Understanding the Legal Framework and Classification of Controlled Substances
The term “controlled substance” carries significant weight, often conjuring images of illicit drugs and strict legal repercussions. However, a deeper understanding reveals a complex legal framework designed to regulate substances that have the potential for abuse or addiction. These substances, whether naturally occurring or synthetically produced, are categorized and controlled by governmental bodies to prevent their misuse, diversion, and trafficking, while still allowing for legitimate medical and scientific purposes. This article will delve into the definition, classification, and regulatory landscape surrounding controlled substances.
Defining Controlled Substances
At its core, a controlled substance is any chemical or drug whose possession, manufacture, distribution, and use are regulated by law. The primary objective behind controlling these substances is to protect public health and safety. This regulation stems from the recognition that certain substances, if misused, can lead to severe health consequences, addiction, crime, and societal disruption. The classification and scheduling of these substances are based on a scientific and medical evaluation of their potential for abuse, addiction, and their accepted medical uses.

Potential for Abuse and Addiction
The defining characteristic of a controlled substance is its potential for abuse. This refers to the likelihood that a drug or chemical will be used in a manner not prescribed by a medical professional or in quantities exceeding recommended dosages. Abuse can lead to a compulsive seeking and use of the substance, even in the face of harmful consequences. Addiction, a more severe and chronic condition, is characterized by a psychological or physical dependence on a substance, making it extremely difficult for an individual to stop using it. Substances with a high potential for abuse and addiction are typically placed under stricter regulatory control.
Accepted Medical Uses
Crucially, the legal status of a controlled substance is not solely determined by its potential for abuse. Its recognized therapeutic value also plays a significant role. Substances that have legitimate and accepted medical uses, such as certain painkillers, anesthetics, or psychotropic medications, are still subject to control, but their regulation aims to balance legitimate medical access with the prevention of misuse. The less accepted or entirely absent the medical use, the more stringent the controls tend to be. For example, a substance with no recognized medical use and a high potential for abuse will be subject to the highest level of control.
Classification and Scheduling of Controlled Substances
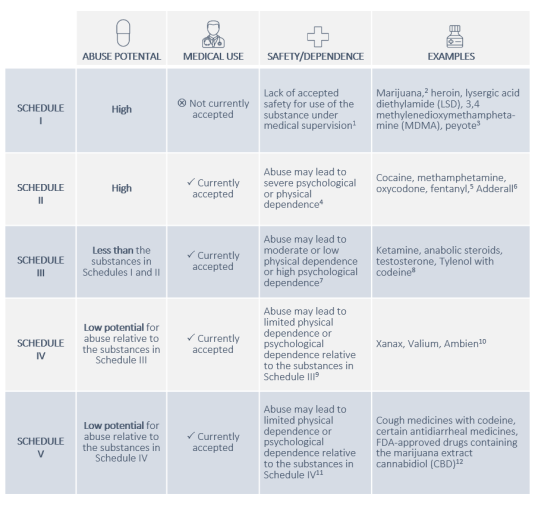
The most prominent framework for classifying controlled substances in the United States is the Controlled Substances Act (CSA) of 1970. This act categorizes drugs and other substances into five distinct schedules, based on their accepted medical use, potential for abuse, and likelihood of causing dependence. These schedules are not static and can be amended by the U.S. Congress or through administrative rule-making by the U.S. Department of Health and Human Services (HHS) and the Drug Enforcement Administration (DEA).
Schedule I Substances
Schedule I substances are those that have a high potential for abuse, no currently accepted medical use in treatment in the United States, and a lack of accepted safety for use under medical supervision. This category represents the most stringent level of control. Examples of Schedule I substances include heroin, lysergic acid diethylamide (LSD), marijuana (though its scheduling is a subject of ongoing debate and legislative action), peyote, and ecstasy (MDMA). The strict classification of Schedule I substances means that their possession, distribution, and manufacturing are generally illegal, with very limited exceptions for approved research purposes.
