Dispersion forces, also known as London dispersion forces or instantaneous dipole-induced dipole forces, are the weakest of the intermolecular forces, yet they play a crucial role in determining the physical properties of many substances. While often overshadowed by stronger interactions like hydrogen bonding or dipole-dipole forces, dispersion forces are ubiquitous, present in all molecules, polar and nonpolar alike. Understanding their nature and influence is fundamental to comprehending phenomena ranging from the boiling points of hydrocarbons to the interactions between biological molecules.
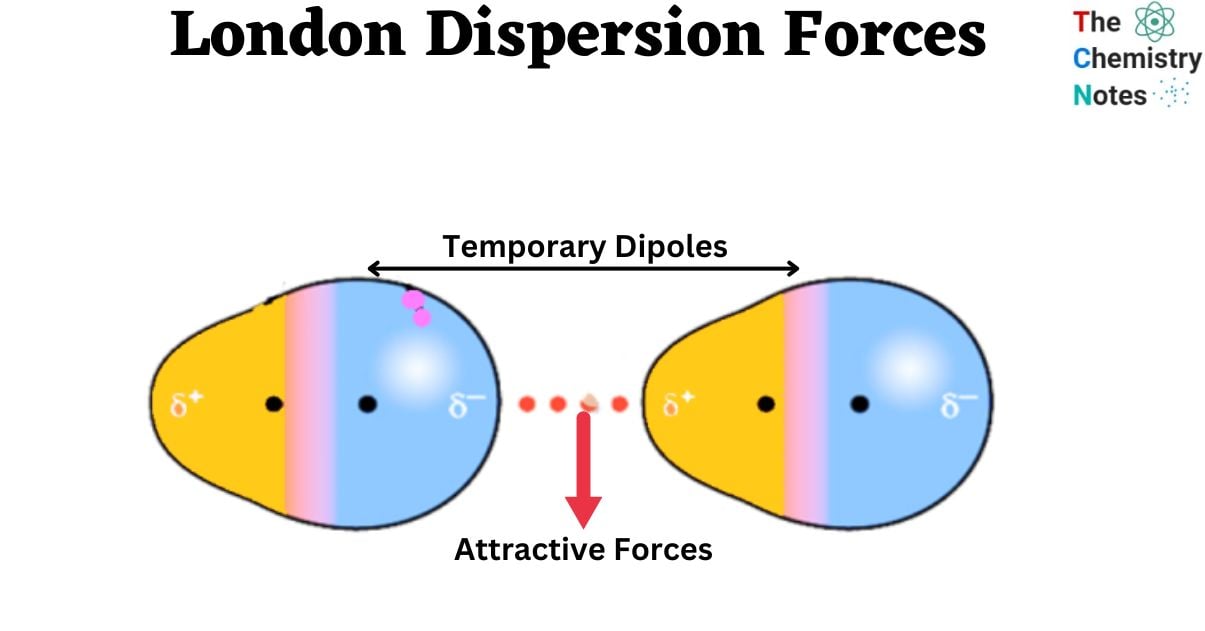
These forces arise from the constant, random motion of electrons within atoms and molecules. Even in molecules that are not permanently polar, the electron cloud is not static. At any given instant, the electron distribution can be uneven, creating a temporary, fleeting dipole – a region of slight positive charge and a region of slight negative charge. This instantaneous dipole can then influence the electron distribution in a neighboring atom or molecule.

The Genesis of Dispersion Forces: Electron Dynamics
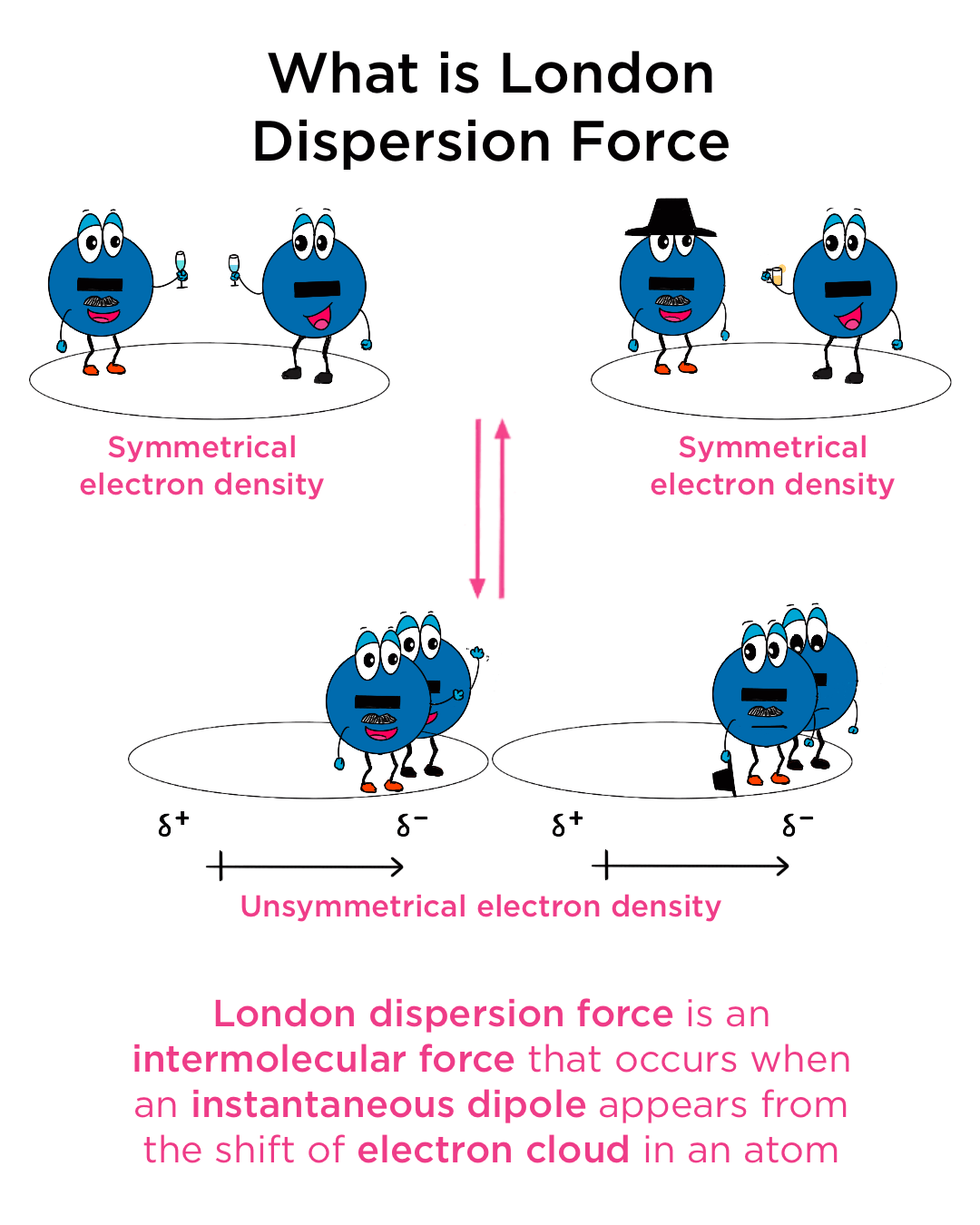
The very existence of dispersion forces is rooted in the dynamic nature of electrons. Electrons, being negatively charged particles, are in perpetual motion around the nucleus of an atom or within the electron cloud of a molecule. This motion is not perfectly uniform. At any infinitesimal point in time, there’s a probability that more electrons will be found on one side of an atom or molecule than on the other. This fluctuating asymmetry in electron distribution leads to the formation of transient, instantaneous dipoles.
Instantaneous Dipoles: A Fleeting Polarity
Imagine a nonpolar molecule, such as methane (CH4) or a diatomic molecule like oxygen (O2). Ideally, these molecules have no permanent separation of charge. However, due to the quantum mechanical nature of electron movement, the electron cloud is constantly shifting. At any given moment, there might be a slight excess of electron density on one side of the molecule and a corresponding deficit on the other. This creates a temporary, instantaneous dipole moment. This dipole is fleeting, lasting only for a fraction of a second before the electrons redistribute themselves.
Induced Dipoles: The Ripple Effect
