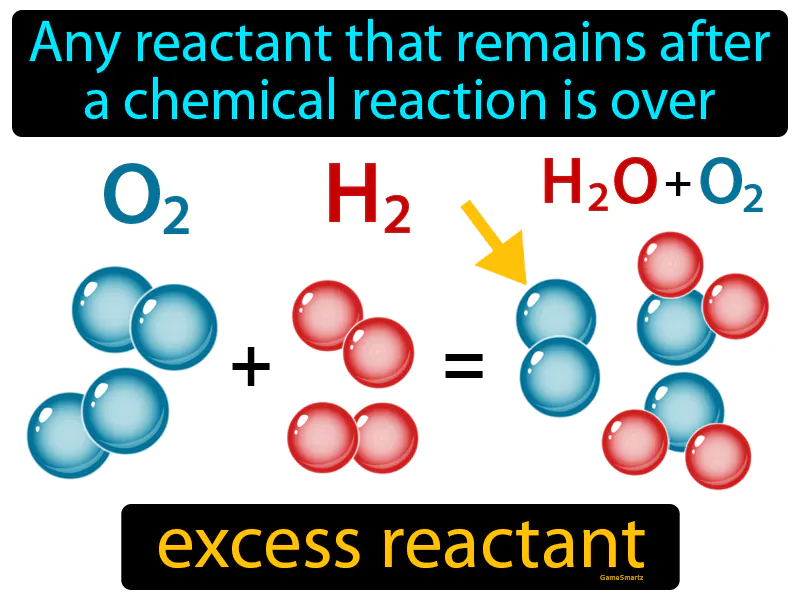
The term “excess reagent” is fundamental to understanding stoichiometry, the quantitative relationship between reactants and products in chemical reactions. In essence, it refers to the reactant that is present in a larger amount than is stoichiometrically required for a complete reaction with the limiting reagent. Identifying and understanding the excess reagent is crucial for predicting the theoretical yield of a product, optimizing reaction efficiency, and controlling the composition of the final product in various chemical processes. This concept is not confined to academic laboratories but has significant implications in industrial chemical manufacturing, where resource management and cost-effectiveness are paramount.

Understanding the Fundamentals of Stoichiometry
Stoichiometry is the bedrock upon which the concept of excess reagents is built. It’s the quantitative study of reactants and products in a chemical reaction, based on the law of conservation of mass. This means that in a closed system, matter is neither created nor destroyed. Chemical equations, when properly balanced, represent the precise molar ratios of reactants and products involved in a reaction. These ratios are derived from the molecular formulas of the substances and are critical for any stoichiometric calculation.
The Importance of Balanced Chemical Equations
A balanced chemical equation is a symbolic representation of a chemical reaction where the number of atoms of each element is the same on both the reactant and product sides. This balance ensures that the law of conservation of mass is upheld. For instance, the reaction between hydrogen and oxygen to form water is represented as:
$2H2 (g) + O2 (g) rightarrow 2H_2O (l)$
In this equation, two moles of hydrogen gas react with one mole of oxygen gas to produce two moles of water. These stoichiometric coefficients (2, 1, and 2) dictate the exact proportions in which these substances react. If we were to deviate from these proportions, one of the reactants would inevitably be left over after the reaction has gone to completion. This leftover reactant is what we identify as the excess reagent.