Schedule II Substances
Schedule II substances also have a high potential for abuse, but they do have a currently accepted medical use in treatment in the United States, or a currently accepted medical use with severe restrictions. Abuse of Schedule II substances may lead to severe psychological or physical dependence. Examples include potent prescription opioids like oxycodone (OxyContin), hydrocodone (Vicodin), morphine, and stimulants such as amphetamines (Adderall) and methamphetamine. Due to their high potential for abuse and addiction, these substances are subject to strict regulations regarding prescribing, dispensing, and record-keeping.
Schedule III Substances
Schedule III substances have a potential for abuse that is less than that of Schedule I and II substances. They have a currently accepted medical use in treatment in the United States, and abuse of Schedule III substances may lead to moderate or low physical dependence or high psychological dependence. This category includes substances like ketamine, anabolic steroids, and certain prescription medications containing lower doses of controlled substances, such as Tylenol with codeine. While still regulated, the controls for Schedule III substances are less restrictive than for Schedules I and II.
Schedule IV Substances
Schedule IV substances have a low potential for abuse relative to the substances in Schedule III. They have a currently accepted medical use in treatment in the United States, and abuse of Schedule IV substances may lead to limited physical dependence or psychological dependence relative to the substances in Schedule III. Examples include benzodiazepines like Xanax, Valium, and Ativan, as well as certain sedatives like Ambien and hypnotics. Prescriptions for Schedule IV substances typically have limitations on refills.
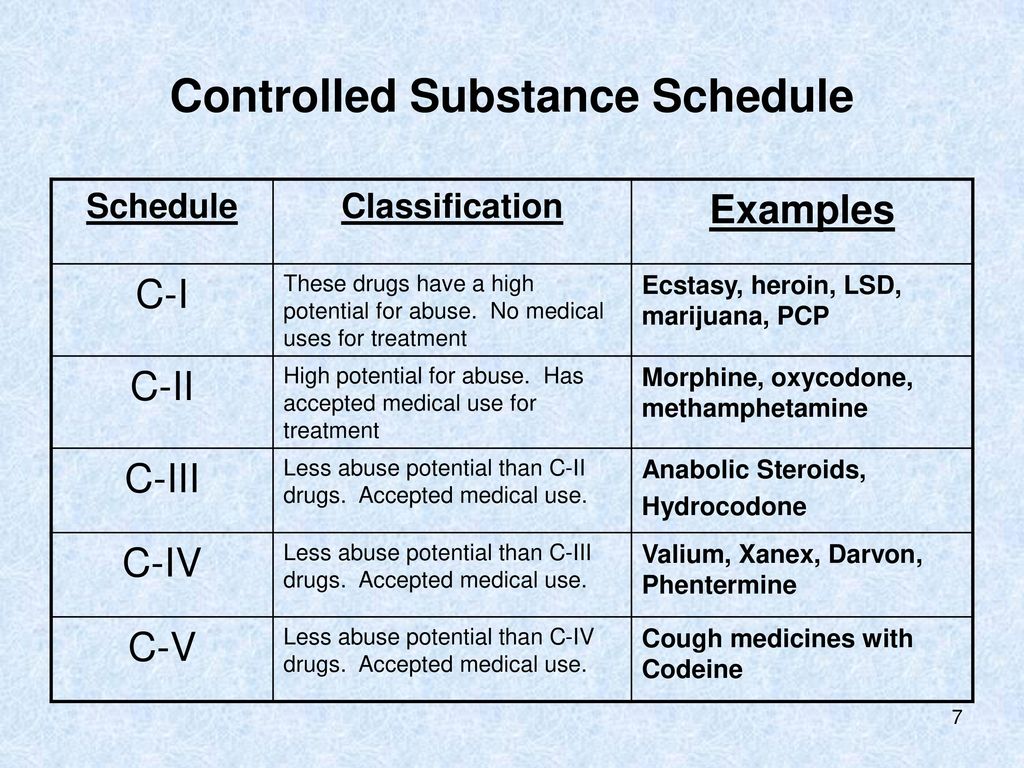
Schedule V Substances
Schedule V substances have a low potential for abuse relative to substances in Schedule IV. They have a currently accepted medical use in treatment in the United States, and abuse of Schedule V substances may lead to limited physical dependence or psychological dependence relative to substances in Schedule IV. These are generally preparations containing small amounts of certain stimulant or depressant drugs that are used for antidiarrheal, antitussive, or analgesic purposes. Examples include cough medicines containing codeine, such as Robitussin AC, in limited quantities. These substances are often available over-the-counter in some jurisdictions but still under specific regulatory controls.