The significance of these instantaneous dipoles lies in their ability to influence their surroundings. When an instantaneous dipole forms in one molecule, the electric field it generates can distort the electron cloud of a neighboring molecule. If the neighboring molecule is also nonpolar, this electric field will pull or push its electrons, inducing a complementary dipole in that molecule. The negative end of the instantaneous dipole will attract the electrons in the neighboring molecule, creating a negative pole on that side. Conversely, the positive end of the instantaneous dipole will repel electrons, creating a positive pole on the opposite side. This process is known as dipole induction, and the resulting temporary dipole is an induced dipole.
The induced dipole in the second molecule will align itself such that its positive end is attracted to the negative end of the instantaneous dipole in the first molecule, and its negative end is attracted to the positive end of the first molecule. This attraction between the instantaneous dipole and the induced dipole is the dispersion force. It’s a weak, short-range attraction that arises from this synchronized, albeit temporary, alignment of charges.
Factors Influencing the Strength of Dispersion Forces
While dispersion forces are present in all molecules, their strength can vary significantly. Several key factors influence the magnitude of these interactions, making them a critical consideration in predicting the macroscopic properties of substances.
Molecular Size and Number of Electrons: The Larger, the Stronger
One of the most significant factors governing the strength of dispersion forces is the size of the molecule, or more precisely, the number of electrons it possesses. Larger molecules, or molecules with a greater number of electrons, generally exhibit stronger dispersion forces. This is because a larger electron cloud is more diffuse and more easily distorted. With more electrons, there’s a greater probability of forming larger and longer-lasting instantaneous dipoles.
Consider the halogens: fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). Fluorine, the smallest, is a gas at room temperature. Chlorine is also a gas. Bromine is a liquid, and iodine is a solid. This trend in physical states directly correlates with the increasing number of electrons and molecular size. As the number of electrons increases down the group, the electron clouds become more polarizable, leading to stronger dispersion forces that require more energy (higher temperature) to overcome, thus increasing the boiling point and melting point.