Molar Ratios and Their Role
The molar ratio, derived from the coefficients in a balanced chemical equation, is the key to determining how much of each reactant is needed to react completely with another. It’s a conversion factor that allows us to relate the amount of one substance in a reaction to the amount of another. For example, in the synthesis of ammonia from nitrogen and hydrogen:
$N2 (g) + 3H2 (g) rightarrow 2NH_3 (g)$
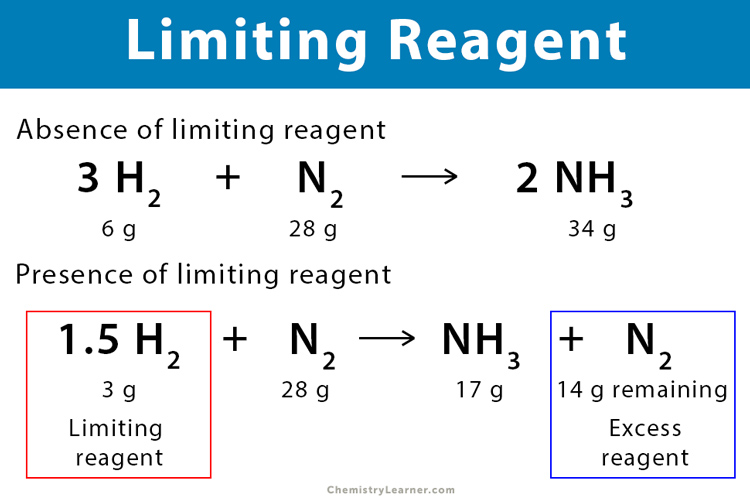
The molar ratio between nitrogen and hydrogen is 1:3. This means that for every one mole of nitrogen that reacts, three moles of hydrogen are required. If we have, say, two moles of nitrogen and only five moles of hydrogen, we can use the molar ratio to determine which reactant is in excess. We would need $2 text{ moles } N2 times frac{3 text{ moles } H2}{1 text{ mole } N2} = 6 text{ moles } H2$ to react completely with two moles of nitrogen. Since we only have five moles of hydrogen, hydrogen is the limiting reagent, and nitrogen will be left over as the excess reagent.
Identifying the Limiting and Excess Reagents
The identification of the limiting and excess reagents is a critical step in any quantitative chemical analysis or synthesis. The limiting reagent is the reactant that is completely consumed first in a chemical reaction, thereby determining the maximum amount of product that can be formed. The excess reagent, conversely, is the reactant that is not completely used up and remains after the limiting reagent has been depleted.
The Concept of the Limiting Reagent
The limiting reagent dictates the theoretical yield of a chemical reaction. Once the limiting reagent is exhausted, the reaction stops, regardless of how much of the other reactants are still present. Imagine baking a cake that requires two cups of flour and one cup of sugar. If you only have one and a half cups of flour but plenty of sugar, the flour is your limiting ingredient, and you can only make a smaller cake. The same principle applies to chemical reactions.
Methods for Determining the Excess Reagent
There are several systematic methods to determine the limiting and excess reagents. One common approach involves calculating the amount of product that could be formed from each reactant, assuming the other reactant is in sufficient supply. The reactant that produces the least amount of product is the limiting reagent. Alternatively, one can compare the mole ratio of the reactants present to the stoichiometric mole ratio.
Let’s consider an example. Suppose we have 10 grams of sodium ($Na$) reacting with 10 grams of chlorine ($Cl_2$) to form sodium chloride ($NaCl$):
$2Na (s) + Cl_2 (g) rightarrow 2NaCl (s)$
First, we convert the masses to moles using their molar masses:
Molar mass of $Na approx 23.0 text{ g/mol}$
Molar mass of $Cl_2 approx 71.0 text{ g/mol}$
Moles of $Na = frac{10 text{ g}}{23.0 text{ g/mol}} approx 0.435 text{ mol}$
Moles of $Cl_2 = frac{10 text{ g}}{71.0 text{ g/mol}} approx 0.141 text{ mol}$
From the balanced equation, the stoichiometric ratio of $Na$ to $Cl_2$ is 2:1.
Now, let’s determine how much $Cl2$ is needed to react with all the $Na$:
Moles of $Cl2$ required = $0.435 text{ mol } Na times frac{1 text{ mol } Cl2}{2 text{ mol } Na} approx 0.218 text{ mol } Cl2$
We only have 0.141 mol of $Cl2$, which is less than the 0.218 mol required. Therefore, $Cl2$ is the limiting reagent.
Alternatively, let’s determine how much $Na$ is needed to react with all the $Cl2$:
Moles of $Na$ required = $0.141 text{ mol } Cl2 times frac{2 text{ mol } Na}{1 text{ mol } Cl_2} approx 0.282 text{ mol } Na$
We have 0.435 mol of $Na$, which is more than the 0.282 mol required. Thus, $Na$ is the excess reagent.
