Glutamate, the most abundant excitatory neurotransmitter in the vertebrate central nervous system, plays a pivotal role in a vast array of cognitive functions, from learning and memory to synaptic plasticity and neuronal development. Far from being a simple chemical messenger, glutamate is a sophisticated molecule whose precise actions are modulated by intricate receptor systems and intricate cellular processes. Understanding its multifaceted roles is fundamental to comprehending how our brains function, process information, and adapt to our environment. Its influence extends to virtually every aspect of neural activity, making it a critical component of both healthy brain function and the pathophysiology of numerous neurological and psychiatric disorders.
The Excitatory Powerhouse: Glutamate as a Primary Neurotransmitter
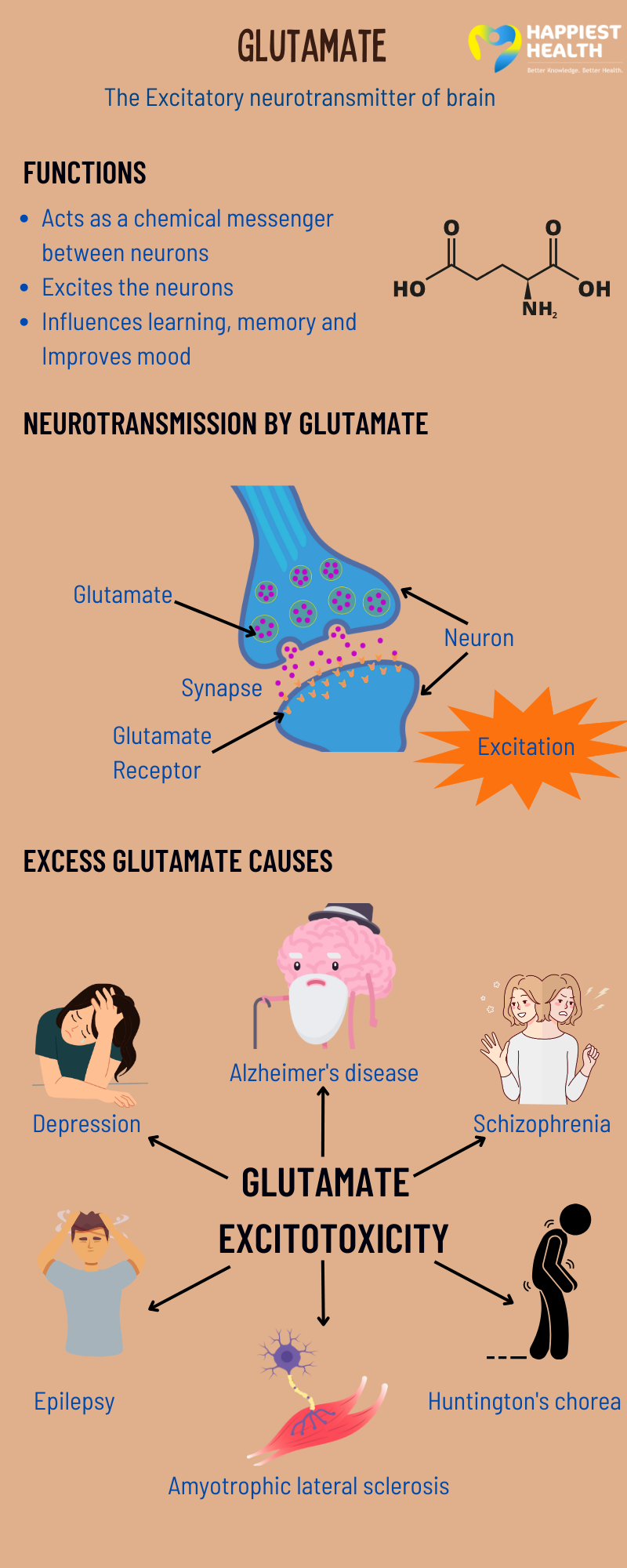
At its core, glutamate’s primary function is to excite neurons. This means that when glutamate is released into the synaptic cleft, it binds to specific receptors on the postsynaptic neuron, increasing the likelihood that this neuron will fire an action potential. This excitatory nature is the bedrock upon which most rapid neural communication in the brain is built.