Molecular Shape: Surface Area Matters
The shape of a molecule also plays a crucial role in the effectiveness of dispersion forces. Molecules with a larger surface area generally experience stronger dispersion forces compared to more compact, spherical molecules of similar molecular weight. This is because the greater the contact area between molecules, the more opportunity there is for the instantaneous and induced dipoles to interact.
For instance, compare n-pentane and neopentane. Both are isomers of C5H12 and have the same molecular weight and number of electrons. However, n-pentane is a linear molecule, while neopentane is spherical. N-pentane has a higher boiling point (36.1 °C) than neopentane (9.5 °C). This difference is attributed to the greater surface area of n-pentane, which allows for more extensive interactions via dispersion forces between adjacent molecules. The linear shape facilitates closer packing and a more effective overlap of electron clouds, leading to stronger attractions.
Polarizability: The Ease of Distortion
The term “polarizability” refers to the ease with which the electron cloud of an atom or molecule can be distorted by an external electric field. As mentioned earlier, this distortion is the basis of induced dipoles. Atoms and molecules with highly polarizable electron clouds will experience stronger dispersion forces. Factors that increase polarizability include a larger number of electrons, a larger atomic or molecular radius, and a loosely held outermost electron shell.
Elements in lower periods of the periodic table, with their more diffuse and less tightly bound valence electrons, are generally more polarizable than elements in higher periods. Similarly, larger molecules, having a greater electron cloud volume, are typically more polarizable than smaller ones, as the outer electrons are further from the nucleus and experience a weaker attractive force. This increased polarizability directly translates to stronger instantaneous and induced dipoles, and consequently, more significant dispersion forces.
The Significance of Dispersion Forces in Chemistry and Beyond
Despite their inherent weakness, dispersion forces are far from insignificant. Their pervasive nature and collective strength make them vital in dictating the physical properties of countless substances and influencing a wide array of chemical and biological processes.
Boiling and Melting Points: The Foundation of Phase Transitions
Perhaps the most direct and observable consequence of dispersion forces is their influence on the boiling and melting points of substances. For nonpolar molecules, dispersion forces are the only intermolecular forces present. Therefore, the energy required to overcome these forces to transition from solid to liquid (melting) or liquid to gas (boiling) is directly proportional to the strength of the dispersion forces. As discussed with the halogens and alkanes, molecules with stronger dispersion forces have higher boiling and melting points.
Even in polar molecules, where dipole-dipole forces and hydrogen bonding may be present, dispersion forces still contribute to the overall intermolecular attractions. In many cases, particularly with larger polar molecules, dispersion forces can become the dominant force, significantly impacting their phase transition temperatures.
Solubility: The “Like Dissolves Like” Principle
The principle of “like dissolves like” often applies to solubility, and dispersion forces are a key factor in this phenomenon. Nonpolar solvents tend to dissolve nonpolar solutes because both have similar intermolecular forces, primarily dispersion forces. The dispersion forces between the solute and solvent molecules are comparable to the dispersion forces within the pure solute and pure solvent, allowing for mixing.
Conversely, polar solvents, with their dipole-dipole interactions or hydrogen bonding, effectively dissolve polar solutes. Attempting to dissolve a highly polar solute in a nonpolar solvent, or vice versa, is usually unsuccessful because the energy required to break the strong intermolecular forces in one substance cannot be compensated by the weak dispersion forces that would form between the dissimilar molecules.
Biological Interactions: A Subtle but Crucial Role
In the realm of biology, dispersion forces, while individually weak, play a surprisingly critical role in many essential processes. The folding of proteins, the binding of enzymes to their substrates, the interaction of DNA base pairs, and the stability of cell membranes all involve a complex interplay of intermolecular forces, with dispersion forces contributing to the overall attraction.
For example, the hydrophobic effect, a major driving force in protein folding and the formation of lipid bilayers in cell membranes, is largely a consequence of dispersion forces. Nonpolar molecules (like hydrocarbon chains) tend to aggregate in aqueous environments to minimize their contact with water. This aggregation is driven by the fact that the dispersion forces between the nonpolar molecules are stronger than the interactions they would have with water molecules, and the release of ordered water molecules into the bulk solvent leads to an increase in entropy, further favoring the aggregation.
Other Applications: From Lubrication to Material Science
The understanding of dispersion forces extends to various practical applications. In lubrication, the ability of molecules to slide past each other easily is influenced by the strength of intermolecular forces. Weaker dispersion forces can lead to better lubrication. In material science, the adhesion between surfaces, the properties of polymers, and the formation of thin films are all influenced by dispersion forces. For instance, the tackiness of adhesives is often related to the strength of dispersion forces between the adhesive and the surface it adheres to.
In conclusion, dispersion forces, though the weakest of intermolecular attractions, are fundamental to understanding the physical world around us. Their origin in the dynamic nature of electrons, their dependence on molecular size and shape, and their pervasive influence on phase transitions, solubility, and even biological processes underscore their profound importance in chemistry and beyond. Recognizing and appreciating these subtle yet powerful forces provides a deeper insight into the molecular basis of matter.