Calculating the Amount of Excess Reagent
Once the limiting and excess reagents are identified, we can calculate the exact amount of the excess reagent remaining. In the previous example, we started with 0.435 mol of $Na$ and determined that 0.282 mol of $Na$ would react with the limiting reagent ($Cl_2$).
Amount of excess $Na$ = Initial moles of $Na$ – Moles of $Na$ reacted
Amount of excess $Na$ = $0.435 text{ mol } – 0.282 text{ mol} = 0.153 text{ mol}$
To express this in grams, we multiply by the molar mass of sodium:
Mass of excess $Na = 0.153 text{ mol} times 23.0 text{ g/mol} approx 3.52 text{ g}$
So, approximately 3.52 grams of sodium would remain unreacted after the reaction.
Practical Applications and Significance of Excess Reagents
The concept of excess reagents extends far beyond theoretical calculations; it has profound practical implications in various scientific and industrial domains. Controlling the amount of reactants used can significantly impact the efficiency, cost, and purity of chemical products.
Industrial Chemical Synthesis
In large-scale industrial processes, the efficient use of raw materials is paramount for economic viability. Often, one reactant is deliberately added in excess to ensure that the more expensive or critical reactant is completely consumed. This strategy is employed to maximize the yield of the desired product and minimize waste. For instance, in the Haber-Bosch process for ammonia synthesis, hydrogen is often used in excess to ensure that nitrogen, a more valuable reactant for the process, is fully utilized.
Analytical Chemistry and Titration
Excess reagents play a crucial role in analytical chemistry, particularly in titration experiments. In an excess titration, a known excess of a reagent is added to react with the analyte. The unreacted excess reagent is then titrated with a standard solution. This method is particularly useful when the reaction between the analyte and the titrant is slow, incomplete, or when the analyte itself cannot be accurately titrated directly. By adding an excess of the first reagent, we ensure that all the analyte has reacted, and the subsequent titration of the excess allows for indirect quantification of the analyte.
Purification and Byproduct Control
Sometimes, an excess of a particular reagent can be used to drive a reaction to completion and simultaneously act as a scavenger for unwanted byproducts. In organic synthesis, for instance, adding an excess of a dehydrating agent can ensure the complete removal of water, which might otherwise inhibit the reaction or lead to undesirable side reactions. Similarly, in certain precipitation reactions, an excess of one ion can be added to ensure that the target ion is completely precipitated, leaving the excess ion in solution, which can then be separated. The control over the presence and amount of excess reagents allows chemists to fine-tune reaction conditions to achieve desired product purity and minimize the formation of unwanted side products.
Factors Influencing the Choice of Excess Reagent
The decision of which reactant to use in excess is not arbitrary; it’s a strategic choice influenced by several critical factors. These decisions are driven by economic considerations, reaction kinetics, safety, and the desired outcome of the chemical process.
Cost and Availability of Reactants
The most significant factor in choosing an excess reagent is often the relative cost and availability of the reactants. If one reactant is considerably cheaper or more readily available than the other, it makes economic sense to use the less expensive one in excess to ensure the complete consumption of the more valuable reactant. This strategy directly impacts the overall production cost and profitability of a chemical process.
Reaction Kinetics and Completion
The rate at which a chemical reaction proceeds is also a crucial consideration. If a reaction is slow or tends to reach equilibrium before completion, adding one reactant in excess can help to push the equilibrium towards product formation, thereby increasing the overall yield. This is particularly relevant in reversible reactions where the removal or excess of one component can significantly influence the reaction’s progress according to Le Chatelier’s principle. For reactions with slow kinetics, using an excess of one reactant can ensure that the limiting reagent is fully utilized within a practical timeframe.
Purity Requirements and Byproduct Formation
The desired purity of the final product and the potential for byproduct formation also guide the selection of an excess reagent. If a particular reactant is prone to forming undesirable byproducts, it might be chosen as the limiting reagent to minimize its presence and potential for side reactions. Conversely, if one reactant can act as a catalyst or a scavenger for impurities, it might be used in excess. For example, in certain esterification reactions, using an excess of the alcohol can help to drive the reaction to completion and minimize side reactions of the carboxylic acid. Understanding the reaction mechanism and potential side pathways is essential in making an informed decision.