Synaptic Transmission: The Basics of Glutamate’s Action
The process of synaptic transmission involving glutamate is a finely tuned cascade. A presynaptic neuron, upon receiving an electrical signal, releases glutamate into the synaptic cleft, the microscopic gap between neurons. This glutamate then diffuses across the cleft and binds to glutamate receptors on the postsynaptic neuron. This binding event triggers a change in the ion permeability of the postsynaptic membrane, typically leading to an influx of positively charged ions, such as sodium (Na+) and calcium (Ca2+), into the postsynaptic neuron. This influx of positive charge depolarizes the postsynaptic membrane, bringing it closer to its firing threshold. If the depolarization is sufficient, the postsynaptic neuron will fire its own action potential, propagating the signal to the next neuron in the circuit. This rapid and efficient transmission of signals is essential for everything from sensory perception to motor control.
Glutamate Receptors: A Diverse Family of Gatekeepers
The diverse effects of glutamate are mediated by a variety of receptor subtypes, each with unique properties and locations. These receptors can be broadly classified into two main categories: ionotropic receptors and metabotropic receptors.
Ionotropic Glutamate Receptors: Fast and Direct Signaling
Ionotropic glutamate receptors are ligand-gated ion channels. This means that when glutamate binds to them, they directly open, allowing ions to flow across the membrane. There are three main subtypes of ionotropic glutamate receptors:
-
NMDA Receptors (N-methyl-D-aspartate receptors): These receptors are particularly important for synaptic plasticity and learning and memory. They are characterized by a unique voltage-dependent magnesium block, meaning that at resting membrane potentials, a magnesium ion (Mg2+) physically blocks the ion channel. Only when the postsynaptic membrane is sufficiently depolarized (often by the activation of AMPA receptors) can the Mg2+ block be removed, allowing ions like Ca2+ to flow through. The influx of Ca2+ through NMDA receptors is a critical trigger for many downstream signaling pathways.
-
AMPA Receptors (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors): These are the primary mediators of fast excitatory synaptic transmission. When glutamate binds to AMPA receptors, they open and allow the rapid influx of Na+ ions, causing depolarization. AMPA receptors are crucial for the initial excitatory postsynaptic potential (EPSP) and are heavily involved in synaptic strength.
-
Kainate Receptors: Similar to AMPA receptors, kainate receptors are ionotropic channels that mediate excitatory transmission. However, they are generally less abundant and their roles are more diverse, sometimes acting presynaptically to modulate neurotransmitter release.
Metabotropic Glutamate Receptors (mGluRs): Slower, Modulatory Effects
Metabotropic glutamate receptors are G protein-coupled receptors (GPCRs). Unlike ionotropic receptors, they do not directly form ion channels. Instead, upon glutamate binding, they activate intracellular signaling cascades via G proteins. These cascades can have a variety of effects, including modulating the opening of ion channels, altering gene expression, and influencing the release of other neurotransmitters. mGluRs are classified into three groups (Group I, II, and III), each with distinct signaling pathways and functions, often acting as modulators of synaptic strength and excitability.
Glutamate’s Crucial Roles in Cognitive Function
The widespread presence and diverse receptor subtypes of glutamate enable it to participate in a remarkable range of cognitive processes. Its excitatory nature is fundamental to how information is encoded, processed, and retrieved by the brain.
Learning and Memory: The Foundation of Synaptic Plasticity
One of the most extensively studied roles of glutamate is its involvement in learning and memory. This connection is primarily mediated through the concept of synaptic plasticity, the ability of synapses to strengthen or weaken over time in response to changes in their activity.
