Combustion, a fundamental chemical process, is the rapid oxidation of a material in the exothermic chemical process of burning, releasing heat and light. While the term often evokes images of roaring flames and intense heat, the phenomenon is a complex interplay of chemical reactions and energy transformations. Understanding what accompanies this rapid oxidation is crucial for a myriad of applications, from controlling industrial processes to appreciating the fundamental physics behind everyday occurrences. At its core, combustion is about the interaction between a fuel and an oxidizer, typically atmospheric oxygen, leading to a cascade of events that result in the release of energy and the formation of new chemical species.
The Essential Ingredients: Fuel and Oxidizer
For combustion to occur, two primary components must be present: a fuel and an oxidizer. The nature of these components dictates the characteristics of the combustion process itself.
Fuel: The Source of Energy
The fuel is the substance that undergoes oxidation. Fuels can exist in solid, liquid, or gaseous states, and their chemical structure significantly influences how readily they combust and the energy they release.
Solid Fuels
Solid fuels, such as wood, coal, and charcoal, are common and have been utilized by humans for millennia. Their combustion is a heterogeneous process, meaning it occurs at the interface between the solid and the gaseous oxidizer. The initial stages involve the thermal decomposition of the solid, releasing volatile gases that then mix with oxygen and ignite. The remaining solid material undergoes direct oxidation, contributing to the overall heat release. The surface area of the fuel plays a critical role; finely divided solids will combust much more rapidly than a solid block due to the increased contact area with the oxidizer. The moisture content of solid fuels is also a significant factor, as energy must first be expended to vaporize this water before combustion can proceed efficiently.
Liquid Fuels
Liquid fuels, like gasoline, kerosene, and diesel, are vaporized before they can combust. This vaporization can occur through direct heating or by mixing with the surrounding air. The ease with which a liquid fuel vaporizes is described by its volatility, a property influenced by its boiling point and vapor pressure. More volatile fuels ignite and burn more readily. In many liquid-fueled combustion systems, a spray of fine droplets is introduced into the combustion chamber, significantly increasing the surface area available for reaction and promoting rapid burning. The composition of the liquid fuel, particularly the type and arrangement of hydrocarbon chains, determines the amount of energy released per unit mass and the byproducts of combustion.
Gaseous Fuels
Gaseous fuels, such as natural gas, propane, and hydrogen, are the most readily combustible because the fuel and oxidizer are already intimately mixed at the molecular level. This homogeneity leads to very rapid and efficient combustion. The primary challenge with gaseous fuels is often containment and controlled delivery to the combustion zone. The flame speed of a gaseous fuel, which is the speed at which the flame front propagates through the fuel-oxidizer mixture, is a critical parameter. Fuels like hydrogen have exceptionally high flame speeds, leading to very rapid combustion. The stoichiometry of the fuel-oxidizer mixture is paramount; too much or too little oxidizer can lead to incomplete combustion or prevent ignition altogether.
Oxidizer: The Reaction Partner
The oxidizer is the substance that reacts with the fuel, accepting electrons and causing the fuel to be oxidized. While atmospheric oxygen is the most common oxidizer, other substances can also act as oxidizers in specific contexts.
Atmospheric Oxygen
Oxygen constitutes approximately 21% of the Earth’s atmosphere and is the ubiquitous oxidizer for most combustion processes. Its presence is essential for the sustained burning of most common fuels. The concentration of oxygen in the air affects the rate of combustion; higher oxygen concentrations generally lead to faster and more intense burning. In some specialized applications, such as in rockets, pure oxygen or oxygen-rich mixtures are used as oxidizers to achieve very high energy output.
Other Oxidizers
In certain scenarios, other chemical compounds can serve as oxidizers. For instance, in rocket propulsion, substances like nitrogen tetroxide or hydrogen peroxide are used as oxidizers, often reacting with a fuel to produce large volumes of hot gas at high pressure. In some chemical reactions, halogens like chlorine or fluorine can act as oxidizers. These non-oxygen oxidizers can lead to different combustion products and energy release characteristics compared to oxygen-based combustion. The selection of an oxidizer is dictated by the specific requirements of the application, including desired performance, safety considerations, and the availability of materials.
