. While the term itself might sound complex, understanding its role reveals a fascinating insight into the sophisticated machinery that governs cellular function.
The Enzymatic Dance: Aminotransferases
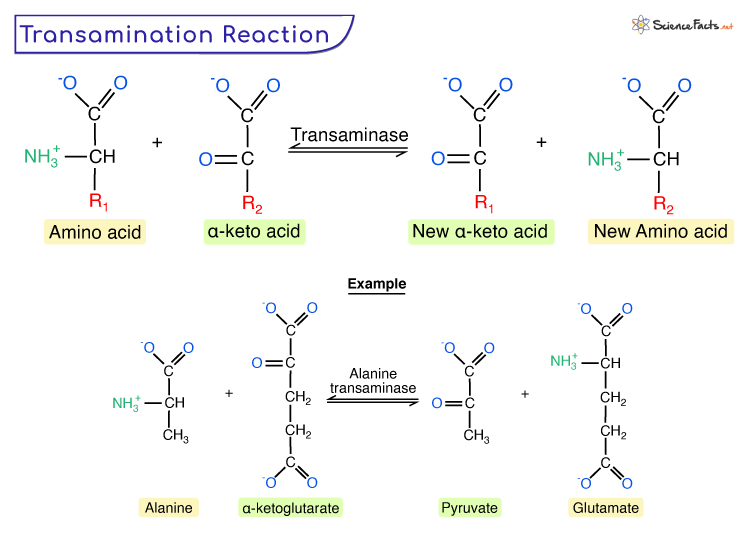
The key players in transamination are a class of enzymes known as aminotransferases, also referred to as transaminases. These enzymes are crucial catalysts, facilitating the reversible transfer of an amino group (–NH₂) from an α-amino acid to an α-keto acid. This seemingly simple exchange is the linchpin for numerous vital biological functions, from the construction of proteins to the generation of energy.
The Catalytic Role of Pyridoxal Phosphate (PLP)
A constant companion to aminotransferases in the transamination process is pyridoxal phosphate (PLP), the active form of vitamin B₆. PLP acts as a coenzyme, a molecule that assists the enzyme in its catalytic activity. Its structure is uniquely suited to participate in the intricate chemical steps involved in amino group transfer.
The mechanism typically begins with the binding of the amino acid substrate to the aminotransferase. PLP, initially bound to the enzyme, undergoes a reversible reaction with the amino acid. This involves the formation of a Schiff base linkage, a transient intermediate where the amino group of the amino acid becomes temporarily attached to the pyridoxal moiety of PLP.
Following the formation of this intermediate, the amino group is cleaved from the original amino acid and transferred to the PLP molecule, forming pyridoxamine phosphate (PMP). Simultaneously, the α-keto acid substrate gains an amino group from the PMP, regenerating PLP and forming a new amino acid. This elegant cyclical process ensures that the enzyme and its coenzyme are ready for the next catalytic round. The versatility of PLP lies in its ability to undergo various tautomerizations and proton transfers, enabling it to accept and then donate the amino group with remarkable efficiency.
Diverse Substrates and Products
The beauty of transamination lies in its broad applicability. A vast array of amino acids can participate as donors, and a corresponding range of α-keto acids can act as acceptors. This allows for the synthesis of virtually all non-essential amino acids, which are amino acids that the body can produce on its own.
For instance, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) are two clinically significant aminotransferases. ALT catalyzes the transfer of an amino group from alanine to α-ketoglutarate, producing pyruvate and glutamate. AST, on the other hand, catalyzes the transfer of an amino group from aspartate to α-ketoglutarate, yielding oxaloacetate and glutamate. The products of these reactions are themselves critical intermediates in central metabolic pathways. Pyruvate is a key player in glycolysis, while oxaloacetate is a central component of the citric acid cycle. Glutamate, formed in both reactions, is a major excitatory neurotransmitter and a crucial intermediate in nitrogen metabolism.
Metabolic Significance of Transamination
Transamination is not merely an isolated enzymatic reaction; it is deeply interwoven into the fabric of cellular metabolism. Its influence extends across energy production, amino acid biosynthesis, and the detoxification of nitrogenous waste.