-
Long-Term Potentiation (LTP): Glutamate, particularly through NMDA receptor activation, is a key player in LTP, a persistent strengthening of synaptic connections that is widely believed to be a cellular mechanism underlying learning and memory. When a synapse is repeatedly activated, the increased influx of Ca2+ through NMDA receptors triggers a cascade of intracellular events that lead to an increase in the number and sensitivity of AMPA receptors on the postsynaptic membrane. This makes the synapse more responsive to future glutamate release, effectively “potentiating” the connection.
-
Long-Term Depression (LTD): Conversely, glutamate also participates in LTD, a weakening of synaptic connections. LTD can occur under conditions of low-frequency stimulation or specific patterns of activity, often involving different patterns of Ca2+ influx and activation of specific intracellular enzymes. This ability to both strengthen and weaken connections allows for the flexible refinement of neural circuits.
Neuronal Development and Circuit Formation
Beyond its role in established neural circuits, glutamate is also essential for the proper development of the brain. It influences neuronal migration, differentiation, and the formation of synaptic connections.
-
Neuronal Growth and Survival: Glutamate signaling is critical for promoting the growth and survival of developing neurons. Inappropriate levels of glutamate during development, however, can have detrimental effects.
-
Synapse Formation and Elimination: Glutamate acts as a cue for synaptogenesis, the process by which new synapses are formed. It also plays a role in the pruning of synapses, a critical process that refines neural circuits during development by eliminating unnecessary connections.
Sensory Processing and Motor Control
The rapid excitatory signaling of glutamate is fundamental to the brain’s ability to process sensory information and execute motor commands.
-
Vision and Audition: Glutamate is the primary neurotransmitter in many sensory pathways, including the visual and auditory systems, allowing for the rapid transmission of sensory input from the periphery to higher brain centers.
-
Motor Function: In the motor cortex and spinal cord, glutamate is essential for the activation of motor neurons that control muscle movement.
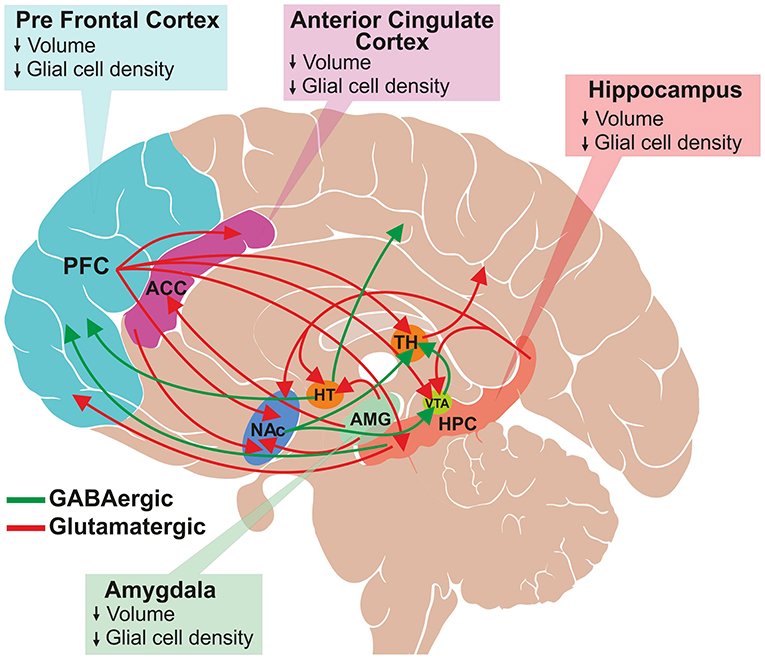
The Delicate Balance: Glutamate Dysregulation and Neurological Disorders
While glutamate is vital for normal brain function, its activity must be tightly regulated. Dysregulation of glutamate signaling, either excessive excitation or insufficient function, is implicated in a wide range of neurological and psychiatric conditions.