Regulatory Bodies and Enforcement
The regulation and enforcement of controlled substance laws are multifaceted, involving various governmental agencies at both the federal and state levels. These bodies work in concert to prevent illegal drug trafficking, diversion of prescription medications, and to ensure that legitimate medical and scientific needs are met.
The Drug Enforcement Administration (DEA)
The Drug Enforcement Administration (DEA) is the primary federal agency responsible for enforcing the CSA. The DEA’s mission includes investigating and prosecuting individuals and organizations involved in the illegal drug trade, as well as regulating the legitimate pharmaceutical industry to prevent diversion. They conduct investigations, gather intelligence, and work with international partners to combat the global drug problem. The DEA also plays a key role in the scheduling and rescheduling of substances under the CSA, based on scientific and medical evaluations.
The Food and Drug Administration (FDA)
The Food and Drug Administration (FDA) is responsible for evaluating the safety and efficacy of drugs for medical use. While the DEA focuses on enforcement and control, the FDA determines whether a drug has accepted medical uses, which is a crucial factor in its scheduling. The FDA’s role in assessing the therapeutic value and safety of a substance directly influences its placement within the CSA schedules.
State and Local Authorities
In addition to federal agencies, state and local law enforcement agencies and regulatory bodies are instrumental in enforcing controlled substance laws. Each state has its own laws and regulations regarding controlled substances, which may be more stringent than federal laws. These authorities are responsible for arresting individuals suspected of drug offenses, seizing illegal substances, and prosecuting drug-related crimes within their jurisdictions. Many states have their own versions of the CSA, often mirroring the federal schedules.
The Importance of Legal Compliance
For individuals, businesses, and healthcare professionals, understanding and adhering to controlled substance laws is paramount. Non-compliance can lead to severe penalties, including hefty fines, imprisonment, loss of professional licenses, and a permanent criminal record.
Prescribing and Dispensing Regulations
Healthcare providers who prescribe or dispense controlled substances must be acutely aware of the regulations governing their practice. This includes obtaining the necessary DEA registration, maintaining meticulous records of all controlled substance transactions, and adhering to prescription requirements, such as the use of secure prescription pads and electronic prescribing systems where applicable. Understanding the specific schedules and associated restrictions for each substance is crucial to avoid accidental violations.
Research and Development
For researchers working with controlled substances, obtaining the necessary DEA registration and permits is essential. Research involving Schedule I substances, in particular, is subject to stringent oversight and requires detailed protocols and approvals to ensure that the substances are used solely for legitimate scientific investigation and are properly accounted for. Diversion of research materials can have serious legal consequences.
Public Health Initiatives and Treatment
Beyond enforcement, controlled substance laws also intersect with public health initiatives aimed at prevention, treatment, and harm reduction. By controlling access to potentially dangerous substances, these laws are intended to reduce addiction rates and the associated societal costs. Resources are often dedicated to providing education about the risks of substance abuse and offering treatment options for individuals struggling with addiction. The classification system helps guide the development of effective treatment strategies and the availability of medications for opioid use disorder and other substance use disorders.
In conclusion, controlled substances are a complex category of chemicals and drugs that are subject to rigorous legal regulation due to their potential for abuse and addiction. The classification system, embodied by the CSA schedules, provides a framework for differentiating substances based on their medical utility and risk profile. The diligent work of regulatory bodies like the DEA and FDA, alongside state and local authorities, aims to strike a balance between facilitating legitimate medical access and preventing the harmful consequences of substance misuse, thereby safeguarding public health and safety. Understanding these classifications and regulations is vital for all stakeholders involved in the handling, prescribing, or research of these powerful substances.