The Energetic Transformation: Heat and Light Release

The hallmark of combustion is the release of energy, primarily in the form of heat and light. This energy release is a direct consequence of the chemical bonds being broken and reformed during the oxidation process.
Heat Generation: The Exothermic Nature
Combustion is an exothermic process, meaning it releases more energy than it consumes. This is because the chemical bonds in the products of combustion (e.g., carbon dioxide and water) are generally stronger and more stable than the chemical bonds in the reactants (fuel and oxygen). The formation of these more stable bonds releases energy. The amount of heat released per unit mass or volume of fuel is known as the enthalpy of combustion or heating value. This value is a critical property for assessing the energy potential of a fuel. The rate at which heat is released depends on the combustion rate, which is influenced by factors such as fuel type, oxidizer concentration, temperature, and pressure. Rapid oxidation leads to a rapid release of heat, resulting in the high temperatures characteristic of flames.
Light Emission: The Visible Flame
The light emitted during combustion originates from several sources. Primarily, it comes from the incandescence of soot particles and hot gases within the flame. As these particles and gas molecules are heated to very high temperatures, they emit electromagnetic radiation across the visible spectrum, making the flame visible. The color of the flame can provide clues about the combustion process. For example, a sooty flame, appearing yellow or orange, indicates incomplete combustion with the presence of incandescent carbon particles. A clean, blue flame, often seen with fuels like natural gas, suggests more complete combustion with fewer soot particles. In some cases, chemiluminescence, where light is emitted directly from chemical reactions, also contributes to the visible output of a flame.
The Chemical Byproducts: What Remains After Burning
Beyond heat and light, combustion results in the formation of new chemical substances, the byproducts of the reaction. The nature and quantity of these byproducts are dependent on the fuel’s composition and the completeness of the combustion process.
Complete Combustion Products
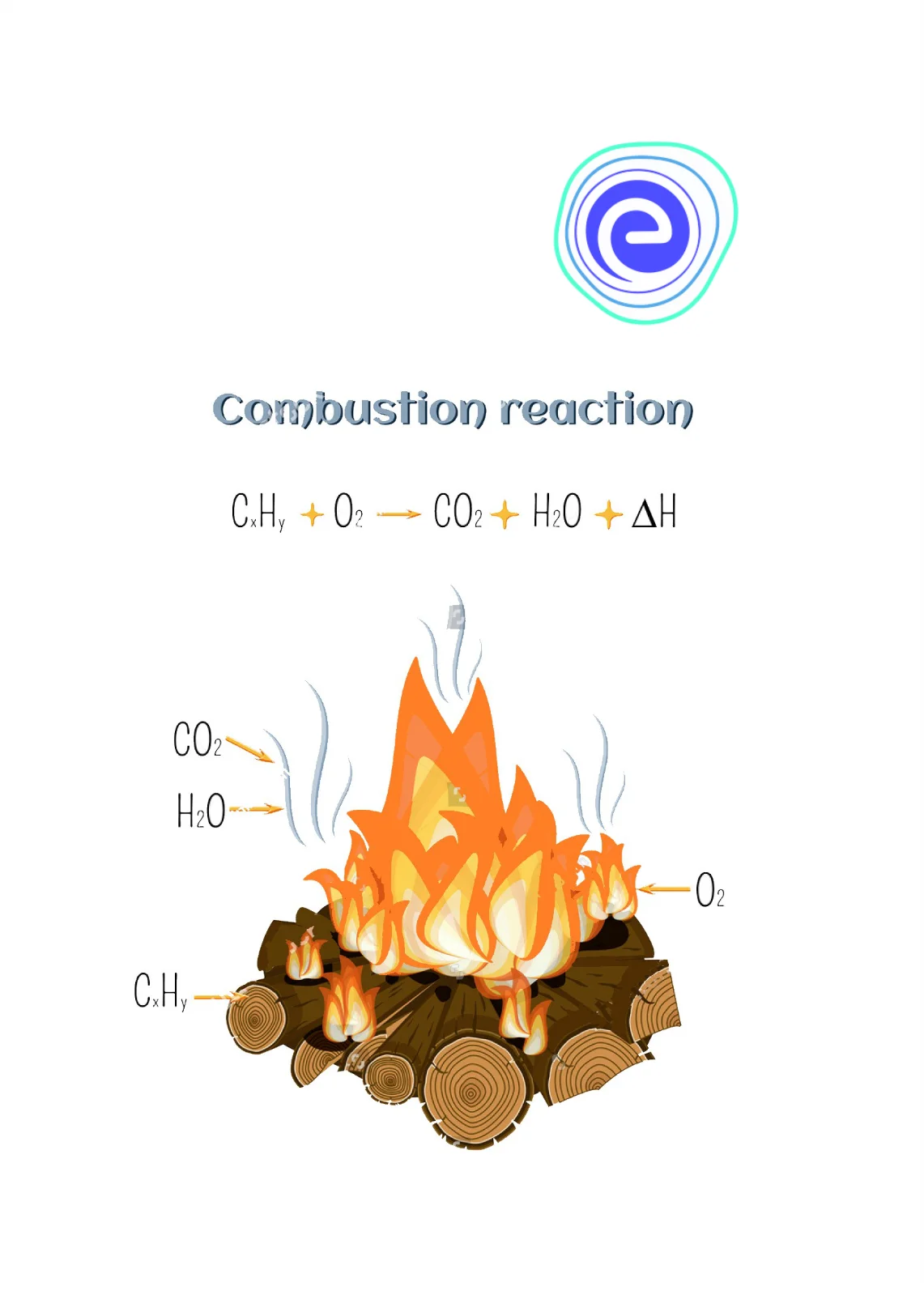
When a hydrocarbon fuel (composed of carbon and hydrogen) undergoes complete combustion with sufficient oxygen, the primary products are carbon dioxide ($text{CO}2$) and water ($text{H}2text{O}$). The chemical equations for these reactions are well-defined:
