The world around us, from the smallest atom to the grandest galaxy, is composed of matter. Understanding matter requires us to explore its inherent characteristics. These characteristics can be broadly classified into two fundamental categories: physical properties and chemical properties. While both describe the nature of a substance, they do so by examining different aspects of its behavior and composition. This article will delve into the definitions, distinctions, and significance of physical and chemical properties, providing a comprehensive overview for anyone seeking to understand the fundamental building blocks of our universe.
Understanding Physical Properties
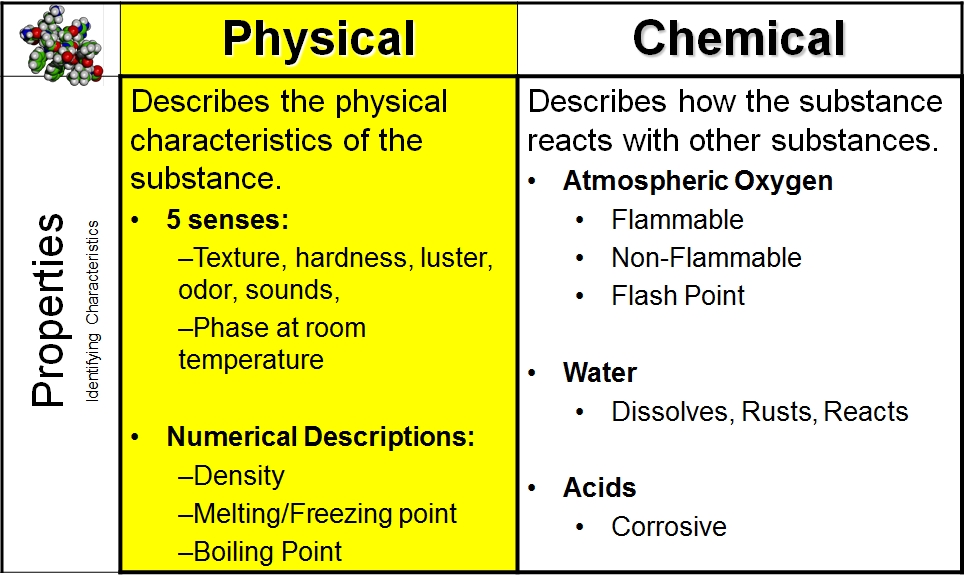
Physical properties are characteristics of a substance that can be observed or measured without changing the substance’s chemical identity. In essence, they describe what a substance is like without altering its fundamental nature. These properties are often reversible and do not involve the formation of new substances. Think of them as the outward appearances and measurable qualities that allow us to identify and differentiate between various materials.

Intrinsic vs. Extrinsic Physical Properties
Physical properties can be further categorized into intrinsic and extrinsic properties.
Intrinsic Physical Properties
Intrinsic physical properties are those that are independent of the amount of substance present. They are characteristic of the substance itself and remain constant regardless of sample size. These are the most useful properties for identifying and classifying substances.
- Melting Point and Boiling Point: The melting point is the temperature at which a solid transitions into a liquid, while the boiling point is the temperature at which a liquid transitions into a gas. For example, water melts at 0°C and boils at 100°C at standard atmospheric pressure. These values are unique to water and do not change whether you have a drop or a gallon.
- Density: Density is a measure of mass per unit volume. It tells us how “packed” the matter is within a given space. For instance, lead is denser than aluminum, meaning a certain volume of lead will weigh more than the same volume of aluminum. Density is calculated as mass divided by volume ($rho = m/V$).
- Solubility: Solubility refers to the ability of a substance (solute) to dissolve in another substance (solvent) to form a homogeneous solution. For example, salt is soluble in water, meaning it dissolves to form saltwater. Different substances have varying solubilities in different solvents.
- Color: The color of a substance is how it appears under visible light. While seemingly simple, color is a physical property that can be crucial for identification. For example, pure gold has a distinct yellow color.
- Odor: The smell of a substance is another observable physical property. For example, ammonia has a pungent odor, while cinnamon has a sweet aroma.
- Hardness: Hardness refers to a material’s resistance to scratching or indentation. The Mohs scale of hardness is a common way to quantify the relative hardness of minerals, with diamond being the hardest.
- Ductility and Malleability: Ductility is the ability of a material to be drawn into thin wires, while malleability is the ability to be hammered or pressed into thin sheets without breaking. Metals like copper are highly ductile and malleable.
- Electrical Conductivity: This property describes how well a substance conducts electricity. Metals are generally excellent electrical conductors, while nonmetals are poor conductors.
- Thermal Conductivity: This property describes how well a substance conducts heat. Similar to electrical conductivity, metals are good thermal conductors.
Extrinsic Physical Properties
Extrinsic physical properties, also known as extensive properties, are dependent on the amount of substance present. These properties change as the size of the sample changes.
- Mass: The amount of matter in an object. A larger piece of gold will have more mass than a smaller piece.
- Volume: The amount of space an object occupies. A larger container will hold more volume of water.
- Length, Width, Height: These are all measurements of dimensions and are directly proportional to the size of the object.
The distinction between intrinsic and extrinsic properties is crucial. While mass and volume can help us quantify a substance, it’s the intrinsic properties like melting point and density that truly define what that substance is.
Changes in Physical State
Physical properties are often observed during physical changes, which are transformations that do not alter the chemical composition of a substance. These changes typically involve a change in state (solid, liquid, gas) or form.
- Melting and Freezing: When a solid melts, it becomes a liquid. The reverse process, freezing, occurs when a liquid becomes a solid. For example, ice melting into water is a physical change.
- Boiling and Condensation: Boiling is the transition of a liquid to a gas. Condensation is the reverse process, where a gas changes into a liquid. Steam rising from boiling water and then condensing on a cool surface is a physical change.