Amino Acid Biosynthesis and Degradation
One of the most fundamental roles of transamination is in the synthesis of amino acids. While essential amino acids must be obtained from the diet, the body can synthesize all 20 standard amino acids required for protein synthesis through various metabolic pathways, with transamination playing a pivotal role in many of these. For example, the synthesis of glutamate from α-ketoglutarate and ammonia, facilitated by glutamate dehydrogenase, can then be the starting point for the transamination reactions that generate other amino acids.

Conversely, transamination is also involved in the catabolism, or breakdown, of amino acids. When amino acids are not needed for protein synthesis or other anabolic purposes, they can be broken down for energy. The first step in the degradation of most amino acids involves the removal of their amino group. This often occurs via transamination, converting the amino acid into its corresponding α-keto acid. These α-keto acids can then enter the central metabolic pathways, such as the citric acid cycle, to be oxidized for energy production. The amino group removed, typically ending up on glutamate, can then be further processed for excretion.
Nitrogen Metabolism and Urea Cycle
Nitrogen is an essential element for life, primarily incorporated into amino acids and nucleic acids. The management of nitrogen within the body is a complex and tightly regulated process, and transamination is integral to it. The amino groups removed from amino acid catabolism are often channeled into the formation of glutamate and then glutamine. These molecules serve as transport forms of ammonia.
In terrestrial vertebrates, excess nitrogen, primarily in the form of ammonia, is detoxified and excreted as urea. This process occurs in the liver through the urea cycle. Transamination reactions are critical in shuttling nitrogen atoms into the urea cycle. For instance, the glutamate formed during amino acid catabolism can be deaminated to produce ammonia, which then enters the urea cycle. Furthermore, aspartate, which participates in transamination reactions, directly contributes an amino group to the urea cycle.
Interconnection with Central Metabolic Pathways
The α-keto acid products of transamination reactions are not dead ends; they are often key intermediates in central metabolic pathways, most notably the citric acid cycle (also known as the Krebs cycle). As mentioned, pyruvate (from alanine transamination) and oxaloacetate (from aspartate transamination) are direct participants in this cycle. This interconnection means that the fate of amino acids is tightly linked to energy metabolism.
When the body needs energy, amino acids can be degraded, with their carbon skeletons entering the citric acid cycle and ultimately leading to ATP production. Conversely, when the body has an excess of energy and building blocks, intermediates from the citric acid cycle can be used to synthesize amino acids through transamination. This metabolic flexibility allows cells to adapt to varying nutrient availability and physiological demands.
Clinical Relevance of Transamination
The enzymes involved in transamination are not only vital for normal physiological function but also serve as important biomarkers in clinical diagnostics. The measurement of certain aminotransferase levels in the blood can provide valuable insights into the health of various organs, particularly the liver and heart.
Liver Function Tests
Aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are routinely measured as part of liver function tests. These enzymes are abundant in liver cells (hepatocytes). When liver cells are damaged or inflamed due to conditions such as viral hepatitis, alcoholic liver disease, or drug toxicity, they release their intracellular enzymes into the bloodstream. Elevated levels of ALT and AST in the blood are therefore indicative of hepatocellular injury. While both enzymes are found in the liver, ALT is considered more specific to the liver, whereas AST is also found in significant amounts in the heart, skeletal muscle, and kidneys. Therefore, patterns of elevation and the AST/ALT ratio can help clinicians differentiate between various causes of liver damage and assess the extent of injury.
Cardiac Markers
AST also plays a role in cardiac muscle. Following a myocardial infarction (heart attack), cardiac muscle cells are damaged, leading to the release of AST into the bloodstream. While troponins are now the primary and more sensitive biomarkers for diagnosing heart attacks, AST was historically used as a cardiac marker. Its elevation in the blood several hours after a heart attack, peaking within 24-48 hours, provided evidence of cardiac muscle damage. However, due to its presence in other tissues, it is less specific for cardiac injury compared to troponins.
Diagnosis of Other Conditions
Beyond liver and heart health, transaminase levels can sometimes provide clues to other conditions. For example, elevated AST levels can be seen in conditions affecting skeletal muscle, such as muscular dystrophy or rhabdomyolysis (muscle breakdown). In the context of kidney disease, some aminotransferases might be affected, though this is less common as a primary diagnostic marker.
The understanding of transamination, therefore, extends beyond the realm of pure biochemistry. It offers practical applications in medicine, allowing for the detection and monitoring of diseases, contributing to more accurate diagnoses and effective patient management. The subtle enzymatic shifts occurring within our cells, orchestrated by transaminases, have a profound impact on our health and well-being.