For methane ($text{CH}4$):
$text{CH}4 text{ (g) } + 2text{O}2 text{ (g) } rightarrow text{CO}2 text{ (g) } + 2text{H}_2text{O} text{ (g) } + text{Energy}$
For a more complex hydrocarbon, like octane ($text{C}8text{H}{18}$):
$2text{C}8text{H}{18} text{ (l) } + 25text{O}2 text{ (g) } rightarrow 16text{CO}2 text{ (g) } + 18text{H}_2text{O} text{ (g) } + text{Energy}$
Complete combustion is desirable from an efficiency standpoint as it maximizes the energy extracted from the fuel. However, it also produces greenhouse gases like carbon dioxide.
Incomplete Combustion Products
Incomplete combustion occurs when there is insufficient oxygen available for the fuel to fully oxidize. This leads to the formation of various undesirable byproducts, including:
- Carbon Monoxide ($text{CO}$): A highly toxic gas formed when carbon oxidizes to a lesser extent than $text{CO}_2$. It is a colorless, odorless gas that can be lethal in enclosed spaces.
- Soot (Particulate Carbon): Tiny solid particles of carbon that are not fully burned. As mentioned, soot contributes to the yellow coloration of flames and can have detrimental effects on air quality and respiratory health.
- Unburned Hydrocarbons: Portions of the fuel that did not react at all. These can contribute to air pollution and have distinct odors.
The presence of incomplete combustion products is a strong indicator of inefficient burning and can pose significant environmental and health hazards. Controlling the fuel-to-oxidizer ratio is paramount to minimizing these byproducts.

Other Combustion Byproducts
Depending on the elemental composition of the fuel, other byproducts can be formed. For example:
- Sulfur Oxides ($text{SOx}$): If the fuel contains sulfur (common in coal and some oils), sulfur dioxide ($text{SO}2$) and sulfur trioxide ($text{SO}3$) can be produced. These contribute to acid rain.
- Nitrogen Oxides ($text{NOx}$): At the high temperatures found in flames, nitrogen and oxygen from the air can react to form nitrogen oxides ($text{NO}$ and $text{NO}_2$). These are also air pollutants and precursors to smog.
Understanding and mitigating the formation of these various byproducts are key aspects of modern combustion engineering and environmental protection efforts. The drive towards cleaner energy sources and more efficient combustion technologies is largely motivated by the desire to minimize the release of harmful substances into the atmosphere.