- Sublimation and Deposition: Sublimation is the direct transition of a solid to a gas, bypassing the liquid phase (e.g., dry ice turning into carbon dioxide gas). Deposition is the reverse process, where a gas directly forms a solid.
- Dissolving: When a solute dissolves in a solvent, it forms a solution. While the solute is dispersed throughout the solvent, its chemical identity remains intact. Evaporating the solvent can often recover the original solute, demonstrating the reversibility of this physical change.
- Changes in Shape and Size: Cutting, bending, or crushing a substance are all physical changes. The material’s chemical composition remains the same, even though its form is altered.
Exploring Chemical Properties
Chemical properties, in contrast to physical properties, describe a substance’s ability to undergo a chemical change or reaction by virtue of its composition. These properties involve the transformation of a substance into one or more new substances with different chemical identities. Observing chemical properties often requires performing a chemical reaction.
Reactivity and Flammability
Chemical properties are inherently linked to how a substance interacts with other substances or energy.
- Flammability: This is the ability of a substance to burn or ignite easily. For example, gasoline is highly flammable and will readily react with oxygen in the presence of an ignition source to produce heat, light, and new substances like carbon dioxide and water.
- Reactivity: This refers to how readily a substance undergoes a chemical reaction. Some substances are highly reactive and will react vigorously with even common substances like air or water, while others are very unreactive and exist in their elemental form for long periods. For instance, alkali metals like sodium are highly reactive with water, producing hydrogen gas and heat.
- Combustibility: Similar to flammability, combustibility describes a substance’s ability to react rapidly with an oxidant, usually oxygen, to produce heat and light. This is the process of burning.
- Oxidation State: The oxidation state of an atom indicates the degree of oxidation of an atom in a chemical compound. It’s a conceptual number used to track electrons in chemical reactions and can reveal a substance’s tendency to gain or lose electrons.
- Acidity and Basicity (pH): While pH can be measured physically, the tendency of a substance to be acidic or basic, its ability to donate or accept protons, is a chemical property. Acids react with bases in a neutralization reaction, forming salt and water.
- Toxicity: The degree to which a substance can damage an organism is a chemical property, as it often involves chemical reactions within biological systems.
- Heat of Combustion: This is the amount of heat released when a substance undergoes complete combustion. It’s a measure of the energy stored within the chemical bonds of the fuel.
Chemical Changes
Chemical changes, also known as chemical reactions, result in the formation of new chemical substances with different properties than the original reactants. These changes are typically irreversible or difficult to reverse. The key indicators of a chemical change include:
- Formation of a Gas: Bubbles appearing in a liquid can indicate the production of a gas.
- Formation of a Precipitate: A solid forming from a solution.
- Change in Color: A permanent change in color that cannot be explained by simple dilution.
- Change in Temperature: Release or absorption of heat (exothermic or endothermic reactions).
- Production of Light or Odor: The emission of light or a new smell.
A classic example of a chemical change is the rusting of iron. Iron reacts with oxygen in the presence of water to form iron oxide, a new substance with entirely different properties (rust) than the original iron.
Distinguishing Between Physical and Chemical Properties
The fundamental difference lies in whether the substance’s chemical identity is altered.
| Feature | Physical Properties | Chemical Properties |
|---|---|---|
| Observation | Without changing chemical identity | By undergoing a chemical change |
| Change in Identity | No new substance formed | New substance(s) formed |
| Reversibility | Often reversible | Generally irreversible or difficult to reverse |
| Focus | What it is like (e.g., color, hardness, melting point) | How it reacts or changes (e.g., flammability, reactivity) |
| Examples | Density, boiling point, color, mass, volume | Reactivity, flammability, acidity, toxicity |
Consider water. Its physical properties include being a colorless liquid at room temperature, having a boiling point of 100°C, and being able to dissolve many substances. These can be observed and measured without turning water into something else. However, water has chemical properties too. It can react with sodium metal in a vigorous chemical reaction to produce hydrogen gas and sodium hydroxide. In this case, the water molecule itself has been broken down and rearranged to form new substances.
Another way to think about it is that physical properties are concerned with the intermolecular forces and arrangement of molecules, while chemical properties are concerned with the intramolecular forces and the actual bonds within the molecules.
The Importance of Understanding Properties
A thorough understanding of both physical and chemical properties is foundational to numerous scientific disciplines and practical applications.
In Chemistry and Material Science
For chemists, identifying and characterizing substances is paramount. Physical properties serve as the initial identifiers, helping to distinguish between different compounds. Chemical properties then reveal how these compounds will behave, enabling chemists to predict reaction outcomes, design new synthesis routes, and understand the fundamental interactions between matter.
In material science, the selection of materials for specific applications hinges entirely on their properties. For instance, an engineer designing an aircraft wing will need materials with high strength-to-weight ratio (physical property) and resistance to corrosion (chemical property). A chef preparing a meal relies on the physical properties of ingredients like texture and solubility, as well as the chemical properties that lead to flavor development during cooking.

In Everyday Life
Even outside the laboratory, we constantly interact with and utilize the physical and chemical properties of matter. We choose clothing based on its physical properties like softness and warmth. We use cleaning products that leverage specific chemical properties to break down stains. Understanding that certain substances are flammable (chemical property) allows us to handle them safely. Recognizing that ice melts at a certain temperature (physical property) informs how we store food.
The distinction between physical and chemical properties allows us to categorize and comprehend the vast array of substances that make up our world. Physical properties describe the observable and measurable characteristics without altering the substance, while chemical properties define its potential to transform into something new. Together, they provide a comprehensive framework for understanding matter and its behavior.