Excitotoxicity: The Peril of Overexcitation
When glutamate levels become excessively high, or when its clearance from the synaptic cleft is impaired, it can lead to overstimulation of neurons. This overstimulation, particularly involving excessive Ca2+ influx through NMDA receptors, can trigger a cascade of damaging events, leading to neuronal death. This phenomenon is known as excitotoxicity and is implicated in conditions such as:
-
Stroke: During a stroke, the disruption of blood flow leads to oxygen and glucose deprivation, which can impair glutamate reuptake mechanisms. This results in a surge of extracellular glutamate and subsequent excitotoxicity, contributing significantly to neuronal damage.
-
Traumatic Brain Injury (TBI): Similar to stroke, TBI can trigger the release of excessive glutamate, leading to excitotoxic neuronal death and further neurological deficits.
-
Neurodegenerative Diseases: While the role is complex and debated, excitotoxicity is thought to contribute to the progressive neuronal loss seen in diseases like Alzheimer’s disease and Huntington’s disease.
Imbalances in Glutamate Signaling and Psychiatric Illnesses
Alterations in glutamate neurotransmission are also implicated in various psychiatric disorders, highlighting the intricate relationship between neurotransmitter balance and mental health.
-
Schizophrenia: Evidence suggests that NMDA receptor hypofunction (reduced activity) may play a role in the cognitive deficits and positive symptoms associated with schizophrenia. Drugs that block NMDA receptors, like phencyclidine (PCP), can induce symptoms that resemble schizophrenia.
-
Depression and Anxiety: While the primary neurotransmitters implicated in depression and anxiety are often serotonin and norepinephrine, there is growing evidence suggesting that glutamate system dysfunction also contributes to these conditions. Antidepressant medications targeting glutamate pathways are an active area of research.
-
Addiction: Glutamate signaling is crucial for the brain’s reward pathways, and chronic drug use can lead to profound alterations in glutamate transmission, contributing to the compulsive drug-seeking behavior characteristic of addiction.
Maintaining Equilibrium: Glutamate Homeostasis
Given the profound impact of glutamate on brain function, the brain has evolved sophisticated mechanisms to maintain glutamate homeostasis – a precise balance between its release, action at receptors, and clearance.
Glutamate Transporters: The Cleanup Crew
Specialized transporter proteins, known as excitatory amino acid transporters (EAATs), are crucial for removing glutamate from the synaptic cleft and surrounding extracellular space. These transporters are located on both neurons and glial cells (particularly astrocytes). Astrocytes, with their extensive network of processes ensheathing synapses, play a particularly vital role in glutamate uptake.
-
Neuronal Transporters (e.g., Excitatory Amino Acid Transporter 3 – EAAT3): These are primarily located on the presynaptic neuron and can be involved in glutamate reuptake for recycling.
-
Astrocyte Transporters (e.g., Excitatory Amino Acid Transporter 1 – EAAT1 and EAAT2): These are the primary mediators of glutamate clearance from the extracellular space. By rapidly removing glutamate, they prevent overstimulation and excitotoxicity, ensuring that synaptic transmission is precisely controlled. Impairment of these astrocyte transporters is strongly linked to neurological disorders.
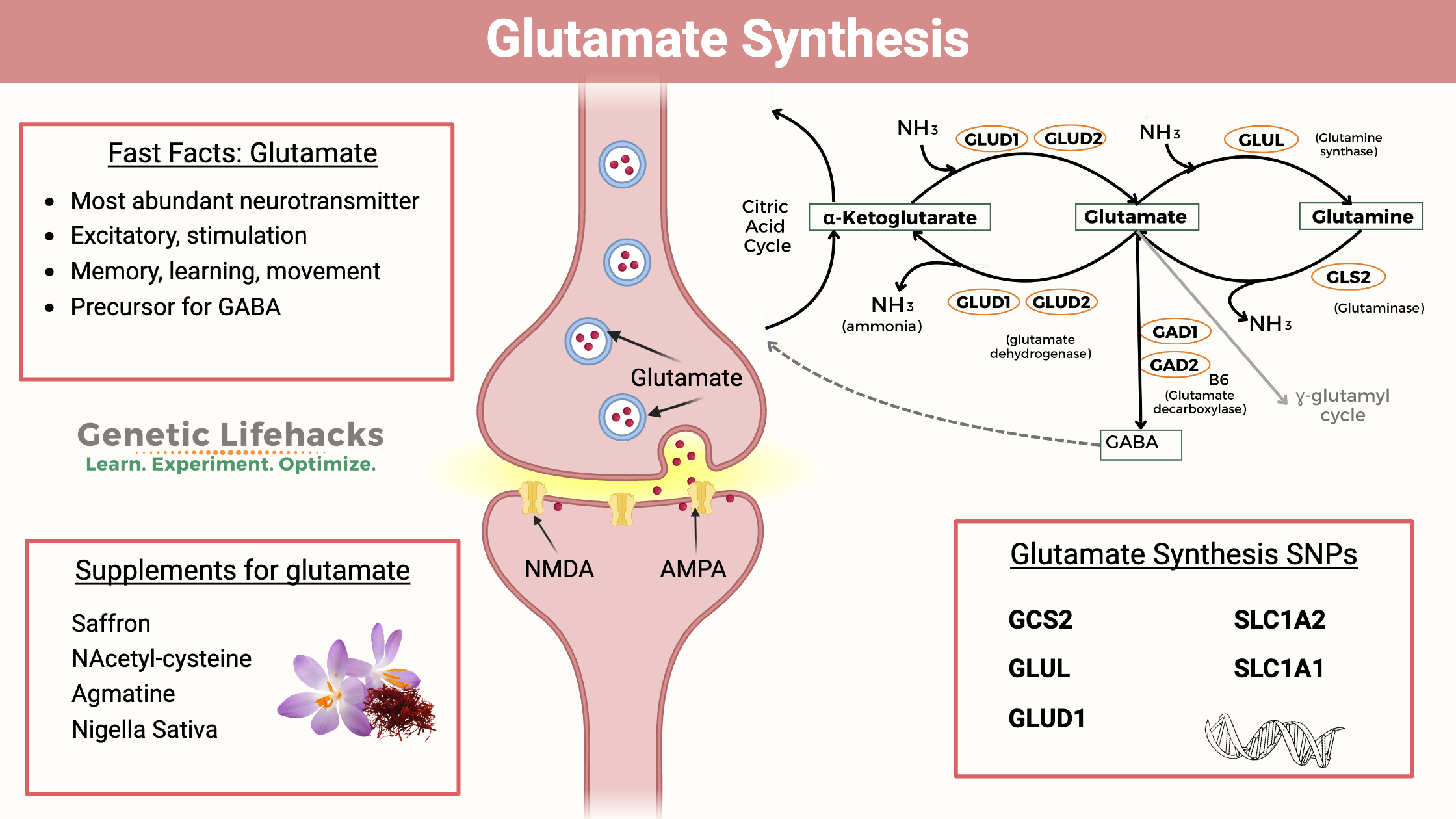
The Glutamate-Glutamine Cycle: Recycling for Efficiency
Once glutamate is taken up by astrocytes, it is converted into glutamine by the enzyme glutamine synthetase. Glutamine is then released from astrocytes and can be taken up by neurons, where it is converted back into glutamate. This glutamate-glutamine cycle is a highly efficient system that ensures a continuous supply of glutamate for neurotransmission while preventing its accumulation in the extracellular space. This cycle is critical for maintaining neuronal function and preventing excitotoxicity.
In conclusion, glutamate is a remarkably versatile and essential molecule in the brain. Its ability to act as a potent excitatory neurotransmitter, coupled with the sophisticated regulation provided by its diverse receptor subtypes and efficient clearance mechanisms, makes it fundamental to nearly every aspect of neural activity, from the most basic sensory perceptions to the highest levels of cognition. Understanding the intricate dance of glutamate is not only key to unraveling the mysteries of the healthy brain but also to developing effective treatments for the myriad of neurological and psychiatric conditions that arise when this delicate balance is disrupted.
